Abstract
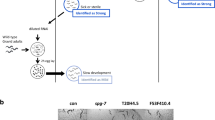
CLN3 disease (Spielmeyer-Vogt-Sjogren-Batten disease, previously known as classic juvenile neuronal ceroid lipofuscinosis, NCL) is a pediatric-onset progressive neurodegenerative disease characterized by progressive vision loss, seizures, loss of cognitive and motor function, and early death. While no precise biochemical mechanism or therapies are known, the pathogenesis of CLN3 disease involves intracellular calcium accumulation that may trigger apoptosis. Our prior work in in vitro cell models of CLN3 deficiency suggested that FDA-approved calcium channel antagonists may have therapeutic value. To further evaluate the potential efficacy of this approach in an otherwise untreatable disorder, we sought to compare the therapeutic effects and underlying mechanisms in an animal model of CLN3 disease. Here, we used the well-characterized XT7 complete cln-3 knockout strain of C. elegans to evaluate the therapeutic efficacy of calcium channel antagonist therapy in a living animal model of Batten disease. Therapeutic effects of five calcium channel antagonists were evaluated on XT7 animal lifespan and in vivo mitochondrial physiology. Remarkably, maximal therapeutic efficacy in this model animal was observed with 1 μM flunarizine, the identical concentration previously identified in cell-based neuronal models of CLN3 disease. Specifically, flunarizine rescued the short lifespan of XT7 worms and prevented their pathophysiologic mitochondrial accumulation. These results confirm the treatment efficacy and dosing of flunarizine in cln-3 disease in a translational model organism. Clinical treatment trials in CLN3 human patients are now needed to test the dosing regimen and efficacy of flunarizine in individuals suffering with this otherwise untreatable and ultimately lethal neurologic disease.



Similar content being viewed by others
References
Seehafer SS, Ramirez-Montealegre D, Wong AM et al (2011) Immunosuppression alters disease severity in juvenile Batten disease mice. J Neuroimmunol 230(1–2):169–172. doi:10.1016/j.jneuroim.2010.08.024
Drack AV, Mullins RF, Pfeifer WL, Augustine EF, Stasheff SF, Hong SD (2015) Immunosuppressive treatment for retinal degeneration in juvenile neuronal ceroid lipofuscinosis (Juvenile Batten Disease). Ophthalmic Genet 36(4):359–364. doi:10.3109/13816810.2014.886271
Haltia M (2003) The neuronal ceroid-lipofuscinoses. J Neuropathol Exp Neurol 62(1):1–13
Phillips SN, Benedict JW, Weimer JM, Pearce DA (2005) CLN3, the protein associated with batten disease: structure, function and localization. J Neurosci Res 79(5):573–583. doi:10.1002/jnr.20367
Rakheja D, Narayan SB, Bennett MJ (2008) The function of CLN3P, the Batten disease protein. Mol Genet Metab 93(3):269–274. doi:10.1016/j.ymgme.2008.01.001
Bennett MJ, Rakheja D (2013) The neuronal ceroid-lipofuscinoses. Dev Disabil Res Rev 17(3):254–259. doi:10.1002/ddrr.1118
Rakheja D, Narayan SB, Pastor JV, Bennett MJ (2004) CLN3P, the Batten disease protein, localizes to membrane lipid rafts (detergent-resistant membranes). Biochem Biophys Res Commun 317(4):988–991. doi:10.1016/j.bbrc.2004.03.146
Narayan SB, Rakheja D, Tan L, Pastor JV, Bennett MJ (2006a) CLN3P, the Batten’s disease protein, is a novel palmitoyl-protein Delta-9 desaturase. Ann Neurol 60(5):570–577. doi:10.1002/ana.20975
Lane SC, Jolly RD, Schmechel DE, Alroy J, Boustany RM (1996) Apoptosis as the mechanism of neurodegeneration in Batten’s disease. J Neurochem 67(2):677–683
Puranam KL, Guo W-X, Qian W-H, Nikbakht K, Boustany R-M (1999) CLN3 defines a novel antiapoptotic pathway operative in neurodegeneration and mediated by ceramide. Mol Genet Metab 66(4):294–308. doi:10.1006/mgme.1999.2834
Dhar S, Bitting RL, Rylova SN et al (2002) Flupirtine blocks apoptosis in batten patient lymphoblasts and in human postmitotic CLN3- and CLN2-deficient neurons. Ann Neurol 51(4):448–466
Persaud-Sawin D-ANW, VanDongen A, Boustany R-MN (2002) Motifs within the CLN3 protein: modulation of cell growth rates and apoptosis. Hum Mol Genet 11(18):2129–2142
Persaud-Sawin DA, Boustany R-MN (2005) Cell death pathways in juvenile Batten disease. Apoptosis 10(5):973–985. doi:10.1007/s10495-005-0733-6
Narayan SB, Rakheja D, Pastor JV et al (2006b) Over-expression of CLN3P, the Batten disease protein, inhibits PANDER-induced apoptosis in neuroblastoma cells: further evidence that CLN3P has anti-apoptotic properties. Mol Genet Metab 88(2):178–183. doi:10.1016/j.ymgme.2006.01.011
Chang J-W, Choi H, Kim H-J et al (2007) Neuronal vulnerability of CLN3 deletion to calcium-induced cytotoxicity is mediated by calsenilin. Hum Mol Genet 16(3):317–326. doi:10.1093/hmg/ddl466
An Haack K, Narayan SB, Li H, Warnock A, Tan L, Bennett MJ (2011) Screening for calcium channel modulators in CLN3 siRNA knock down SH-SY5Y neuroblastoma cells reveals a significant decrease of intracellular calcium levels by selected L-type calcium channel blockers. Biochim Biophys Acta 1810(2):186–191. doi:10.1016/j.bbagen.2010.09.004
Warnock A, Tan L, Li C, An Haack K, Narayan SB, Bennett MJ (2013) Amlodipine prevents apoptotic cell death by correction of elevated intracellular calcium in a primary neuronal model of Batten disease (CLN3 disease). Biochem Biophys Res Commun 436(4):645–649. doi:10.1016/j.bbrc.2013.04.113
de Voer G, van der Bent P, Rodrigues AJG, van Ommen G-JB, Peters DJM, Taschner PEM (2005) Deletion of the Caenorhabditis elegans homologues of the CLN3 gene, involved in human juvenile neuronal ceroid lipofuscinosis, causes a mild progeric phenotype. J Inherit Metab Dis 28(6):1065–1080. doi:10.1007/s10545-005-0125-7
McCormack S, Polyak E, Ostrovsky J et al (2015a) Pharmacologic targeting of sirtuin and PPAR signaling improves longevity and mitochondrial physiology in respiratory chain complex I mutant Caenorhabditis elegans. Mitochondrion 22:45–59. doi:10.1016/j.mito.2015.02.005
Dingley S, Polyak E, Lightfoot R et al (2010) Mitochondrial respiratory chain dysfunction variably increases oxidant stress in Caenorhabditis elegans. Mitochondrion 10(2):125–136. doi:10.1016/j.mito.2009.11.003
McCormack S, Polyak E, Ostrovsky J et al (2015b) Pharmacologic targeting of sirtuin and PPAR signaling improves longevity and mitochondrial physiology in respiratory chain complex I mutant Caenorhabditis elegans. Mitochondrion 22:45–59. doi:10.1016/j.mito.2015.02.005
Schindelin J, Arganda-Carreras I, Frise E et al (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9(7):676–682. doi:10.1038/nmeth.2019
Palmer DN, Martinus RD, Cooper SM, Midwinter GG, Reid JC, Jolly RD (1989) Ovine ceroid lipofuscinosis. The major lipopigment protein and the lipid-binding subunit of mitochondrial ATP synthase have the same NH2-terminal sequence. J Biol Chem 264(10):5736–5740
Elkayam U, Shotan A, Mehra A, Ostrzega E (1993) Calcium channel blockers in heart failure. J Am Coll Cardiol 22(4 Suppl A):139A–144A
Elkayam U (1998) Calcium channel blockers in heart failure. Cardiology 89(Suppl 1):38–46
de Vries RJ, van Veldhuisen DJ, Dunselman PHJ (2000) Efficacy and safety of calcium channel blockers in heart failure: focus on recent trials with second-generation dihydropyridines. Am Heart J 139(2):185–194. doi:10.1016/S0002-8703(00)90224-2
Trepakova ES, Dech SJ, Salata JJ (2006) Flunarizine is a highly potent inhibitor of cardiac hERG potassium current. J Cardiovasc Pharmacol 47(2):211–220. doi:10.1097/01.fjc.0000200810.18575.80
Cao K, Han F, Lin A et al (2016) Zhengtian Capsule versus flunarizine in patients with migraine: a multi-center, double-blind, double-dummy, randomized controlled, non-inferior clinical trial. BMC Complement Altern Med 16:356. doi:10.1186/s12906-016-1321-8
Lepcha A, Amalanathan S, Augustine AM, Tyagi AK, Balraj A (2014) Flunarizine in the prophylaxis of migrainous vertigo: a randomized controlled trial. Eur Arch Otorhinolaryngol 271(11):2931–2936. doi:10.1007/s00405-013-2786-4
Hasan M, Pulman J, Marson AG (2013) Calcium antagonists as an add-on therapy for drug-resistant epilepsy. Cochrane Database Syst Rev 3:CD002750. doi:10.1002/14651858.CD002750.pub2
Pessetto ZY, Weir SJ, Sethi G, Broward MA, Godwin AK (2013) Drug repurposing for gastrointestinal stromal tumor. Mol Cancer Ther 12(7):1299–1309. doi:10.1158/1535-7163.MCT-12-0968
Beaulieu CL, Samuels ME, Ekins S et al (2012) A generalizable pre-clinical research approach for orphan disease therapy. Orphanet J Rare Dis 7:39. doi:10.1186/1750-1172-7-39
Xu K, Coté TR (2011) Database identifies FDA-approved drugs with potential to be repurposed for treatment of orphan diseases. Brief Bioinform 12(4):341–345. doi:10.1093/bib/bbr006
Acknowledgments
We would like to thank P.E.M. Taschner for providing the XT7 strain used in this study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
None.
Informed Consent and Animal Rights
This article does not contain any studies with human or vertebrate animal subjects performed by the any of the authors.
Funding
This work was funded in part by the National Institutes of Health (R01-HD065858 to MJF). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Additional information
Communicated by: Robin Lachmann
Marni J. Falk and Michael J. Bennett contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kwon, Y.J., Falk, M.J. & Bennett, M.J. Flunarizine rescues reduced lifespan in CLN3 triple knock-out Caenorhabditis elegans model of batten disease. J Inherit Metab Dis 40, 291–296 (2017). https://doi.org/10.1007/s10545-016-9986-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-016-9986-1