Abstract
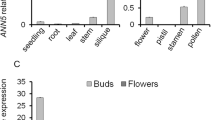
Cysteine proteases play important roles in intracellular protein degradation, programmed cell death and responses to environmental stimuli in plant. Although subclassification and biochemical analysis of major plant papain-like cysteine proteases (PLCPs) have been studied, the biological function of many PLCPs remained unknown. In this study, we identified a PLCP gene Cysteine Protease 51 (CP51) which participates in exine formation and anther development in Arabidopsis thaliana. Promoter-GUS fusion detection showed its specific expression in anthers at stages 9–12. RNA interference (RNAi) transgenic plants with reduced CP51 transcriptional levels exhibited a male sterile phenotype with aborted microspores, shortened siliques and fewer or no seeds. Cytological analysis indicated that pollen abortion occurred due to defective pollen exine and the tapetum degraded earlier during the transition from the uninucleated stage to the binucleated stage. Scanning electron microscopy demonstrated that aborted microspores lacked complete or normal reticulate exine, and the intine membrane was extruded in the pollens of CP51-RNAi plants. Transmission electron microscopy further revealed that the tapetum degeneration was initiated early and that normal tectum connections to the bacula were missing in anthers of CP51-RNAi plants. Taken together, these results suggested that CP51 critically mediates tapetum stability and pollen exine formation.








Similar content being viewed by others
Abbreviations
- PLCPs:
-
Papain-like cysteine proteases
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- PCD:
-
Programmed cell death
- WT:
-
Wild-type
- GUS:
-
β-Glucuronidase
- Kan:
-
Kanamycin
- NPTII :
-
Neomycin phosphotransferase
- HPT :
-
Hygromycin B phosphotransferase
- DAPI:
-
4′,6′-Diamidino-2-phenylindole
- ORF:
-
Open reading frame
References
Alexander MP (1969) Differential staining of aborted and nonaborted pollen. Stain Technol 44:117–122
Ariizumi T, Toriyama K (2011) Genetic regulation of sporopollenin synthesis and pollen exine development. Annu Rev Plant Biol 62:437–460
Bate N, Twell D (1998) Functional architecture of a late pollen promoter: pollen-specific transcription is developmentally regulated by multiple stage-specific and codependent activator elements. Plant Mol Biol 37:859–869
Bedinger P (1992) The remarkable biology of pollen. Plant Cell 4:879–889
Beers EP, Jones AM, Dickerman AW (2004) The S8 serine, C1A cysteine and A1 aspartic protease families in Arabidopsis. Phytochemistry 65:43–58
Beyene G, Foyer CH, Kunert KJ (2006) Two new cysteine proteinases with specific expression patterns in mature and senescent tobacco (Nicotiana tabacum L.) leaves. J Exp Bot 57:1431–1443
Blackmore S, Wortley AH, Skvarla JJ, Rowley JR (2007) Pollen wall development in flowering plants. New Phytol 174:483–498
Chang HS, Zhang C, Chang YH, Zhu J, Xu XF, Shi ZH, Zhang XL, Ling Xu, Huang H, Zhang S, Yang ZN (2012) No primexine and plasma membrane undulation is essential for primexine deposition and plasma membrane undulation during microsporogenesis in Arabidopsis. Plant Physiol 158:264–272
Chen YN, Lei SL, Zhou ZF, Zeng FQ, Yi B et al (2009) Analysis of gene expression profile in pollen development of recessive genic male sterile Brassica napus L. line S45A. Plant Cell Rep 28:1363–1372
Chen LQ, Hou BH, Lalonde S et al (2010) Sugar transporters for intercellular exchange and nutrition of pathogens. Nature 468:527–532
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
DeGuzman R, Riggs CD (2000) A survey of proteinases active during meiotic development. Planta 210:921–924
Diaz-Mendoza M, Velasco-Arroyo B, Gonzalez-Melendi P, Martinez M, Diaz I (2014) C1A cysteine protease-cystatin interactions in leaf senescence. J Exp Bot. doi:10.1093/jxb/eru043
Dobritsa AA, Lei Z, Nishikawa S, Urbanczyk-Wochniak E, Huhman DV, Preuss D, Sumner LW (2010) LAP5 and LAP6 encode anther-specific proteins with similarity to chalcone synthase essential for pollen exine development in Arabidopsis. Plant Physiol 153:937–955
Dun XL, Zhou ZF, Xia SQ, Wen J, Yi B, Shen JX, Ma CZ, Tu JX, Fu TD (2011) BnaC.Tic40, a plastid inner membrane translocon originating from Brassica oleracea, is essential for tapetal function and microspore development in Brassica napus. Plant J 68:532–545
Fan J, Yang YW, Gao X, Deng W, Falara V, Kanellis AK, Li ZG (2009) Expression of a senescence-associated cysteine protease gene related to peel pitting of navel orange (Citrus sinensis L. Osbeck). Plant Cell Tissue Cult 98:281–289
Goldberg RB, Beals TP, Sanders PM (1993) Anther development: basic principles and practical applications. Plant Cell 5:1217–1229
Guan YF, Huang XY, Zhu J, Gao JF, Zhang HX, Yang ZN (2008) RUPTURED POLLEN GRAIN1, a member of the MtN3/saliva gene family, is crucial for exine pattern formation and cell integrity of microspores in Arabidopsis. Plant Physiol 147:852–863
Guerrero C, Calle M, Reid MS, Valpuesta V (1998) Analysis of the expression of two thioprotease genes from day lily (Hemerocallis spp.) during flower senescence. Plant Mol Biol 36:565–571
Heslop-Harrison J (1971) Wall pattern formation in angiosperm microsporogenesis. Symp Soc Exp Biol 25:277–300
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database. Nucleic Acids Res 27:297–300
Ho SL, Tong WF, Yu SM (2000) Multiple mode regulation of a cysteine proteinase gene expression in rice. Plant Physiol 122:57–66
Ito T, Shinozaki K (2002) The MALE STERILITY1 gene of Arabidopsis, encoding a nuclear protein with a PHD-finger motif, is expressed in tapetal cells and is required for pollen maturation. Plant Cell Physiol 43:1285–1292
Ito T, Nagata N, Yoshiba Y, Ohme-Takagi M, Ma H, Shinozaki K (2007) Aabidopsis MALE STERILITY1 encodes a PHD-type transcription factor and regulates pollen and tapetum development. Plant Cell 19:3549–3562
Kamphuis IG, Drenth J, Baker EN (1985) Thiol proteases: comparative studies based on the high-resolution structures of papain and actinidin, and on amino acid sequence information from cathepsins B and H, and stem bromelain. J Mol Biol 182:317–329
Kapoor S, Kobayashi A, Takatsuji H (2002) Silencing of the tapetum-specific zinc finger gene TAZ1 causes premature degeneration of tapetum and pollen abortion in Petunia. Plant Cell 14:2353–2367
Karrer KM, Peiffer SL, Ditomas ME (1993) Two distinct gene subfamilies within the family of cysteine protease genes. Proc Natl Acad Sci USA 90:3063–3067
Kim YJ, Yeu SY, Park BS, Koh HJ, Song HT, Seo HS (2012) Protein disulfide isomerase-like protein 1-1 controls endosperm development through regulation of the amount and composition of seed proteins in rice. PLoS ONE 7:e44493
Koizumi M, Yamaguchi-Shinozaki K, Tsuji H, Shinozaki K (1993) Structure and expression of two genes that encode distinct drought-inducible cysteine proteinases in Arabidopsis thaliana. Gene 129:175–182
Ku S, Yoon H, Suh HS, Chung YY (2003) Male-sterility of thermosensitive genic male-sterile rice is associated with premature programmed cell death of the tapetum. Planta 217:559–565
Lee S, Jung KH, An G, Chung YY (2004) Isolation and characterization of a rice cysteine protease gene, OsCP1, using T-DNA gene trap system. Plant Mol Biol l54:755–765
Li N, Zhang DS, Liu HS, Yin CS, Li XX, Lian WQ, Yuan Z, Xu B, Chu HW, Jia Wang J, Wen TQ, Huang H, Luo D, Ma H, Zhang DB (2006) The rice tapetum degeneration retardation gene is required for tapetum degradation and anther development. Plant Cell 18:2999–3014
Li XW, Gao XQ, Wei Y, Deng L, Ouyang YD, Chen GX, Li XH, Zhang QF, Wu CY (2011) Rice APOPTOSIS INHIBITOR 5 coupled with two DEAD-box adenosine 5′-triphosphate-dependent RNA helicases regulates tapetum degeneration. Plant Cell 23:1416–1434
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−△△Ct method. Methods 25:402–408
Martinez M, Cambra I, Carrillo L, Mercedes DM, Diaz I (2009) Characterization of the entire cystatin gene family in Barley and their target cathepsin L-like cysteine-proteases, partners in the hordein mobilization during seed germination. Plant Physiol 151:1531–1545
McLellan H, Gilroy EM, Yun B-W, Birch PRJ, Loake GJ (2009) Functional redundancy in the Arabidopsis cathepsin B gene family contributes to basal defence, the hypersensitive response and senescence. New Phytol 183:408–418
Meyers BC, Lee DK, Vu TH, Tej SS, Edberg SB, Matvienko M, Tindell LD (2004) Arabidopsis MPSS. An online resource for quantitative expression analysis. Plant Physiol 135:801–813
Parish RW, Li SF (2010) Death of a tapetum: a programme of developmental altruism. Plant Sci 178:73–89
Paxson-Sowders DM, Owen HA, Makaroff CA (1997) A comparative ultrastructural analysis of exine pattern development in WT Arabidopsis and a mutant defective in pattern formation. Protoplasma 198:53–65
Piffanelli P, Ross JHE, Murphy DJ (1998) Biogenesis and function of the lipidic structures of pollen grains. Sex Plant Reprod 11:65–80
Qin P, Tu B, Wang Y, Deng L, Quilichini TD, Li T, Wang H, Ma B, Li S (2013) ABCG15 encodes an ABC transporter protein, and is essential for post-meiotic anther and pollen exine development in rice. Plant Cell Physiol 54:138–154
Quilichini TD, Friedmann MC, Samuels AL, Douglas CJ (2010) ATP-Binding Cassette transporter G26 is required for male fertility and pollen exine formation in Arabidopsis. Plant Physiol 154:678–690
Rawlings ND, Barrett AJ, Bateman A (2010) MEROPS: the peptidase database. Nucleic Acids Res 38:227–233
Richau KH, Kaschani F, Verdoes M, Pansuriya TC, Niessen S et al (2012) Subclassification and biochemical analysis of plant papain-like cysteine proteases displays subfamily-specific characteristics. Plant Physiol 158:1583–1599
Rogers HJ, Bate N, Combe J, Sullivan J, Sweetman J, Swan C, Lonsdale DM, Twell D (2001) Functional analysis of cis-regulatory elements within the promoter of the tobacco late pollen gene g10. Plant Mol Biol 45:577–585
Ross KJ, Fransz P, Jones GH (1996) A light microscopic atlas of meiosis in Arabidopsis thaliana. Chromosome Res 4:507–516
Sanders PM, Bui AQ, Weterings K, McIntire KN, Hsu Y-C, Lee PY, Truong MT, Beals TP, Goldberg RB (1999) Anther developmental defects in Arabidopsis thaliana male-sterile mutants. Sex Plant Reprod 11:297–322
Scott RJ, Spielman M, Dickinson HG (2004) Stamen structure and function. Plant Cell (Suppl) 16:S46–S60
Sessions A, Weigel D, Yanofsky MF (1999) The Arabidopsis thaliana MERISTEM LAYER 1 promoter specifies epidermal expression in meristems and young primordia. Plant J 20:259–263
Shutov AD, Vaintrub JA (1987) Degradation of storage proteins in germinating seeds. Phytochemistry 26:1557–1566
Solomon M, Beleghi B, Delledonne M, Menachem E, Levine A (1999) The involvement of cysteine proteases and protease inhibitor genes in the regulation of programmed cell death in plants. Plant Cell 11:431–443
Sorensen A, Krober S, Unte US, Huijser P, Dekker K, Saedler H (2003) The Arabidopsis ABORTED MICROSPORES (AMS) gene encodes a MYC class transcription factor. Plant J 33:413–423
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Van der Hoorn RAL (2008) Plant proteases: from phenotypes to molecular mechanism. Annu Rev Plant Biol 59:191–223
Vizcay-Barrena G, Wilson ZA (2006) Altered tapetal PCD and pollen wall development in the Arabidopsis ms1 mutant. J Exp Bot 57:2709–2717
Wesley SV, Helliwell CA, Smith NA, Wang MB, Rouse DT et al (2001) Construct design for efficient, effective and high-throughput gene silencing in plants. Plant J 27:581–590
Wilson ZA, Morroll SM, Dawson J, Swarup R, Tighe PJ (2001) The Arabidopsis MALE STERILITY1 (MS1) gene is a transcriptional regulator of male gametogenesis, with homology to the PHD-finger family of transcription factors. Plant J 28:27–39
Yang C, Vizcay-Barrena G, Conner K, Wilson ZA (2007) MALE STERILITY1 is required for tapetal development and pollen wall biosynthesis. Plant Cell 19:3530–3548
Yi B, Zeng FQ, Lei SL, Yao XQ, Zhu Y, Wen J et al (2010) Two duplicate CYP704B1 homologous genes BnMs1 and BnMs2 are required for pollen exine formation and tapetal development in Brassica napus. Plant J 63:925–938
Zhang XM, Wang Y, Lv XM, Li H, Sun P, Lu H, Li FI (2009) NtCP56, a new cysteine protease in Nicotiana tabacum L., involved in pollen grain development. J Exp Bot 60:1569–1577
Zhou ZF, Dun XL, Xia SQ, Shi DY, Qin MM et al (2012) BnMs3 is required for tapetal differentiation and degradation, microspore separation, and pollen-wall biosynthesis in Brassica napus. J Exp Bot 63:2041–2058
Zhu L, Shi JX, Zhao GC, Zhang DB, Liang WQ (2013) Post-meiotic Deficient Anther1 (PDA1) encodes an ABC transporter required for the development of anther cuticle and pollen exine in rice. J Plant Biol 56:59–68
Zimmermann P, Hirsch-Hoffmann M, Hennig L, Gruissem W (2004) Genevestigator. Arabidopsis microarray database and analysis toolbox. Plant Physiol 136:2621–2632
Zinkl GM, Zwiebel BI, Grier DG, Preuss D (1999) Pollen-stigma adhesion in Arabidopsis: a species-specific interaction mediated by lipophilic molecules in the pollen exine. Develop 126:5431–5440
Acknowledgments
We thank Jianbo Cao (Huazhong Agricultural University, China) for help with scanning electron microscopy and transmission electron microscope. This work was supported by the National Basic Research Program of China (2011CB109305), the State High-Tech Development Plan of China (2013AA102602), Commonweal Specialized Research Fund of China Agriculture (201103016), Innovation Program of Wuhan Academy of Agricultural Science and Technology (CX201242) and the Hubei Agriculture Science and Technology Innovation Center.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yongxue Yang and Caihua Dong have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yang, Y., Dong, C., Yu, J. et al. Cysteine Protease 51 (CP51), an anther-specific cysteine protease gene, is essential for pollen exine formation in Arabidopsis . Plant Cell Tiss Organ Cult 119, 383–397 (2014). https://doi.org/10.1007/s11240-014-0542-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-014-0542-0