Abstract
Background
This trial was designed to evaluate the safety and clinical responses to a combination of temozolomide (TMZ) chemotherapy and immunotherapy with fusions of DCs and glioma cells in patients with glioblastoma (GBM).
Method
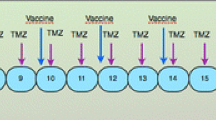
GBM patients were assigned to two groups: a group of recurrent GBMs after failing TMZ-chemotherapy against the initially diagnosed glioma (Group-R) or a group of newly diagnosed GBMs (Group-N). Autologous cultured glioma cells obtained from surgical specimens were fused with autologous DCs using polyethylene glycol. The fusion cells (FC) were inoculated intradermally in the cervical region. Toxicity, progression-free survival (PFS), and overall survival (OS) of this trial were evaluated. Expressions of WT-1, gp-100, and MAGE-A3, recognized as chemoresistance-associated peptides (CAP), were confirmed by immunohistochemistry of paraffin-embedded tumor samples. Patient’s PBMCs of pre- and post-vaccination were evaluated by tetramer and ELISPOT assays.
Results
FC-immunotherapy was well tolerated in all patients. Medians of PFS and OS of Group-R (n = 10) were 10.3 and 18.0 months, and those of Group-N (n = 22) were 18.3 and 30.5 months, respectively. Up-regulation and/or cytoplasmic accumulation of CAPs was observed in the recurrent tumors of Group-R patients compared with their initially excised tumors. Specific immune responses against CAPs were observed in the tetramer and ELISPOT assays.
Conclusions
The combination of TMZ-treatment leading to up-regulation and/or cytoplasmic accumulation of CAPs, with FC-immunotherapy as a means of producing specific immunity against CAPs, may safely induce anti-tumor effects in patients with GBM.





Similar content being viewed by others
Abbreviations
- AA:
-
Anaplastic astrocytoma
- AO:
-
Anaplastic oligodendroglioma
- CAP:
-
Chemoresistance-associated peptide
- CI:
-
Confidence intervals
- FC:
-
Fusion cell
- GBM:
-
Glioblastoma multiforme
- gp-100:
-
Glycoprotein-100
- IR:
-
Injection site reaction
- KPS:
-
Karnofsky performance status
- LL:
-
Leukopenia/lymphopenia
- MAGE:
-
Melanoma-associated antigen gene
- PFS:
-
Progression-free survival
- siRNA:
-
Small interference RNA
- TMZ:
-
Temozolomide
- TRP:
-
Tyrosinase-related protein
- WT-1:
-
Wilms’ Tumor 1
References
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996. doi:10.1056/NEJMoa043330
Chen G, Emens LA (2013) Chemoimmunotherapy: reengineering tumor immunity. Cancer Immunol Immunother 62:203–216. doi:10.1007/s00262-012-1388-0
Heimberger AB, Sun W, Hussain SF et al (2008) Immunological responses in a patient with glioblastoma multiforme treated with sequential courses of temozolomide and immunotherapy: case study. Neuro Oncol 10:98–103. doi:10.1215/15228517-2007-046
Gunzer M, Janich S, Varga G, Grabbe S (2001) Dendritic cells and tumor immunity. Semin Immunol 13:291–302. doi:10.1006/smim.2001.0325
Banchereau J, Steinman RM (1998) Dendritic cells and the control of immunity. Nature 392:245–252. doi:10.1038/32588
Guermonprez P, Valladeau J, Zitvogel L, Thery C, Amigorena S (2002) Antigen presentation and T cell stimulation by dendritic cells. Annu Rev Immunol 20:621–667. doi:10.1146/annurev.immunol.20.100301.064828
Kikuchi T, Akasaki Y, Irie M, Homma S, Abe T, Ohno T (2001) Results of a phase I clinical trial of vaccination of glioma patients with fusions of dendritic and glioma cells. Cancer Immunol Immunother 50:337–344
Akasaki Y, Kikuchi T, Homma S, Abe T, Kofe D, Ohno T (2001) Antitumor effect of immunizations with fusions of dendritic and glioma cells in a mouse brain tumor model. J Immunother 24:106–113
Kikuchi T, Akasaki Y, Abe T, Fukuda T, Saotome H, Ryan JL, Kufe DW, Ohno T (2004) Vaccination of glioma patients with fusions of dendritic and glioma cells and recombinant human interleukin 12. J Immunother 27:452–459
Akasaki Y, Kikuchi T, Irie M, Yamamoto Y, Arai T, Tanaka T, Joki T, Abe T (2011) Cotransfection of Poly(I: C) and siRNA of IL-10 into fusions of dendritic and glioma cells enhances antitumor T helper type 1 induction in patients with glioma. J Immunother 34:121–128. doi:10.1097/CJI.0b013e3181e5c278
Koido S, Homma S, Okamoto M et al (2014) Treatment with chemotherapy and dendritic cells pulsed with multiple Wilms’ tumor 1 (WT1)-specific MHC class I/II-restricted epitopes for pancreatic cancer. Clin Cancer Res 20:4228–4239. doi:10.1158/1078-0432.CCR-14-0314
Yu JS, Wheeler CJ, Zeltzer PM et al (2001) Vaccination of malignant glioma patients with peptide-pulsed dendritic cells elicits systemic cytotoxicity and intracranial T-cell infiltration. Cancer Res 61:842–847
Phuphanich S, Wheeler CJ, Rudnick JD et al (2013) Phase I trial of a multi-epitope-pulsed dendritic cell vaccine for patients with newly diagnosed glioblastoma. Cancer Immunol Immunother 62:125–135. doi:10.1007/s00262-012-1319-0
Butterfield LH, Gooding W, Whiteside TL (2008) Development of a potency assay for human dendritic cells: IL-12p70 production. J Immunother 31:89–100. doi:10.1097/CJI.0b013e318158fce0
Trinchieri G (2003) Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nat Rev Immunol 3:133–146. doi:10.1038/nri1001
Lasek W, Zagozdzon R, Jakobisiak M (2014) Interleukin 12: still a promising candidate for tumor immunotherapy? Cancer Immunol Immunother 63:419–435. doi:10.1007/s00262-014-1523-1
Okada H, Kalinski P, Ueda R et al (2011) Induction of CD8+ T-cell responses against novel glioma-associated antigen peptides and clinical activity by vaccinations with {alpha}-type 1 polarized dendritic cells and polyinosinic-polycytidylic acid stabilized by lysine and carboxymethylcellulose in patients with recurrent malignant glioma. J Clin Oncol 29:330–336. doi:10.1200/JCO.2010.30.7744
Liu G, Akasaki Y, Khong HT, Wheeler CJ, Das A, Black KL, Yu JS (2005) Cytotoxic T cell targeting of TRP-2 sensitizes human malignant glioma to chemotherapy. Oncogene 24:5226–5234. doi:10.1038/sj.onc.1208519
Chen MY, Clark AJ, Chan DC, Ware JL, Holt SE, Chidambaram A, Fillmore HL, Broaddus WC (2011) Wilms’ tumor 1 silencing decreases the viability and chemoresistance of glioblastoma cells in vitro: a potential role for IGF-1R de-repression. J Neurooncol 103:87–102. doi:10.1007/s11060-010-0374-7
Xie T, Nguyen T, Hupe M, Wei ML (2009) Multidrug resistance decreases with mutations of melanosomal regulatory genes. Cancer Res 69:992–999. doi:10.1158/0008-5472.CAN-08-0506
Monte M, Simonatto M, Peche LY, Bublik DR, Gobessi S, Pierotti MA, Rodolfo M, Schneider C (2006) MAGE-A tumor antigens target p53 transactivation function through histone deacetylase recruitment and confer resistance to chemotherapeutic agents. Proc Natl Acad Sci USA 103:11160–11165. doi:10.1073/pnas.0510834103
Blum JS, Wearsch PA, Cresswell P (2013) Pathways of antigen processing. Annu Rev Immunol 31:443–473. doi:10.1146/annurev-immunol-032712-095910
Benteyn D, Anguille S, Van Lint S et al (2013) Design of an Optimized Wilms’ Tumor 1 (WT1) mRNA Construct for Enhanced WT1 Expression and Improved Immunogenicity In Vitro and In Vivo. Mol Ther Nucleic Acids 2:e134. doi:10.1038/mtna.2013.54
Takahara A, Koido S, Ito M et al (2011) Gemcitabine enhances Wilms’ tumor gene WT1 expression and sensitizes human pancreatic cancer cells with WT1-specific T-cell-mediated antitumor immune response. Cancer Immunol Immunother 60:1289–1297. doi:10.1007/s00262-011-1033-3
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264. doi:10.1038/nrc3239
Acknowledgments
The authors thank Dr. Masako Nishikawa for her advice on statistical analyses, Dr. Kostadin L. Karagiozov for editing the manuscript, and Ms. Yukiko Kobayashi and Ms. Akiko Kuhara for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Akasaki, Y., Kikuchi, T., Homma, S. et al. Phase I/II trial of combination of temozolomide chemotherapy and immunotherapy with fusions of dendritic and glioma cells in patients with glioblastoma. Cancer Immunol Immunother 65, 1499–1509 (2016). https://doi.org/10.1007/s00262-016-1905-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-016-1905-7