Abstract
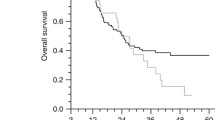
Myelodysplastic syndrome (MDS) is a rare clonal hematopoietic disorder in children. The risk stratification system and treatment strategy for adults are unfit for children. The role of hypomethylating agents (HMAs) in higher-risk childhood MDS has not been identified. This study aimed to investigate the outcomes of hematopoietic stem cell transplantation (HSCT) in children with higher-risk MDS at one single center. A retrospective study was conducted in children with higher-risk MDS undergoing HSCT between September 2019 and March 2023 at Blood Diseases Hospital CAMS. The clinical characteristics and transplantation information were reviewed and analyzed. A total of 27 patients were analyzed, including 11 with MDS with excess blasts (MDS-EB), 14 with MDS-EB in transformation (MDS-EBt) or acute myeloid leukemia with myelodysplasia-related changes (AML-MRC), and 2 with therapy-related MDS/AML (t-MDS/AML). Eight patients harbored monosomy 7. Before transplantation, induction therapy was administered to 25 patients, and 19 of them achieved bone marrow blasts <5% before HSCT. The stem cell source was unmanipulated-related bone marrow or peripheral blood stem cells for nineteen patients and unrelated cord blood for eight. All patients received decitabine-containing and Bu/Cy-based myeloablative conditioning; 26 patients achieved initial engraftment. The cumulative incidences of grade II–IV and grade III–IV acute graft-versus-host disease (GvHD) at 100 days were 65.4% and 42.3%, respectively. The incidence of cGvHD was 38.5%. The median follow-up was 26 (range 4–49) months after transplantation. By the end of follow-up, two patients died of complications and two died of disease progression. The probability of 3-year overall survival (OS) was 84.8% (95%CI, 71.1 to 98.5%). In summary, decitabine-containing myeloablative conditioning resulted in excellent outcomes for children with higher-risk MDS undergoing allogeneic HSCT.

Similar content being viewed by others
References
WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. Revised 4th edition. 2017
Wlodarski MW, Sahoo SS, Niemeyer CM (2018) Monosomy 7 in pediatric myelodysplastic syndromes. Hematol Oncol Clin North Am 32(4):729–743. https://doi.org/10.1016/j.hoc.2018.04.007
Locatelli F, Strahm B (2018) How I treat myelodysplastic syndromes of childhood. Blood. 131(13):1406–1414. https://doi.org/10.1182/blood-2017-09-765214
Strahm B, Nöllke P, Zecca M et al (2011) Hematopoietic stem cell transplantation for advanced myelodysplastic syndrome in children: results of the EWOG-MDS 98 study. Leukemia 25(3):455–462. https://doi.org/10.1038/leu.2010.297
Suo P, Wang S, Xue Y et al (2020) Unmanipulated haploidentical hematopoietic stem cell transplantation for children with myelodysplastic syndrome. Pediatr Transplant 24(7):e13864. https://doi.org/10.1111/petr.13864
Strahm B, Locatelli F, Bader P et al (2007) Reduced intensity conditioning in unrelated donor transplantation for refractory cytopenia in childhood. Bone Marrow Transplant 40(4):329–333. https://doi.org/10.1038/sj.bmt.1705730
Inagaki J, Fukano R, Kurauchi K et al (2015) Hematopoietic stem cell transplantation in children with refractory cytopenia of childhood: single-center experience using high-dose cytarabine containing myeloablative and aplastic anemia oriented reduced-intensity conditioning regimens. Biol Blood Marrow Transplant 21(3):565–569. https://doi.org/10.1016/j.bbmt.2014.12.003
Gao J, Hu Y, Gao L et al (2022) The effect of decitabine-combined minimally myelosuppressive regimen bridged allo-HSCT on the outcomes of pediatric MDS from 10 years’ experience of a single center. BMC Pediatr. 22(1):312. https://doi.org/10.1186/s12887-022-03376-1
Kantarjian H, Issa JP, Rosenfeld CS et al (2006) Decitabine improves patient outcomes in myelodysplastic syndromes: results of a phase III randomized study. Cancer. 106(8):1794–1803. https://doi.org/10.1002/cncr.21792
Qin Y, Kuang P, Zeng Q et al (2019) Hypomethylating agents for patients with myelodysplastic syndromes prior to hematopoietic stem cell transplantation: a systematic review and meta-analysis. Ann Hematol. 98(11):2523–2531. https://doi.org/10.1007/s00277-019-03811-x
Waespe N, Van Den Akker M, Klaassen RJ et al (2016) Response to treatment with azacitidine in children with advanced myelodysplastic syndrome prior to hematopoietic stem cell transplantation. Haematologica 101(12):1508–1515. https://doi.org/10.3324/haematol.2016.145821
Winters AC, Maloney KW, Treece AL et al (2020) Single-center pediatric experience with venetoclax and azacitidine as treatment for myelodysplastic syndrome and acute myeloid leukemia. Pediatr Blood Cancer 67(10):e28398. https://doi.org/10.1002/pbc.28398
Almstedt M, Blagitko-Dorfs N, Duque-Afonso J et al (2010) The DNA demethylating agent 5-aza-2'-deoxycytidine induces expression of NY-ESO-1 and other cancer/testis antigens in myeloid leukemia cells. Leuk Res 34(7):899–905. https://doi.org/10.1016/j.leukres.2010.02.004
Tang X, Valdez BC, Ma Y et al (2021) Low-dose decitabine as part of a modified Bu-Cy conditioning regimen improves survival in AML patients with active disease undergoing allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 56(7):1674–1682. https://doi.org/10.1038/s41409-021-01238-5
Cao YG, He Y, Zhang SD et al (2020) Conditioning regimen of 5-day decitabine administration for allogeneic stem cell transplantation in patients with myelodysplastic syndrome and myeloproliferative neoplasms. Biol Blood Marrow Transplant 26(2):285–291. https://doi.org/10.1016/j.bbmt.2019.09.001
Valdez BC, Li Y, Murray D et al (2010) 5-Aza-2'-deoxycytidine sensitizes busulfan-resistant myeloid leukemia cells by regulating expression of genes involved in cell cycle checkpoint and apoptosis. Leuk Res 34(3):364–372. https://doi.org/10.1016/j.leukres.2009.08.014
Cany J, Roeven MWH, Hoogstad-van Evert JS et al (2018) Decitabine enhances targeting of AML cells by CD34+ progenitor-derived NK cells in NOD/SCID/IL2Rgnull mice. Blood 131(2):202–214. https://doi.org/10.1182/blood-2017-06-790204
D'Angelo CR, Hall A, Woo KM et al (2020) Decitabine induction with myeloablative conditioning and allogeneic hematopoietic stem cell transplantation in high-risk patients with myeloid malignancies is associated with a high rate of infectious complications. Leuk Res 96:106419. https://doi.org/10.1016/j.leukres.2020.106419
Pastor V, Hirabayashi S, Karow A et al (2017) Mutational landscape in children with myelodysplastic syndromes is distinct from adults: specific somatic drivers and novel germline variants. Leukemia 31(3):759–762. https://doi.org/10.1038/leu.2016.342
Schwartz JR, Ma J, Lamprecht T et al (2017) The genomic landscape of pediatric myelodysplastic syndromes. Nat Commun 8(1):1557. https://doi.org/10.1038/s41467-017-01590-5
Brown AL, Hahn CN, Scott HS (2020) Secondary leukemia in patients with germline transcription factor mutations (RUNX1, GATA2, CEBPA). Blood 136(1):24–35. https://doi.org/10.1182/blood.2019000937
Yoshida M, Tanase-Nakao K, Shima H et al (2020) Prevalence of germline GATA2 and SAMD9/9L variants in paediatric haematological disorders with monosomy 7. Br J Haematol 191(5):835–843. https://doi.org/10.1111/bjh.17006
Wang Y, Wang HX, Lai YR et al (2016) Haploidentical transplant for myelodysplastic syndrome: registry-based comparison with identical sibling transplant. Leukemia 30(10):2055–2063. https://doi.org/10.1038/leu.2016.110
Yoo JW, Im HJ, Kim H et al (2020) Improved outcomes of allogeneic hematopoietic stem cell transplantation including haploidentical transplantation for childhood myelodysplastic syndrome. Bone Marrow Transplant 55(8):1595–1603. https://doi.org/10.1038/s41409-020-0814-8
Madureira AB, Eapen M, Locatelli F et al (2011) Analysis of risk factors influencing outcome in children with myelodysplastic syndrome after unrelated cord blood transplantation. Leukemia 25(3):449–454. https://doi.org/10.1038/leu.2010.285
Milano F, Gooley T, Wood B et al (2016) Cord-blood transplantation in patients with minimal residual disease. N Engl J Med 375(10):944–953. https://doi.org/10.1056/NEJMoa1602074
Mufti GJ, Figes A, Hamblin TJ et al (1986) Immunological abnormalities in myelodysplastic syndromes. I. Serum immunoglobulins and autoantibodies. Br J Haematol 63(1):143–147. https://doi.org/10.1111/j.1365-2141.1986.tb07504.x
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (82070201[Y.G.]) and the CAMS Innovation Fund for Medical Sciences (CIFMS) (2021-12M-1-041).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Exemption of informed consent approved by the Ethics Committee of Blood Diseases Hospital, Chinese Academy of Medical Sciences (IIT2021009-EC-1).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ren, Y., Liu, F., Chen, X. et al. Decitabine-containing conditioning improved outcomes for children with higher-risk myelodysplastic syndrome undergoing allogeneic hematopoietic stem cell transplantation. Ann Hematol 103, 1345–1351 (2024). https://doi.org/10.1007/s00277-024-05628-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-024-05628-9