Abstract
Annotation
Malignant gliomas are the most common primary brain tumor. Despite the variety of modern treatments, it is still a fatal disease with an extremely poor prognosis. The use of immunotherapy as a technique for the treatment of malignant tumors has great promise, retraining and exploiting the patient’s immune response against tumors.
Objective
Evaluation of the effectiveness of dendritic cell vaccine in patients with malignant brain gliomas in the structure of complex treatment in comparison with the control group of patients without immunotherapy in the structure of treatment.
Materials and methods
In a single-center, prospective, cohort study, taking place on the basis of the RNSI named after prof. A.L. Polenov, 91 patients with morphologically established malignant glial tumor (glioblastoma) took part. The main group of 41 patients who, in addition to standard treatment (surgical, radiation and chemotherapy), underwent specific antitumor immunotherapy. 50 patients received only standard treatment, without immunotherapy.
Results
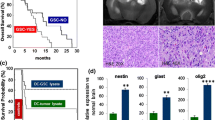
Median survival was 21.7 months in the immunotherapy group (95% CI 4–37 months) and 15.8 months (95% CI 3–22 months) in the non-immunotherapy group (p = 0.002). The median relapse-free period in the group with immunotherapy was 13.8 months (95% CI 1–20 months), and in the group without immunotherapy 7.9 months (95% CI 1–12 months) (p = 0.003).
Conclusion
In general, the use of immunotherapy in the structure of complex treatment of patients with malignant gliomas demonstrates a clear positive trend in terms of overall survival and median relapse-free period. But nevertheless, immunotherapy requires further development as a therapeutic tool, study and improvement, which will take into account immunosuppression in malignant gliomas and means of overcoming it, optimization in terms of target antigen selection, cell preparation and integration of dendritic vaccines into other treatment regimens.




Similar content being viewed by others
Data availability
The datasets generated during and analysed during the current study are available from the corresponding author on reasonable request.
References
Ostrom QT, Cioffi G, Waite K, Kruchko C, Barnholtz-Sloan JS (2021) CBTRUS Statistical Report: primary brain and other Central Nervous System tumors diagnosed in the United States in 2014–2018. Neuro Oncol 23:III1–III105. https://doi.org/10.1093/NEUONC/NOAB200
Poon MTC, Sudlow CLM, Figueroa JD, Brennan PM (2020) Longer-term (≥ 2 years) survival in patients with glioblastoma in population-based studies pre- and post-2005: a systematic review and meta-analysis. Sci Rep 10:11622. https://doi.org/10.1038/s41598-020-68011-4
Ostrom QT, Cote DJ, Ascha M, Kruchko C, Barnholtz-Sloan J (2018) Adult glioma incidence and survival by race or ethnicity in the United States from 2000 to 2014. JAMA Oncol 4:1254–1262. https://doi.org/10.1001/jamaoncol.2018.1789
Rynda AYu, Olyushin VE, Rostovtsev DM, Zabrodskaya YM, Tastanbekov MM, Papayan GV (2021) Intraoperative fluorescence control with chlorin E6 in resection of glial brain tumors. Zhurnal Voprosy Neirokhirurgii Imeni N N Burdenko 85(4):20–28 (In Russ.). https://doi.org/10.17116/neiro20218504120
Rynda AYu, Olyushin VE, Rostovtsev DM, Zabrodskaya YM, Ulitin AYu, Papayan GV (2023) Intraoperative photodynamic therapy in complex treatment of malignant gliomas. Burdenko’s Journal of Neurosurgery = Zhurnal Voprosy neirokhirurgii imeni N. N Burdenko 87(1):25–34 (In Russ.). https://doi.org/10.17116/neiro20238701125
Rynda AYu, Olyushin VE, Rostovtsev DM, Zabrodskaya YM, Papayan GV (2022) Comparative analysis of 5-ALA and chlorin E6 fluorescence-guided navigation in malignant glioma Surgery. Pirogov Russian Journal of Surgery = Khirurgiya Zurnal Im N I Pirogova 15–14 (In Russ.). https://doi.org/10.17116/hirurgia20220115
Chekhonin IV, Kobiakov GL, Gurina OI (2020) Dendritic cell vaccines in neurological oncology. Zhurnal Voprosy Neirokhirurgii Imeni N N Burdenko 84(1):76–85 (In Russ.). https://doi.org/10.17116/neiro20208401176
Medikonda R, Dunn G, Rahman M, Fecci P, Lim M (2020) A review of glioblastoma immunotherapy. J Neuro-Oncol 151:41–53. https://doi.org/10.1007/s11060-020-03448-1
Marin-Acevedo JA, Dholaria B, Soyano AE, Knutson KL, Chumsri S, Lou Y (2018) Next generation of immune checkpoint therapy in cancer: new developments and challenges. J Hematol Oncol 11:39. https://doi.org/10.1186/s13045-018-0582-8
Barrueto L, Caminero F, Cash L, Makris C, Lamichhane P, Deshmukh RR (2020) Resistance to checkpoint inhibition in cancer immunotherapy. Transl Oncol 13:100738. https://doi.org/10.1016/j.tranon.2019.12.010
Chauvin J-M, Zarour HM (2020) TIGIT in cancer immunotherapy. J Immunother Cancer 8:e000957. https://doi.org/10.1016/10.1136/jitc-2020-000957
Kim JE, Lim M (2015) The role of checkpoints in the treatment of GBM. J Neuro-Oncol 123:413–423. https://doi.org/10.1007/s11060-015-1747-8
Bao Z, Wang Y, Wang Q, Fang S, Shan X, Wang J et al (2021) Intratumor heterogeneity, microenvironment, and mechanisms of drug resistance in glioma recurrence and evolution. Front Med 15:551–561. https://doi.org/10.1007/s11684-020-0760-2
Andersen RS, Anand A, Harwood DSL, Kristensen BW (2021) Tumor-associated microglia and macrophages in the glioblastoma microenvironment and their implications for therapy. Cancers 13:4255. https://doi.org/10.3390/cancers13174255
Rynda A, Olyushin V, Rostovtsev D (2021) Immunotherapy in the complex treatment of HGG. Neurooncology 23(2):i47. https://doi.org/10.1093/neuonc/noab180.164
Datsi A, Sorg RV (2021) Dendritic cell vaccination of Glioblastoma: Road to Success or Dead End. Front Immunol 12:770390. https://doi.org/10.3389/fimmu.2021.770390
Liau LM, Black KL, Martin NA, Sykes SN, Bronstein JM, Jouben-Steele L et al (2000) Treatment of a patient by Vaccination with autologous dendritic cells pulsed with allogeneic major histocompatibility complex class I-Matched Tumor peptides. Case Rep Neurosurg Focus 9:e8. https://doi.org/10.3171/foc.2000.9.6.9
Burster T, Gärtner F, Bulach C, Zhanapiya A, Gihring A, Knippschild U (2021) Regulation of MHC I molecules in glioblastoma cells and the sensitizing of NK cells. Pharmaceuticals 14:236. https://doi.org/10.3390/ph14030236
Hung AL, Maxwell R, Theodros D, Belcaid Z, Mathios D, Luksik AS et al (2018) TIGIT and PD-1 dual checkpoint blockade enhances antitumor immunity and survival in GBM. Oncoimmunology 7(8):e1466769. https://doi.org/10.1080/2162402X.2018.1466769
Litak J, Mazurek M, Grochowski C, Kamieniak P, Roliński J (2019) PD-L1/PD-1 axis in Glioblastoma Multiforme. Int J Mol Sci 20:5347. https://doi.org/10.3390/ijms20215347
Wu A, Maxwell R, Xia Y, Cardarelli P, Oyasu M, Belcaid Z et al (2019) Combination anti-CXCR4 and anti-PD-1 immunotherapy provides survival benefit in glioblastoma through immune cell modulation of Tumor microenvironment. J Neuro Oncol 143:241–249. https://doi.org/10.1007/s11060-019-03172-5
Yu JS, Wheeler CJ, Zeltzer PM et al (2001) Vaccination of malignant glioma patients with peptide-pulsed dendritic cells elicits systemic cytotoxicity and intracranial T-cell infiltration. Cancer Res 61:842–847
Chan HY, Choi J, Jackson C, Lim M (2021) Combination immunotherapy strategies for glioblastoma. J Neuro-Oncol 151:375–391. https://doi.org/10.1007/s11060-020-03481-0
Lamano JB, Lamano JB, Li YD, DiDomenico JD, Choy W, Veliceasa D et al (2019) Glioblastoma-derived IL6 induces immunosuppressive peripheral myeloid cell PD-L1 and promotes Tumor growth. Clin Cancer Res 25:3643–3657. https://doi.org/10.1158/1078-0432.CCR-18-2402
Bausart M, Préat V, Malfanti A (2022) Immunotherapy for glioblastoma: the promise of combination strategies. J Exp Clin Cancer Res 41:35. https://doi.org/10.1186/s13046-022-02251-2
Jin K, Mao C, Chen L, Wang L, Liu Y, Yuan J (2021) Adenosinergic pathway: a hope in the immunotherapy of glioblastoma. Cancers 13:229. https://doi.org/10.3390/cancers13020229
Olyushin V, Rynda A, Rostovtsev D (2020) Immunotherapy with autologous dendritic cells as part of the complex treatment of patients with malignant gliomas. Ann Oncol 31(4):S405. https://doi.org/10.1016/j.annonc.2020.08.493
Yao Y, Luo F, Tang C, Chen D, Qin Z, Hua W et al (2018) Molecular subgroups and B7-H4 expression levels predict responses to dendritic cell vaccines in Glioblastoma: an exploratory Randomized Phase II Clinical Trial. Cancer Immunol Immunother 67:1777–1788. https://doi.org/10.1007/s00262-018-2232-y
Cho DY, Yang WK, Lee HC, Hsu DM, Lin HL, Lin SZ et al (2012) Adjuvant immunotherapy with whole-cell lysate dendritic cells vaccine for Glioblastoma Multiforme: a phase II clinical trial. World Neurosurg 77:736–744. https://doi.org/10.1016/j.wneu.2011.08.020
Wen PY, Reardon DA, Armstrong TS, Phuphanich S, Aiken RD, Landolfi JC et al (2019) A Randomized double-blind placebo-controlled phase II trial of dendritic cell vaccine Ict-107 in newly diagnosed patients with Glioblastoma. Clin Cancer Res 25:5799–5807. https://doi.org/10.1158/1078-0432.CCR-19-0261
Liau LM, Ashkan K, Tran DD, Campian JL, Trusheim JE, Cobbs CS et al (2018) First results on Survival from a large phase 3 clinical trial of an autologous dendritic cell vaccine in newly diagnosed Glioblastoma. J Transl Med 16:142. https://doi.org/10.1186/s12967-018-1507-6
McGranahan T, Therkelsen KE, Ahmad S, Nagpal S (2019) Current state of immunotherapy for treatment of glioblastoma. Curr Treat Options in Oncol 20:24. https://doi.org/10.1007/s11864-019-0619-4
Jie X, Hua L, Jiang W, Feng F, Feng G, Hua Z (2012) Clinical application of a dendritic cell vaccine raised against Heat-Shocked Glioblastoma. Cell Biochem Biophys 62:91–99. https://doi.org/10.1007/s12013-011-9265-6
Buchroithner J, Erhart F, Pichler J, Widhalm G, Preusser M, Stockhammer G et al (2018) Audencel Immunotherapy based on dendritic cells has no effect on overall and progression-free survival in newly diagnosed glioblastoma: a phase II randomized trial. Cancers (Basel) 10:372. https://doi.org/10.3390/cancers10100372
Louis DN, Perry A, Wesseling P et al (2021) The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro Oncol 23(8):1231–1251. https://doi.org/10.1093/neuonc/noab106
Karschnia P, Vogelbaum MA, van den Bent M et al (2021) Evidence-based recommendations on categories for extent of resection in diffuse glioma. Eur J Cancer 149:23–33. https://doi.org/10.1016/j.ejca.2021.03.002
Scheffel TB, Grave N, Vargas P, Diz FM, Rockenbach L, Morrone FB (2021) Immunosuppression in gliomas via PD-1/PD-L1 axis and adenosine pathway. Front Oncol 10:617385. https://doi.org/10.3389/fonc.2020.617385
Zhang N, Wei L, Ye M, Kang C, You H (2020) Treatment progress of immune checkpoint blockade therapy for glioblastoma. Front Immunol 11:592612. https://doi.org/10.3389/fimmu.2020.592612
Skaga E, Kulesskiy E, Fayzullin A, Sandberg CJ, Potdar S, Kyttälä A et al (2019) Intertumoral heterogeneity in patient-specific drug sensitivities in treatment-naïve glioblastoma. BMC Cancer 19:628. https://doi.org/10.1186/s12885-019-5861-4
Funding
The study was performed without external funding.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by A.R., V.O., D.R. and Yu.Z. The first draft of the manuscript was written by A.R. and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
This study was conducted in accordance with the principles of the Declaration of Helsinki. Approval was received by the Ethics Committee of the Russian Research Neurosurgical Institute named after Professor A.L. Polenov (Date 03/07/2013/№ 3577).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rynda, A.Y., Rostovthev, D.M., Zabrodskaya, Y.M. et al. Immunotherapy with autologous dendritic cells in the complex treatment of malignant gliomas - results. J Neurooncol 166, 309–319 (2024). https://doi.org/10.1007/s11060-023-04559-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-023-04559-1