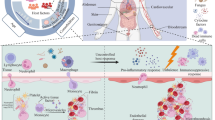
Abstract
There is a decades old association between cytomegalovirus reactivation and sepsis in immune-competent hosts. Much has been learned about this relationship, which has been described as bidirectional, meaning that the virus incites and is incited by the host’s inflammatory response. More recent work has suggested that chronic viral infection leaves the host with exaggerated immunity to bacterial infections. In this review, the relationship between CMV and host responses to sepsis are reviewed, with particular attention to the impact that tissue viral load contributes to this phenomenon.

Similar content being viewed by others
References
Bordes J, Maslin J, Prunet B, d'Aranda E, Lacroix G, Goutorbe P, Dantzer E, Meaudre E (2011) Cytomegalovirus infection in severe burn patients monitoring by real-time polymerase chain reaction: a prospective study. Burns 37(3):434–439. https://doi.org/10.1016/j.burns.2010.11.006
Heininger A, Haeberle H, Fischer I, Beck R, Riessen R, Rohde F, Meisner C, Jahn G, Koenigsrainer A, Unertl K, Hamprecht K (2011) Cytomegalovirus reactivation and associated outcome of critically ill patients with severe sepsis. Crit Care 15(2):R77. https://doi.org/10.1186/cc10069
Heininger A, Jahn G, Engel C, Notheisen T, Unertl K, Hamprecht K (2001) Human cytomegalovirus infections in nonimmunosuppressed critically ill patients. Crit Care Med 29(3):541–547
Chilet M, Aguilar G, Benet I, Belda J, Tormo N, Carbonell JA, Clari MA, Costa E, Navarro D (2010) Virological and immunological features of active cytomegalovirus infection in nonimmunosuppressed patients in a surgical and trauma intensive care unit. J Med Virol 82(8):1384–1391. https://doi.org/10.1002/jmv.21825
Ziemann M, Sedemund-Adib B, Reiland P, Schmucker P, Hennig H (2008) Increased mortality in long-term intensive care patients with active cytomegalovirus infection. Crit Care Med 36(12):3145–3150
Limaye AP, Kirby KA, Rubenfeld GD, Leisenring WM, Bulger EM, Neff MJ, Gibran NS, Huang M-L, Santo Hayes TK, Corey L, Boeckh M (2008) Cytomegalovirus reactivation in critically ill immunocompetent patients. JAMA 300(4):413–422. https://doi.org/10.1001/jama.300.4.413
von Muller L, Klemm A, Weiss M, Schneider M, Suger-Wiedeck H, Durmus N, Hampl W, Mertens T (2006) Active cytomegalovirus infection in patients with septic shock. Emerg Infect Dis 12(10):1517–1522
Kutza AS, Muhl E, Hackstein H, Kirchner H, Bein G (1998) High incidence of active cytomegalovirus infection among septic patients. Clin Infect Dis 26(5):1076–1082
Papazian L, Fraisse A, Garbe L, Zandotti C, Thomas P, Saux P, Pierrin G, Gouin F (1996) Cytomegalovirus. An unexpected cause of ventilator-associated pneumonia. Anesthesiology 84(2):280–287
Domart Y, Trouillet JL, Fagon JY, Chastre J, Brun-Vezinet F, Gibert C (1990) Incidence and morbidity of cytomegaloviral infection in patients with mediastinitis following cardiac surgery [see comments]. Chest 97(1):18–22
Walton AH, Muenzer JT, Rasche D, Boomer JS, Sato B, Brownstein BH, Pachot A, Brooks TL, Deych E, Shannon WD, Green JM, Storch GA, Hotchkiss RS (2014) Reactivation of multiple viruses in patients with sepsis. PLoS ONE 9(6):e98819. https://doi.org/10.1371/journal.pone.0098819
Coisel Y, Bousbia S, Forel J-M, Hraiech S, Lascola B, Roch A, Zandotti C, Million M, Jaber S, Raoult D, Papazian L (2012) Cytomegalovirus and herpes simplex virus effect on the prognosis of mechanically ventilated patients suspected to have ventilator-associated pneumonia. PLoS ONE 7(12):e51340. https://doi.org/10.1371/journal.pone.0051340
Smith CA, Conroy LT, Pollock M, Ruddy J, Binning A, McCruden EA (2010) Detection of herpes viruses in respiratory secretions of patients undergoing artificial ventilation. J Med Virol 82(8):1406–1409. https://doi.org/10.1002/jmv.21794
Jaber S, Chanques G, Borry J, Souche B, Verdier R, Perrigault P-F, Eledjam J-J (2005) Cytomegalovirus infection in critically Ill patients: associated factors and consequences. Chest 127(1):233–241
Friedrichs I, Bingold T, Keppler OT, Pullmann B, Reinheimer C, Berger A (2013) Detection of herpesvirus EBV DNA in the lower respiratory tract of ICU patients: a marker of infection of the lower respiratory tract? Med Microbiol Immunol 202(6):431–436. https://doi.org/10.1007/s00430-013-0306-1
Chiche L, Forel JM, Roch A, Guervilly C, Pauly V, Allardet-Servent J, Gainnier M, Zandotti C, Papazian L (2009) Active Cytomegalovirus infection is common in mechanically ventilated medical intensive care unit patients. Crit Care Med 37(6):1850–1857
Cook CH, Martin LC, Yenchar JK, Lahm MC, McGuinness B, Davies EA, Ferguson RM (2003) Occult herpes family viral infections are endemic in critically ill surgical patients. Crit Care Med 31(7):1923–1929
Cook CH, Yenchar JK, Kraner TO, Davies EA, Ferguson RM (1998) Occult herpes family viruses may increase mortality in critically ill surgical patients. Am J Surg 176(4):357–360
Desachy A, Ranger-Rogez S, Francois B, Venot C, Traccard I, Gastinne H, Denis F, Vignon P (2001) Reactivation of human herpesvirus type 6 in multiple organ failure syndrome. Clin Infect Dis 32(2):197–203
Razonable RR, Fanning C, Brown RA, Espy MJ, Rivero A, Wilson J, Kremers W, Smith TF, Paya CV (2002) Selective reactivation of human herpesvirus 6 variant a occurs in critically ill immunocompetent hosts [see comment]. J Infect Dis 185(1):110–113
Stephan F, Meharzi D, Ricci S, Fajac A, Clergue F, Bernaudin JF (1996) Evaluation by polymerase chain reaction of cytomegalovirus reactivation in intensive care patients under mechanical ventilation. Intensive Care Med 22(11):1244–1249
Vogel T, Vadonis R, Kuehn J, Eing BR, Senninger N, Haier J (2008) Viral reactivation is not related to septic complications after major surgical resections. APMIS 116(4):292–301
Cinel G, Pekcan S, Özçelik U, Alp A, Yalçın E, Doğru Ersöz D, Kiper N (2014) Cytomegalovirus infection in immunocompetent wheezy infants: the diagnostic value of CMV PCR in bronchoalveolar lavage fluid. J Clin Pharm Ther 39(4):399–403. https://doi.org/10.1111/jcpt.12169
Singh N, Inoue M, Osawa R, Wagener MM, Shinohara ML (2017) Inflammasome expression and cytomegalovirus viremia in critically ill patients with sepsis. J Clin Virol 93:8–14. https://doi.org/10.1016/j.jcv.2017.05.012
Ong DY, Spitoni C, Klein Klouwenberg PC, Verduyn Lunel F, Frencken J, Schultz M, van der Poll T, Kesecioglu J, Bonten MM, Cremer O (2016) Cytomegalovirus reactivation and mortality in patients with acute respiratory distress syndrome. Intensive Care Med 42(3):333–341. https://doi.org/10.1007/s00134-015-4071-z
Castón JJ, Cantisán S, González-Gasca F, Páez-Vega A, Abdel-Hadi H, Illescas S, Alonso G, Torre-Cisneros J (2016) Interferon-γ production by CMV-specific CD8+ T lymphocytes provides protection against cytomegalovirus reactivation in critically ill patients. Intensive Care Med 42(1):46–53. https://doi.org/10.1007/s00134-015-4077-6
Frantzeskaki FG, Karampi ES, Kottaridi C, Alepaki M, Routsi C, Tzanela M, Vassiliadi DA, Douka E, Tsaousi S, Gennimata V, Ilias I, Nikitas N, Armaganidis A, Karakitsos P, Papaevangelou V, Dimopoulou I (2015) Cytomegalovirus reactivation in a general, nonimmunosuppressed intensive care unit population: incidence, risk factors, associations with organ dysfunction, and inflammatory biomarkers. J Crit Care 30(2):276–281. https://doi.org/10.1016/j.jcrc.2014.10.002
Ong DSY, Bonten MJM, Spitoni C, Verduyn Lunel FM, Frencken JF, Horn J, Schultz MJ, van der Poll T, Klein Klouwenberg PMC, Cremer OL, Consortium ftMDaRSoS (2017) Epidemiology of multiple herpes viremia in previously immunocompetent patients with septic shock. Clin Infect Dis 64(9):1204–1210. https://doi.org/10.1093/cid/cix120
Kalil AC, Florescu DF (2009) Prevalence and mortality associated with cytomegalovirus infections in non-immunosuppressed ICU patients. Crit Care Med 37(8):2350–2358
Lachance P, Chen J, Featherstone R, Sligl WI (2017) Association between cytomegalovirus reactivation and clinical outcomes in immunocompetent critically ill patients: a systematic review and meta-analysis. Open Forum Infect Dis 4(2):ofx029. https://doi.org/10.1093/ofid/ofx029
Mansfield SA, Cook CH (2017) Antiviral prophylaxis of cytomegalovirus reactivation in immune competent patients-the jury remains out. J Thorac Dis 9(8):2221–2223. https://doi.org/10.21037/jtd.2017.06.130
Cowley NJ, Owen A, Shiels SC, Millar J, Woolley R, Ives N, Osman H, Moss P, Bion JF (2017) Safety and efficacy of antiviral therapy for prevention of cytomegalovirus reactivation in immunocompetent critically ill patients: a randomized clinical trial. JAMA Intern Med. https://doi.org/10.1001/jamainternmed.2017.0895
Limaye AP, Stapleton RD, Peng L, Gunn SR, Kimball LE, Hyzy R, Exline MC, Files DC, Morris PE, Frankel SK, Mikkelsen ME, Hite D, Enfield KB, Steingrub J, O'Brien J, Parsons PE, Cuschieri J, Wunderink RG, Hotchkin DL, Chen YQ, Rubenfeld GD, Boeckh M (2017) Effect of ganciclovir on IL-6 levels among cytomegalovirus-seropositive adults with critical illness: a randomized clinical trial. JAMA 318(8):731–740. https://doi.org/10.1001/jama.2017.10569
Seckert CK, Griessl M, Buttner JK, Freitag K, Lemmermann N, Hummel M, Liu XF, Abecassis M, Angulo A, Messerle M, Cook CH, Reddehase M (2013) Immune surveillance of cytomegalovirus latency and reactivation in murine models: link to memory inflation. In: Reddehase MJ (ed) Cytomegaloviruses. Caister Academic Press 1, Norfolk, pp 374–416
Dorsch-Hasler K, Keil GM, Weber F, Jasin M, Schaffner W, Koszinowski UH (1985) A long and complex enhancer activates transcription of the gene coding for the highly abundant immediate early mRNA in murine cytomegalovirus. Proc Natl Acad Sci USA 82(24):8325–8329
Liu B, Stinski MF (1992) Human cytomegalovirus contains a tegument protein that enhances transcription from promoters with upstream ATF and AP-1 cis-acting elements. J Virol 66(7):4434–4444
Cornell TT, Wynn J, Shanley TP, Wheeler DS, Wong HR (2010) Mechanisms and regulation of the gene-expression response to sepsis. Pediatrics 125(6):1248–1258. https://doi.org/10.1542/peds.2009-3274
Li Y, Alam HB (2011) Modulation of acetylation: creating a pro-survival and anti-inflammatory phenotype in lethal hemorrhagic and septic shock. J Biomed Biotechnol 2011:523481. https://doi.org/10.1155/2011/523481
Docke WD, Prosch S, Fietze E, Kimel V, Zuckermann H, Klug C, Syrbe U, Kruger DH, von Baehr R, Volk HD (1994) Cytomegalovirus reactivation and tumour necrosis factor. Lancet 343(8892):268–269
Prosch S, Staak K, Stein J, Liebenthal C, Stamminger T, Volk HD, Kruger DH (1995) Stimulation of the human cytomegalovrus IE enhancer/promoter in HL-60 Cells by TNFalpha is mediated via induction of NF-kappaB. Virology 208(1):197–206
Stein J, Volk HD, Liebenthal C, Kruger DH, Prosch S (1993) Tumour necrosis factor alpha stimulates the activity of the human cytomegalovirus major immediate early enhancer/promoter in immature monocytic cells. J Gen Virol 74(11):2333–2338
Kline JN, Hunninghake GM, He B, Monick MM, Hunninghake GW (1998) Synergistic activation of the human cytomegalovirus major immediate early promoter by prostaglandin E2 and cytokines. Exp Lung Res 24(1):3–14
Hunninghake GW, Monick MM, Liu B, Stinski MF (1989) The promoter-regulatory region of the major immediate-early gene of human cytomegalovirus responds to T-lymphocyte stimulation and contains functional cyclic AMP-response elements. J Virol 63(7):3026–3033
Laegreid A, Medvedev A, Nonstad U, Bombara MP, Ranges G, Sundan A, Espevik T (1994) Tumor necrosis factor receptor p75 mediates cell-specific activation of nuclear factor kappa B and induction of human cytomegalovirus enhancer. J Biol Chem 269(10):7785–7791
Cook C, Zhang X, McGuinness B, Lahm M, Sedmak D, Ferguson R (2002) Intra-abdominal bacterial infection reactivates latent pulmonary cytomegalovirus in immunocompetent mice. J Infect Dis 185:1395–1400
Cook CH, Trgovcich J, Zimmerman PD, Zhang Y, Sedmak DD (2006) Lipopolysaccharide, tumor necrosis factor alpha, or interleukin-1{beta} triggers reactivation of latent cytomegalovirus in immunocompetent mice. J Virol 80(18):9151–9158
Hummel M, Zhang Z, Yan S, DePlaen I, Golia P, Varghese T, Thomas G, Abecassis MI (2001) Allogeneic transplantation induces expression of cytomegalovirus immediate-early genes in vivo: a model for reactivation from latency. J Virol 75(10):4814–4822
Simon CO, Seckert CK, Dreis D, Reddehase MJ, Grzimek NKA (2005) Role for tumor necrosis factor alpha in murine cytomegalovirus transcriptional reactivation in latently infected lungs. J Virol 79(1):326–340
Simon CO, Kuhnapfel B, Reddehase MJ, Grzimek NKA (2007) Murine cytomegalovirus major immediate-early enhancer region operating as a genetic switch in bidirectional gene pair transcription. J Virol 81(14):7805–7810. https://doi.org/10.1128/jvi.02388-06
Loser P, Jennings GS, Strauss M, Sandig V (1998) Reactivation of the previously silenced cytomegalovirus major immediate-early promoter in the mouse liver: involvement of NFkappaB. J Virol 72(1):180–190
Murphy JC, Fischle W, Verdin E, Sinclair JH (2002) Control of cytomegalovirus lytic gene expression by histone acetylation. EMBO J 21(5):1112–1120. https://doi.org/10.1093/emboj/21.5.1112
Reeves MB, MacAry PA, Lehner PJ, Sissons JGP, Sinclair JH (2005) Latency, chromatin remodeling, and reactivation of human cytomegalovirus in the dendritic cells of healthy carriers. Proc Natl Acad Sci 102(11):4140–4145. https://doi.org/10.1073/pnas.0408994102
Liu XF, Yan S, Abecassis M, Hummel M (2008) Establishment of murine cytomegalovirus latency in vivo is associated with changes in histone modifications and recruitment of transcriptional repressors to the major immediate-early promoter. J Virol 82(21):10922–10931. https://doi.org/10.1128/jvi.00865-08
Hummel M, Yan S, Li Z, Varghese TK, Abecassis M (2007) Transcriptional reactivation of murine cytomegalovirus ie gene expression by 5-aza-2'-deoxycytidine and trichostatin A in latently infected cells despite lack of methylation of the major immediate-early promoter. J Gen Virol 88(4):1097–1102. https://doi.org/10.1099/vir.0.82696-0
Dağ F, Dölken L, Holzki J, Drabig A, Weingärtner A, Schwerk J, Lienenklaus S, Conte I, Geffers R, Davenport C, Rand U, Köster M, Weiß S, Adler B, Wirth D, Messerle M, Hauser H, Čičin-Šain L (2014) Reversible silencing of cytomegalovirus genomes by type I interferon governs virus latency. PLoS Pathog 10(2):e1003962. https://doi.org/10.1371/journal.ppat.1003962
Simon CO, Holtappels R, Tervo H-M, Bohm V, Daubner T, Oehrlein-Karpi SA, Kuhnapfel B, Renzaho A, Strand D, Podlech J, Reddehase MJ, Grzimek NKA (2006) CD8 T cells control cytomegalovirus latency by epitope-specific sensing of transcriptional reactivation. J Virol 80(21):10436–10456
Kurz SK, Reddehase MJ (1999) Patchwork pattern of transcriptional reactivation in the lungs indicates sequential checkpoints in the transition from murine cytomegalovirus latency to recurrence. J Virol 73(10):8612–8622
Boomer JS, To K, Chang KC, Takasu O, Osborne DF, Walton AH, Bricker TL, Jarman SD, Kreisel D, Krupnick AS, Srivastava A, Swanson PE, Green JM, Hotchkiss RS (2011) Immunosuppression in patients who die of sepsis and multiple organ failure. JAMA J Am Med Assoc 306(23):2594–2605. https://doi.org/10.1001/jama.2011.1829
Hotchkiss RS, Nicholson DW (2006) Apoptosis and caspases regulate death and inflammation in sepsis. Nat Rev Immunol 6(11):813–822
Campbell J, Trgovcich J, Kincaid M, Zimmerman PD, Klenerman P, Sims S, Cook CH (2012) Transient CD8-memory contraction: a potential contributor to latent cytomegalovirus reactivation. J Leukoc Biol 92(5):933–937. https://doi.org/10.1189/jlb.1211635
Hotchkiss RS, Tinsley KW, Swanson PE, Schmieg RE, Hui JJ, Chang KC, Osborne DF, Freeman BD, Cobb JP, Buchman TG, Karl IE (2001) Sepsis-induced apoptosis causes progressive profound depletion of B and CD4+ T lymphocytes in humans. J Immunol 166(11):6952–6963
Jonjic S, Pavic I, Polic B, Crnkovic I, Lucin P, Koszinowski UH (1994) Antibodies are not essential for the resolution of primary cytomegalovirus infection but limit dissemination of recurrent virus. J Exp Med 179(5):1713–1717. https://doi.org/10.1084/jem.179.5.1713
Krmpotić A, Podlech J, Reddehase MJ, Britt WJ, Jonjić S (2019) Role of antibodies in confining cytomegalovirus after reactivation from latency: three decades’ résumé. Med Microbiol Immunol. https://doi.org/10.1007/s00430-019-00600-1
Holtappels A, Pahl-Seibert MF, Thomas D, Reddehase MJ (2000) Enrichment of immediate-early 1 (m123/pp89) peptide-specific CD8 T cells in a pulmonary CD62L10 memory-effector cell pool during latent murine cytomegalovirus infection of the lungs. J Virol 74(24):11495–11503
Sylwester AW, Mitchell BL, Edgar JB, Taormina C, Pelte C, Ruchti F, Sleath PR, Grabstein KH, Hosken NA, Kern F, Nelson JA, Picker LJ (2005) Broadly targeted human cytomegalovirus-specific CD4+ and CD8+ T cells dominate the memory compartments of exposed subjects. J Exp Med 202(5):673–685. https://doi.org/10.1084/jem.20050882
Vescovini R, Biasini C, Fagnoni FF, Telera AR, Zanlari L, Pedrazzoni M, Bucci L, Monti D, Medici MC, Chezzi C, Franceschi C, Sansoni P (2007) Massive load of functional effector CD4+ and CD8+ T cells against cytomegalovirus in very old subjects. J Immunol 179(6):4283–4291
Seckert CK, Griessl M, Buttner JK, Scheller S, Simon CO, Kropp KA, Renzaho A, Kuhnapfel B, Grzimek NK, Reddehase MJ (2012) Viral latency drives 'memory inflation': a unifying hypothesis linking two hallmarks of cytomegalovirus infection. Med Microbiol Immunol. https://doi.org/10.1007/s00430-012-0273-y
Snyder CM, Cho KS, Bonnett EL, van Dommelen S, Shellam GR, Hill AB (2008) Memory inflation during chronic viral infection is maintained by continuous production of short-lived, functional T cells. Immunity 29(4):650–659
Baars PA, Sierro S, Arens R, Tesselaar K, Hooibrink B, Klenerman P, van Lier RAW (2005) Properties of murine CD8+CD27− T cells. Eur J Immunol 35(11):3131–3141
Deutschman CS, Konstantinides FN, Tsai M, Simmons RL, Cerra FB (1987) Physiology and metabolism in isolated viral septicemia: further evidence of an organism-independent, Host-Dependent Response. Arch Surg 122(1):21–25. https://doi.org/10.1001/archsurg.1987.01400130027003
Cook CH, Zhang Y, Sedmak DD, Martin LC, Jewell S, Ferguson RM (2006) Pulmonary cytomegalovirus reactivation causes pathology in immunocompetent mice. Crit Care Med 34(3):842–849
Barton ES, White DW, Cathelyn JS, Brett-McClellan KA, Engle M, Diamond MS, Miller VL, Virgin HW (2007) Herpesvirus latency confers symbiotic protection from bacterial infection. Nature 447(7142):326–329
Cook CH, Trgovcich J (2011) Cytomegalovirus reactivation in critically ill immunocompetent hosts: a decade of progress and remaining challenges. Antiviral Res 90(3):151–159. https://doi.org/10.1016/j.antiviral.2011.03.179
Welsh RM, Selin LK (2002) No one is naive: the significance of heterologous T-cell immunity. Nat Rev Immunol 2(6):417–426
Seok J, Warren HS, Cuenca AG, Mindrinos MN, Baker HV, Xu W, Richards DR, McDonald-Smith GP, Gao H, Hennessy L, Finnerty CC, Lopez CM, Honari S, Moore EE, Minei JP, Cuschieri J, Bankey PE, Johnson JL, Sperry J, Nathens AB, Billiar TR, West MA, Jeschke MG, Klein MB, Gamelli RL, Gibran NS, Brownstein BH, Miller-Graziano C, Calvano SE, Mason PH, Cobb JP, Rahme LG, Lowry SF, Maier RV, Moldawer LL, Herndon DN, Davis RW, Xiao W, Tompkins RG (2013) Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.1222878110
Mansfield S, Grießl M, Gutknecht M, Cook C (2015) Sepsis and cytomegalovirus: foes or conspirators? Med Microbiol Immunol. https://doi.org/10.1007/s00430-015-0407-0
Beura LK, Hamilton SE, Bi K, Schenkel JM, Odumade OA, Casey KA, Thompson EA, Fraser KA, Rosato PC, Filali-Mouhim A, Sekaly RP, Jenkins MK, Vezys V, Haining WN, Jameson SC, Masopust D (2016) Normalizing the environment recapitulates adult human immune traits in laboratory mice. Nature 532(7600):512–516. https://doi.org/10.1038/nature17655
Jergovic M, Contreras NA, Nikolich-Zugich J (2019) Impact of CMV upon immune aging: facts and fiction. Med Microbiol Immunol. https://doi.org/10.1007/s00430-019-00605-w
Soderberg-Naucler C, Fish KN, Nelson JA (1997) Reactivation of latent human cytomegalovirus by allogeneic stimulation of blood cells from healthy donors. Cell 91(1):119–126
Larsson S, Soderberg-Naucler C, Wang FZ, Moller E (1998) Cytomegalovirus DNA can be detected in peripheral blood mononuclear cells from all seropositive and most seronegative healthy blood donors over time. Transfusion 38(3):271–278
Slobedman B, Mocarski ES (1999) Quantitative analysis of latent human cytomegalovirus. J Virol 73(6):4806–4812
Dioverti MV, Razonable RR (2015) Clinical utility of cytomegalovirus viral load in solid organ transplant recipients. Curr Opin Infect Dis 28(4):317–322. https://doi.org/10.1097/QCO.0000000000000173
Reddehase M, Lemmermann N (2019) Cellular reservoirs of latent cytomegaloviruses. Med Microbiol Immunol. https://doi.org/10.1007/s00430-019-00592-y
Reddehase MJ, Balthesen M, Rapp M, Jonjic S, Pavic I, Koszinowski UH (1994) The conditions of primary infection define the load of latent viral genome in organs and the risk of recurrent cytomegalovirus disease. J Exp Med 179(1):185–193
Adler SP, Reddehase M (2019) Pediatric roots of cytomegalovirus recurrence and memory inflation in the elderly. Med Microbiol Immunol. https://doi.org/10.1007/s00430-019-00609-6
Redeker A, Welten SPM, Arens R (2014) Viral inoculum dose impacts memory T-cell inflation. Eur J Immunol 44(4):1046–1057. https://doi.org/10.1002/eji.201343946
Trgovcich J, Kincaid M, Thomas A, Griessl M, Zimmerman P, Dwivedi V, Bergdall V, Klenerman P, Cook CH (2016) Cytomegalovirus reinfections stimulate CD8 T-memory inflation. PLoS ONE 11(11):e0167097. https://doi.org/10.1371/journal.pone.0167097
Redeker A, Remmerswaal EBM, van der Gracht ETI, Welten SPM, Höllt T, Koning F, Cicin-Sain L, Nikolich-Žugich J, ten Berge IJM, van Lier RAW, van Unen V, Arens R (2018) The contribution of cytomegalovirus infection to immune senescence is set by the infectious dose. Front Immunol 8(1953):1–15. https://doi.org/10.3389/fimmu.2017.01953
Mansfield SA, Dwivedi V, Elgharably H, Griessl M, Zimmerman PD, Limaye AP, Cook CH (2019) Cytomegalovirus immunoglobulin-G titers do not predict reactivation risk in immunocompetent hosts. J Med Virol. https://doi.org/10.1002/jmv.25389
Booth TW, Scalzo AA, Carrello C, Lyons PA, Farrell HE, Singleton GR, Shellam GR (1993) Molecular and biological characterization of new strains of murine cytomegalovirus isolated from wild mice. Arch Virol 132(1–2):209–220
Kotsimbos ATC, Sinickas V, Glare EM, Esmore DS, Snell GI, Walters EH, Williams TJ (1997) Quantitative detection of human cytomegalovirus DNA in lung transplant recipients. Am J Respir Crit Care Med 156(4):1241–1246
Thomas AC, Forster MR, Bickerstaff AA, Zimmerman PD, Wing BA, Trgovcich J, Bergdall VK, Klenerman P, Cook CH (2010) Occult cytomegalovirus in vivarium-housed mice may influence transplant allograft acceptance. Transpl Immunol 23(1–2):86–91
Stowe RP, Kozlova EV, Yetman DL, Walling DM, Goodwin JS, Glaser R (2007) Chronic herpesvirus reactivation occurs in aging. Exp Gerontol 42(6):563–570. https://doi.org/10.1016/j.exger.2007.01.005
Parry HM, Zuo J, Frumento G, Mirajkar N, Inman C, Edwards E, Griffiths M, Pratt G, Moss P (2016) Cytomegalovirus viral load within blood increases markedly in healthy people over the age of 70 years. Immun Ageing 13(1):1. https://doi.org/10.1186/s12979-015-0056-6
Leng S, Qu T, Semba R, Li H, Yao X, Nilles T, Yang X, Manwani B, Walston J, Ferrucci L, Fried L, Margolick J, Bream J (2011) Relationship between cytomegalovirus (CMV) IgG serology, detectable CMV DNA in peripheral monocytes, and CMV pp65&495–503-specific CD8+ T cells in older adults. Age 33(4):607–614. https://doi.org/10.1007/s11357-011-9205-9
Leng SX, Li H, Xue QL, Tian J, Yang X, Ferrucci L, Fedarko N, Fried LP, Semba RD (2011) Association of detectable cytomegalovirus (CMV) DNA in monocytes rather than positive CMV IgG serology with elevated neopterin levels in community-dwelling older adults. Exp Gerontol 46(8):679–684. https://doi.org/10.1016/j.exger.2011.04.002
Toro AI, Ossa J (1996) PCR activity of CMV in healthy CMV-seropositive individuals: does latency need redefinition? Res Virol 147(4):233–238
Robert FP, Hutto C (1986) Group day care and cytomegaloviral infections of mothers and children. Rev Infect Dis 8(4):599–605
Zanghellini F, Boppana SB, Pass RF, Griffiths PD, Emery VC (1999) Asymptomatic primary cytomegalovirus infection: virologic and immunologic features. J Infect Dis 180(3):702–707. https://doi.org/10.1086/314939
Arora N, Novak Z, Fowler KB, Boppana SB, Ross SA (2010) Cytomegalovirus viruria and DNAemia in healthy seropositive women. J Infect Dis 202(12):1800–1803. https://doi.org/10.1086/657412
Mehta SK, Stowe RP, Feiveson AH, Tyring SK, Pierson DL (2000) Reactivation and shedding of cytomegalovirus in astronauts during spaceflight. J Infect Dis 182(6):1761–1764. https://doi.org/10.1086/317624
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Additional information
Edited by: Matthias J. Reddehase.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Special Issue on Immunological Imprinting during Chronic Viral Infection.
Rights and permissions
About this article
Cite this article
Marandu, T., Dombek, M. & Cook, C.H. Impact of cytomegalovirus load on host response to sepsis. Med Microbiol Immunol 208, 295–303 (2019). https://doi.org/10.1007/s00430-019-00603-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00430-019-00603-y