Abstract
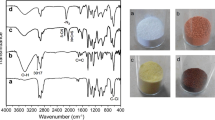
N-phenylmaleimide (NPMI) has a specially five-membered ring and N-aryl substituted structure, which allows its homopolymers and copolymers to exhibit remarkable thermal resistance properties. By introducing a desired amount of NPMI into polyvinyl acetate (PVAc) and polyvinyl alcohol (PVA) main chain, their thermal resistance properties will hopefully be significantly improved. There is a very large gap in the reactivity ratios between NPMI (rNPMI = 86.12) and vinyl acetate (VAc, rVAc = 0.001) in methanol by counting experimental results. As a result, the co-polymerization of NPMI (a relatively high reactive monomer) with low-reactive VAc remains a great challenge in industrial synthesis. In this paper, VAc was copolymerized with different mole ratios NPMI by starvation feeding method. The DSC curves indicated that the introduction of 4% NPMI improved Tg of PVAc/NPMI by about 10 °C. After hydrolysis, the corresponding PVA/NPMI also exhibits higher Tg compared to that of pure PVA under identical polymerization conditions. Interestingly, a visible and rapid discoloration can be observed after the incorporation of a base catalyst during the alcoholysis of PVAc/NPMI, and such color change is reversible under acid or base conditions. The mechanism of color change has been ascribed to interconversion between keto and enol under the interplay of acid or base conditions.









Similar content being viewed by others
References
Zhao C, Dong J, Li S, Fan Z (2012) Synthesis and characterization of heat-resistant N-phenylmaleimide-styrene-maleic anhydride copolymers and application in acrylonitrile-butadiene-styrene resin. J Appl Polym Sci 126:169–178. https://doi.org/10.1002/app.36544
Yuan Y, Siegmann A, Narkis M, Bell JP (1996) Emulsion copolymerization of N-phenylmaleimide with styrene. J Appl Polym Sci 61:1049–1054. https://doi.org/10.1002/(sici)1097-4628(19960808)61:6%3c1049::Aid-app19%3e3.0.Co;2-r
Krishnamoorthy S, Haria M, Fortier-McGill BE, Mazumder MAJ, Robinson EI, Xia Y, Burke NAD, Stover HDH (2011) High Tg microspheres by dispersion copolymerization of N-phenylmaleimide with styrenic or alkyl vinyl ether monomers. J Polym Sci Pol Chem 49:192–202. https://doi.org/10.1002/pola.24440
Lu Y, Sun W, Shen Z (2002) Copolymerization of N-phenylmaleimide with styrene by rare earth coordination catalyst. Eur Polym J 38:1275–1279. https://doi.org/10.1016/s0014-3057(02)00013-7
Zhou JM, Zhang CX, Shen CL, Wang Y (2019) Synthesis of poly(2-dimethylaminoethyl methacrylate)-block-poly(styrene-alt-N-phenylmaleimide) and its thermo-tolerant nanoporous films prepared by selective swelling. Polymer 164:126–133. https://doi.org/10.1016/j.polymer.2019.01.013
Dean BD (1987) Styrene/N-phenylmaleimide copolymer miscible blends. J Appl Polym Sci 33:2259–2261. https://doi.org/10.1002/app.1987.070330634
Shan GR, Weng ZX, Huang ZM, Pan ZR (1997) Free radical copolymerization and kinetic treatment of styrene with N-phenylmaleimide. J Appl Polym Sci 63:1535–1542. https://doi.org/10.1002/(SICI)1097-4628(19970321)63:123.0.CO;2-P
Mohamed AA, Jebrael FH, Elsabee MZ (2002) Copolymerization of styrene with N-arylmaleimides. Macromolecules 19:32–37. https://doi.org/10.1021/ma00155a006
Doi T, Akimoto A, Matsumoto A, Oki Y, Otsu T (1996) Alternating copolymerization of N-(alkyl-substituted phenyl)maleimides with isobutene and thermal properties of the resulting copolymers. J Polym Sci A Polym Chem 34:2499–2505. https://doi.org/10.1002/(sici)1099-0518(19960915)34:122499::Aid-pola233.0.Co;2-2
Doi T, Yukioka S, Inoue H, Akimoto A (1997) A new class of transparent polymeric materials. III. Miscible blends of poly(N-methylmaleimide-alt-isobutene) with poly(acrylonitrile-co-styrene) and the properties of the blends. J Appl Polym Sci 63:925–929. https://doi.org/10.1002/(sici)1097-4628(19970214)63:7925::Aid-app133.0.Co;2-q
Patel JD, Patel MR (2006) Free radical copolymerizations of N-arylmaleimides. J Macromol Sci A 19:801–812. https://doi.org/10.1080/00222338308060777
Senel S, Rzaev ZMO, Piskin E (2003) Copolymerization of N-phenylmaleimide with 2-hydroxyethyl and ethyl methacrylates. Polym Int 52:713–721. https://doi.org/10.1002/pi.1117
Mohamed AA, Hasan MEH (1991) Copolymerization of N-phenyl maleimide with methyl acrylate, ethyl acrylate, methyl methacrylate, and butyl methacrylate. Acta Polym 42:544–547. https://doi.org/10.1002/actp.1991.010421102
Janović Z, Matusinović TT, Ranogajec F (1993) Copolymerization and copolymers of N-(2,4,6-tribromophenyl)maleimide with methyl acrylate and methyl methacrylate. Die Makromolekulare Chemie 194:1915–1923. https://doi.org/10.1002/macp.1993.021940706
Du M, Weng Z-X, Shan G-R, Huang Z-M, Pan Z-R (2000) Control and optimization for vinyl chloride/N-phenylmaleimide copolymer composition. Eur Polym J 36:1455–1462. https://doi.org/10.1016/s0014-3057(99)00202-5
Barrales-rienda JM, De La Campa JIG, Ramos JG (1977) Free-radical copolymerizations of N-phenyl Maleimide. J Macromol Sci A 11:267–286. https://doi.org/10.1080/00222337708061267
Elsabee MZ, Sabaa MW, Mokhtar S (1983) Copolymerization of N-arylmaleimides with vinyl acetate. Polym J 15:429–434. https://doi.org/10.1295/polymj.15.429
Cui S, Li L, Wang Q (2016) Enhancing glass transition temperature and mechanical properties of poly (propylene carbonate) by intermacromolecular complexation with poly (vinyl alcohol). Compos Sci Technol 127:177–184. https://doi.org/10.1016/j.compscitech.2016.03.007
Omayu A, Matsumoto A (2008) Thermal properties of N-phenylmaleimide-isobutene alternating copolymers containing polar groups to form intermolecular and intramolecular hydrogen bonding. Polym J 40:736–742. https://doi.org/10.1295/polymj.PJ2008073
Liu G, Li X, Zhang L, Qu X, Liu P, Yang L, Gao J (2002) Thermal analysis of solution copolymers of styrene with N-phenylmaleimide. J Appl Polym Sci 83:417–422. https://doi.org/10.1002/app.10065
Fleš DD, Vuković R, Ranogajec F (1989) Copolymerization of α-methylstyrene and N-phenylmaleimide. J Polym Sci, Part A: Polym Chem 27:3227–3236. https://doi.org/10.1002/pola.1989.080271004
Semsarzadeh MA, Karimi A, Eshtad M (1997) Polymerizations of vinyl acetate in solution. Iranian Poylmer Journal 6:261–268
Han H, Zhang J (2013) Lightly branched poly(vinyl alcohol) for fluid loss additive. J Appl Polym Sci:n/a-n/a. https://doi.org/10.1002/app.39741
Perea LFT, Okorafo OE, Hutchinson RA, Guay M (2010) Development of on-line optimization-based control strategies for a starved-feed semibatch copolymerization reactor. Control Eng Pract 18:131–139. https://doi.org/10.1016/j.conengprac.2009.10.003
Arzamendi G, Asua JM (1989) Monomer addition policies for copolymer composition control in semicontinuous emulsion copolymerization. J Appl Polym Sci 38:2019–2036. https://doi.org/10.1002/app.1989.070381106
Nasresfahani A, Idowu LA, Hutchinson RA (2019) Extractable content of functional acrylic resins produced by radical copolymerization: a comparison of experiment and stochastic simulation. Chem Eng Sci 378. https://doi.org/10.1016/j.cej.2019.122087
Fineman M, Ross SD (1950) Linear method for determining monomer reactivity ratios in copolymerization. J Polym Sci 5:259–262. https://doi.org/10.1002/pol.1950.120050210
Otsu T, Matsumoto A, Kubota T, Mori S (1990) Reactivity in radical polymerization of N-substituted maleimides and thermal stability of the resulting polymers. Polym Bull 23:43–50. https://doi.org/10.1007/bf00983962
Yoshida Y, Endo T (2017) Dependence of color change of vinylethylene carbonate copolymers having N-substituted maleimides on chemical structure by acid-base switching in solution and solid state. React Funct Polym 120:139–146. https://doi.org/10.1016/j.reactfunctpolym.2017.10.001
Cao TT, Male U, Huh DS (2018) Fabrication of pore-selective carboxyl group functionalized polyimide honeycomb-patterned porous films using KOH humidity. Polymer 153:86–94. https://doi.org/10.1016/j.polymer.2018.08.006
Pistorius AM, Groenen PJ, De Grip WJ (1993) Infrared analysis of peptide succinimide derivatives. Int J Pept Protein Res 42:570–577. https://doi.org/10.1111/j.1399-3011.1993.tb00366.x
Yoshida Y, Endo T (2017) Color change of alternating copolymers with phenyl vinylethylene carbonate and N-phenylmaleimide in a solution and in the solid-state, depending on their structure. RSC Adv 7:9373–9380. https://doi.org/10.1039/c6ra28446a
Khan MN (1987) The kinetics and mechanism of alkaline hydrolysis of N-substituted phthalimides. Int J Chem Kinet 19:143–153. https://doi.org/10.1002/kin.550190206
Chen Y, Tong L, Zhang D, Yang W, Deng J (2012) Thermostable microspheres consisting of poly(N-phenylmaleimide-co-α-methyl styrene) prepared by precipitation polymerization. Ind Eng Chem Res 51:15610–15617. https://doi.org/10.1021/ie301941r
Yang LT, Zhang LC, Liu GD, Gao JG, Yang MS, Jin RG (2001) Synthesis and thermal analysis of emulsion terpolymers of N-phenylmaleimide, methyl methacrylate, and acrylonitrile. J Appl Polym Sci 81:2455–2462. https://doi.org/10.1002/app.1687
Matsumoto M, Maeda M (1955) Kinetics of the polymerization of vinyl acetate in benzene solution. J Polym Sci 17:435–438. https://doi.org/10.1002/pol.1955.120178516
Troitskii BB, Razuvaev GA, Khokhlova LV, Bortnikov GN (1973) On the mechanism of the thermal degradation of polyvinyl acetate. Journal of Polymer Science: Polymer Symposia 42:1363–1375. https://doi.org/10.1002/polc.5070420336
Ballistreri A, Foti S, Montaudo G, Scamporrino E (1980) Evolution of aromatic compounds in the thermal decomposition of vinyl polymers. J Polym Sci A Polym Chem 18:1147–1153. https://doi.org/10.1002/pol.1980.170180401
Acknowledgements
The authors would like to appreciate The State Key Laboratory of Polymer Materials Engineering, New Materials R & D Center, Institute of Chemical Material, China Academy of Engineering Physics.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhao, X., Tian, P., Deng, J. et al. Synthesis and characterization of vinyl acetate/N-phenylmaleimide copolymers and the corresponding polyvinyl alcohol/N-phenylmaleimide with color cycle change property. Colloid Polym Sci 300, 11–20 (2022). https://doi.org/10.1007/s00396-021-04920-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-021-04920-8