Abstract
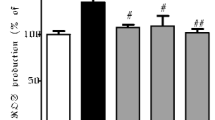
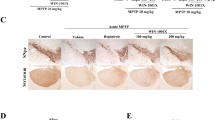
Coriandrum sativum L. (coriander), which is an annual herb of the Apiaceae family, has been traditionally used as a remedy. Here we tested whether heated extract of coriander leaf protects nigral dopaminergic neurodegeneration after exposure to 6-hydroxydopamine (6-OHDA). After injection of 6-OHDA into the rat substantia nigra pars compacta (SNpc), dopaminergic degeneration, which was determined by tyrosine hydroxylase immunostaining, was rescued by co-injection of CaEDTA, an extracellular Zn2+ chelator, suggesting that extracellular Zn2+ influx is involved in neurodegeneration. Both intracellular Zn2+ dysregulation determined by ZnAF-2 fluorescence and dopaminergic degeneration in the SNpc induced by 6-OHDA were rescued by co-injection of 0.25% coriander extract, which also reduced reactive oxygen species (ROS) production in the SNpc determined by aminophenyl fluorescein fluorescence. The present study suggests that coriander leaf extract protects nigral dopaminergic neurodegeneration induced by intracellular Zn2+ dysregulation. It is likely that the nutraceutical property of coriander leaf extract contributes to the protection via reducing ROS production.




Similar content being viewed by others
Data availability
Not applicable.
References
Kouli A, Torsney KM, Kuan WL, Thomas BS, Greenland JC (2018) In: Parkinson’s Disease: Pathogenesis and Clinical Aspects [Internet]. Brisbane (AU): Codon Publications, pp 3–26. https://doi.org/10.15586/codonpublications.parkinsonsdisease.2018
Rodriguez-Pallares J, Parga JA, Joglar B, Guerra MJ, Labandeira-Garcia JL (2009) The mitochondrial ATP-sensitive potassium channel blocker 5-hydroxydecanoate inhibits toxicity of 6-hydroxydopamine on dopaminergic neurons. Neurotox Res 15:82–95. https://doi.org/10.1007/s12640-009-9010-8
Tamano H, Nishio R, Morioka H, Takeda A (2019) Extracellular Zn2+ influx into nigral dopaminergic neurons plays a key role for pathogenesis of 6-hydroxydopamine-induced Parkinson’s disease in rats. Mol Neurobiol 56:435–443. https://doi.org/10.1007/s12035-018-1075-z
Nishio R, Morioka H, Takeuchi A, Saeki N, Furuhata R, Katahira M, Chinenn T, Tamura H, Tamano H, Takeda A (2022) Intracellular hydrogen peroxide produced by 6-hydroxydopamine is a trigger for nigral dopaminergic degeneration of rats via rapid influx of extracellular Zn2+. Neurotoxicology 89:1–8. https://doi.org/10.1016/j.neuro.2021
Laribi B, Kouki K, M'Hamdi M, Bettaieb T (2015) Coriander (Coriandrum sativum L.) and its bioactive constituents. Fitoterapia. 2015 103:9–26. https://doi.org/10.1016/j.fitote.2015.03.012
Gazwi HSS, Mahmoud ME, Toson EMA (2022) Analysis of the phytochemicals of Coriandrum sativum and Cichorium intybus aqueous extracts and their biological effects on broiler chickens. Sci Rep 12:6399. https://doi.org/10.1038/s41598-022-10329-2
Verma T, Sinha M, Bansal N, Yadav SR, Shah K, Chauhan NS (2021) Plants used as antihypertensive. Nat Prod Bioprospect 11:155–184. https://doi.org/10.1007/s13659-020-00281-x
Gray AM, Flatt PR (1999) Insulin-releasing and insulin-like activity of the traditional anti-diabetic plant Coriandrum sativum (coriander). Br J Nutr 81:203–209. https://doi.org/10.1017/S0007114599000392
Cioanca O, Hritcu L, Mihasan M, Hancianu M (2013) Cognitive-enhancing and antioxidant activities of inhaled coriander volatile oil in amyloid β (1–42) rat model of Alzheimer’s disease. Physiol Behav 120:193–202. https://doi.org/10.1016/j.physbeh.2013.08.006
Nishio R, Tamano H, Morioka H, Takeuchi A, Takeda A (2019) Intake of heated leaf extract of Coriandrum sativum contributes to resistance to oxidative stress via decreases in heavy metal concentrations in the kidney. Plant Foods Hum Nutr 74:204–209. https://doi.org/10.1007/s11130-019-00720-2
Kim EY, Koh JY, Kim YH, Sohn S, Joe E, Gwag BJ (1999) Zn2+ entry produces oxidative neuronal necrosis in cortical cell cultures. Eur J Neurosci 11:327–334. https://doi.org/10.1046/j.1460-9568.1999.00437.x
Kim YH, Eom JW, Koh JY (2020) Mechanism of zinc excitotoxicity: a focus on AMPK. Front Neurosci 14:577958. https://doi.org/10.3389/fnins.2020.577958
Sensi SL, Paoletti P, Bush AI, Sekler I (2009) Zinc in the physiology and pathology of the CNS. Nat Rev Neurosci 10:780–791. https://doi.org/10.1038/nrn2734
Medvedeva YV, Ji SG, Yin HZ, Weiss JH (2017) Differential vulnerability of CA1 versus CA3 pyramidal neurons after ischemia: possible relationship to sources of Zn2+ accumulation and its entry into and prolonged effects on mitochondria. J Neurosci 37:726–737. https://doi.org/10.1523/JNEUROSCI.3270-16.2016
Ji SG, Medvedeva YV, Wang HL, Yin HZ, Weiss JH (2019) Mitochondrial Zn 2+ accumulation: a potential trigger of hippocampal ischemic injury. Neuroscientist 25:126–138. https://doi.org/10.1177/1073858418772548
Ji SG, Medvedeva YV, Weiss JH (2020) Zn 2+ entry through the mitochondrial calcium uniporter is a critical contributor to mitochondrial dysfunction and neurodegeneration. Exp Neurol 325:113161. https://doi.org/10.1016/j.expneurol.2019.113161
Noh KM, Koh JY (2000) Induction and activation by zinc of NADPH oxidase in cultured cortical neurons and astrocytes. J Neurosci 20:RC111. https://doi.org/10.1523/JNEUROSCI.20-23-j0001.2000
Kabuto H, Yamanushi TT (2011) Effects of zingerone [4-(4-hydroxy-3-methoxyphenyl)-2-butanone] and eugenol [2-methoxy-4-(2-propenyl)phenol] on the pathological progress in the 6-hydroxydopamine-induced Parkinson’s disease mouse model. Neurochem Res 36:2244–2249. https://doi.org/10.1007/s11064-011-0548-5
Bigham M, Mohammadipour A, Hosseini M, Malvandi AM, Ebrahimzadeh-Bideskan A (2021) Neuroprotective effects of garlic extract on dopaminergic neurons of substantia nigra in a rat model of Parkinson’s disease: motor and non-motor outcomes. Metab Brain Dis 36:927–937. https://doi.org/10.1007/s11011-021-00705-8
Koppula S, Alluri R, Kopalli SR (2021) Coriandrum sativum attenuates microglia mediated neuroinflammation and MPTP-induced behavioral and oxidative changes in Parkinson's disease mouse model. EXCLI J 20:835–850. https://doi.org/10.17179/excli2021-3668
Funding
The authors received no funding in the present paper. The present paper contains non-financial interests.
Author information
Authors and Affiliations
Contributions
Conceptualization; Atsushi Takeda: Data curation; Haruna Tamano: Formal analysis; Nana Saeki, Azusa Takeuchi, Misa Katahira, Ryusuke Nishio, Haruna Tamura: Methodology; Nana Saeki, Atsushi Takeda: Project administration; Atsushi Takeda: Resources; Atsushi Takeda: Software; Nana Saeki: Supervision; Haruna Tamano: Validation; Atsushi Takeda: Roles/Writing—original draft; Atsushi Takeda: Writing—review & editing; Atsushi Takeda.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
The Ethics Committee for Experimental Animals has permitted the present study in the University of Shizuoka.
Consent to Participate
The present paper has been approved by all named authors.
Consent for Publication
The present paper, which is original, has not been published before and is not currently being considered for publication elsewhere.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saeki, N., Tamano, H., Takeuchi, A. et al. Heated Leaf Extract of Coriandrum sativum L. Protects Nigral Dopaminergic Degeneration in Rats. Plant Foods Hum Nutr 77, 455–459 (2022). https://doi.org/10.1007/s11130-022-00999-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-022-00999-8