Abstract
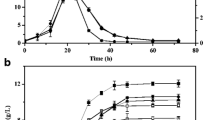
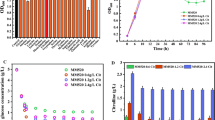
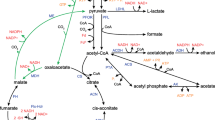
The redox-sensing transcriptional repressor Rex (Rex) displayed diverse functions in different microbial species. Nowadays, only part function of rex has been verified in vitro and alcohol dehydrogenase gene (adhE) as the target of Rex has been widely reported. In this study, rex was knocked out in Thermoanaerobacterium aotearoense SCUT27 (GDMCC 60765) and the carbon metabolic distribution analysis was performed. Results showed that the ethanol yield (mol product/mol carbon) of SCUT27(Δrex) had increased by 75.00–90.91%, cell growth improved by 27.27–36.36%, and acetic acid and lactic acid decreased by 58.33–61.54% accompanied with the yield of hydrogen decreased by 46.15–58.35% within different carbon sources. The ability of sugar consumption of SCUT27(Δrex) had improved about 74.19–130.55% with the improvement of total ATP concentration and the cofactors NADH and NAD+ concentrations. In addition, the specific activities of alcohol dehydrogenase of SCUT27(Δrex) with NADH and NADPH as cofactors were improved by 119.26–140.28% and 35.66–47.69%, respectively. After ldh was further knocked out in SCUT27(Δrex), SCUT27(ΔldhΔrex) showed higher ethanol production and yield when various carbon resources were used as substrates (including glucose, xylose, glucose/xylose mixture and three kinds of lignocellulosic hydrolysates). This study confirms that Rex is an important regulator for determining products distribution in SCUT27 and deletion of rex and ldh is a promising strategy for enhanced ethanol production.





Similar content being viewed by others
References
Ahring BK, Licht D, Schmidt AS, Sommer P, Thomsen AB (1999) Production of ethanol from wet oxidised wheat straw by Thermoanaerobacter mathranii. Bioresour Technol 68(1):3–9
Almarsdottir AR, Sigurbjornsdottir MA, Orlygsson J (2012) Effect of various factors on ethanol yields from lignocellulosic biomass by Thermoanaerobacterium AK17. Biotechnol Bioeng 109(3):686–694. https://doi.org/10.1002/bit.24346
Balat M (2005) Current alternative engine fuels. Energy Sources 27(6):569–577. https://doi.org/10.1080/00908310490450458
Beri D, Olson DG, Holwerda EK, Lynd LR (2016) Nicotinamide cofactor ratios in engineered strains of Clostridium thermocellum and Thermoanaerobacterium saccharolyticum. FEMS Microbiol Lett 363(11). https://doi.org/10.1093/femsle/fnw091
Berriosrivera SJ (2002) Metabolic engineering of cofactors (NADH/NAD+) in Escherichia coli. PhD thesis, Rice University, Houston
Bitoun JP, Liao S, Yao X, Xie GG, Wen ZT (2012) The redox-sensing regulator Rex modulates central carbon metabolism, stress tolerance response and biofilm formation by Streptococcus mutans. PLoS One 7(9):e44766. https://doi.org/10.1371/journal.pone.0044766
Busic A, Mardetko N, Kundas S, Morzak G, Belskaya H, Ivancic Santek M, Komes D, Novak S, Santek B (2018) Bioethanol production from renewable raw materials and its separation and purification: a review. Food Technol Biotechnol 56(3):289–311. https://doi.org/10.17113/ftb.56.03.18.5546
Cai Y, Lai C, Li S, Liang Z, Zhu M, Liang S, Wang J (2011) Disruption of lactate dehydrogenase through homologous recombination to improve bioethanol production in Thermoanaerobacterium aotearoense. Enzym Microb Technol 48(2):155–161. https://doi.org/10.1016/j.enzmictec.2010.10.006
Chen X, Li S, Liu L (2014) Engineering redox balance through cofactor systems. Trends Biotechnol 32(6):337–343. https://doi.org/10.1016/j.tibtech.2014.04.003
Eminoglu A, Murphy SJ, Maloney M, Lanahan A, Giannone RJ, Hettich RL, Tripathi SA, Belduz AO, Lynd LR, Olson DG (2017) Deletion of the hfsB gene increases ethanol production in Thermoanaerobacterium saccharolyticum and several other thermophilic anaerobic bacteria. Biotechnol Biofuels 10:282. https://doi.org/10.1186/s13068-017-0968-9
Fu H, Yang ST, Wang M, Wang J, Tang IC (2017) Butyric acid production from lignocellulosic biomass hydrolysates by engineered Clostridium tyrobutyricum overexpressing xylose catabolism genes for glucose and xylose co-utilization. Bioresour Technol 234:389–396. https://doi.org/10.1016/j.biortech.2017.03.073
Georgieva TI, Ahring BK (2007) Evaluation of continuous ethanol fermentation of dilute-acid corn stover hydrolysate using thermophilic anaerobic bacterium Thermoanaerobacter BG1L1. Appl Microbiol Biotechnol 77(1):61–68. https://doi.org/10.1007/s00253-007-1149-8
Georgieva TI, Mikkelsen MJ, Ahring BK (2008) Ethanol production from wet-exploded wheat straw hydrolysate by thermophilic anaerobic bacterium Thermoanaerobacter BG1L1 in a continuous immobilized reactor. Appl Biochem Biotechnol 145(1–3):99–110. https://doi.org/10.1007/s12010-007-8014-1
Green J, Paget MS (2004) Bacterial redox sensors. Nat Rev Microbiol 2(12):954–966. https://doi.org/10.1038/nrmicro1022
He Q, Lokken PM, Chen S, Zhou J (2009) Characterization of the impact of acetate and lactate on ethanolic fermentation by Thermoanaerobacter ethanolicus. Bioresour Technol 100(23):5955–5965. https://doi.org/10.1016/j.biortech.2009.06.084
Herring CD, Kenealy WR, Shaw AJ, Raman B, Tschaplinski TJ, Brown SD, Davison BH, Covalla SF, Sillers WR, Xu H (2012) Final report on development of Thermoanaerobacterium saccharolyticum for the conversion of lignocellulose to ethanol. US DeP Energy Tech Rep DOE/GO/017057-1
Herring CD, Kenealy WR, Joe Shaw A, Covalla SF, Olson DG, Zhang J, Ryan Sillers W, Tsakraklides V, Bardsley JS, Rogers SR, Thorne PG, Johnson JP, Foster A, Shikhare ID, Klingeman DM, Brown SD, Davison BH, Lynd LR, Hogsett DA (2016) Strain and bioprocess improvement of a thermophilic anaerobe for the production of ethanol from wood. Biotechnol Biofuels 9:125. https://doi.org/10.1186/s13068-016-0536-8
Hon S, Holwerda EK, Worthen RS, Maloney MI, Tian L, Cui J, Lin PP, Lynd LR, Olson DG (2018) Expressing the Thermoanaerobacterium saccharolyticum pforA in engineered Clostridium thermocellum improves ethanol production. Biotechnol Biofuels 11:242. https://doi.org/10.1186/s13068-018-1245-2
Kaparaju P, Serrano M, Thomsen AB, Kongjan P, Angelidaki I (2009) Bioethanol, biohydrogen and biogas production from wheat straw in a biorefinery concept. Bioresour Technol 100(9):2562–2568. https://doi.org/10.1016/j.biortech.2008.11.011
Kim JS, Park SC, Kim JW, Park JC, Park SM, Lee JS (2010) Production of bioethanol from lignocellulose: status and perspectives in Korea. Bioresour Technol 101(13):4801–4805. https://doi.org/10.1016/j.biortech.2009.12.059
Klinke HB, Thomsen AB, Ahring BK (2001) Potential inhibitors from wet oxidation of wheat straw and their effect on growth and ethanol production by Thermoanaerobacter mathranii. Appl Microbiol Biotechnol 57(5–6):631–638
Lee JM, Venditti RA, Jameel H, Kenealy WR (2011) Detoxification of woody hydrolyzates with activated carbon for bioconversion to ethanol by the thermophilic anaerobic bacterium Thermoanaerobacterium saccharolyticum. Biomass Bioenergy 35(1):626–636. https://doi.org/10.1016/j.biombioe.2010.10.021
Lee S, Nam D, Jung JY, Oh MK, Sang BI, Mitchell RJ (2012) Identification of Escherichia coli biomarkers responsive to various lignin-hydrolysate compounds. Bioresour Technol 114:450–456. https://doi.org/10.1016/j.biortech.2012.02.085
Li S, Lai C, Cai Y, Yang X, Yang S, Zhu M, Wang J, Wang X (2010) High efficiency hydrogen production from glucose/xylose by the ldh-deleted Thermoanaerobacterium strain. Bioresour Technol 101(22):8718–8724. https://doi.org/10.1016/j.biortech.2010.06.111
Li HF, Knutson BL, Nokes SE, Lynn BC, Flythe MD (2012) Metabolic control of Clostridium thermocellum via inhibition of hydrogenase activity and the glucose transport rate. Appl Microbiol Biotechnol 93(4):1777–1784. https://doi.org/10.1007/s00253-011-3812-3
Liao Z, Suo Y, Xue C, Fu H, Wang J (2018) Improving the fermentation performance of Clostridium acetobutylicum ATCC 824 by strengthening the VB1 biosynthesis pathway. Appl Microbiol Biotechnol 102(18):8107–8119. https://doi.org/10.1007/s00253-018-9208-x
Lin L, Song H, Tu Q, Qin Y, Zhou A, Liu W, He Z, Zhou J, Xu J (2011) The Thermoanaerobacter glycobiome reveals mechanisms of pentose and hexose co-utilization in bacteria. PLoS Genet 7(10):e1002318. https://doi.org/10.1371/journal.pgen.1002318
Lo J, Zheng T, Hon S, Olson DG, Lynd LR (2015) The bifunctional alcohol and aldehyde dehydrogenase gene, adhE, is necessary for ethanol production in Clostridium thermocellum and Thermoanaerobacterium saccharolyticum. J Bacteriol 197(8):1386–1393. https://doi.org/10.1128/jb.02450-14
Mai V, Lorenz WW, Wiegel J (1997) Transformation of Thermoanaerobacterium sp. strain JW/SL-YS485 with plasmid pIKM1 conferring kanamycin resistance. FEMS Microbiol Lett 148(2):163–167. https://doi.org/10.1111/j.1574-6968.1997.tb10283.x
Miyazaki K, Irbis C, Takada J, Matsuura A (2008) An ability of isolated strains to efficiently cooperate in ethanolic fermentation of agricultural plant refuse under initially aerobic thermophilic conditions: oxygen deletion process appended to consolidated bioprocessing (CBP). Bioresour Technol 99(6):1768–1775. https://doi.org/10.1016/j.biortech.2007.03.045
Moshi AP, Hosea KM, Elisante E, Mamo G, Mattiasson B (2015) High temperature simultaneous saccharification and fermentation of starch from inedible wild cassava (Manihot glaziovii) to bioethanol using Caloramator boliviensis. Bioresour Technol 180:128–136. https://doi.org/10.1016/j.biortech.2014.12.087
Nelson D, Cox M (2005) Lehninger principles of biochemistry. W.H. Freeman; 2005. 947–948
Pagels M, Fuchs S, Pane-Farre J, Kohler C, Menschner L, Hecker M, McNamarra PJ, Bauer MC, von Wachenfeldt C, Liebeke M, Lalk M, Sander G, von Eiff C, Proctor RA, Engelmann S (2010) Redox sensing by a Rex-family repressor is involved in the regulation of anaerobic gene expression in Staphylococcus aureus. Mol Microbiol 76(5):1142–1161. https://doi.org/10.1111/j.1365-2958.2010.07105.x
Palmqvist E, Hahn-Hägerdal B (2000) Fermentation of lignocellulosic hydrolysates. II: inhibitors and mechanisms of inhibition. Bioresour Technol 74(1):25–33. https://doi.org/10.1016/S0960-8524(99)00161-3
Pei J, Zhou Q, Jing Q, Li L, Dai C, Li H, Wiegel J, Shao W (2011) The mechanism for regulating ethanol fermentation by redox levels in Thermoanaerobacter ethanolicus. Metab Eng 13(2):186–193. https://doi.org/10.1016/j.ymben.2010.12.006
Rahayu F, Kawai Y, Iwasaki Y, Yoshida K, Kita A, Tajima T, Kato J, Murakami K, Hoshino T, Nakashimada Y (2017) Thermophilic ethanol fermentation from lignocellulose hydrolysate by genetically engineered Moorella thermoacetica. Bioresour Technol 245(Pt B):1393–1399. https://doi.org/10.1016/j.biortech.2017.05.146
Ravcheev DA, Li X, Latif H, Zengler K, Leyn SA, Korostelev YD, Kazakov AE, Novichkov PS, Osterman AL, Rodionov DA (2012) Transcriptional regulation of central carbon and energy metabolism in bacteria by redox-responsive repressor Rex. J Bacteriol 194(5):1145–1157. https://doi.org/10.1128/jb.06412-11
Shaw AJ, Jenny FE Jr, Adams MWW, Lynd LR (2008a) End-product pathways in the xylose fermenting bacterium, Thermoanaerobacterium saccharolyticum. Enzym Microb Technol 42(6):453–458. https://doi.org/10.1016/j.enzmictec.2009.01.005
Shaw AJ, Podkaminer KK, Desai SG, Bardsley JS, Rogers SR, Thorne PG, Hogsett DA, Lynd LR (2008b) Metabolic engineering of a thermophilic bacterium to produce ethanol at high yield. Proc Natl Acad Sci U S A 105(37):13769–13774. https://doi.org/10.1073/pnas.0801266105
Silveira MH, Morais AR, da Costa Lopes AM, Olekszyszen DN, Bogel-Lukasik R, Andreaus J, Pereira Ramos L (2015) Current pretreatment technologies for the development of cellulosic ethanol and biorefineries. ChemSusChem 8(20):3366–3390. https://doi.org/10.1002/cssc.201500282
Sudha RK, Swamy MV, Seenayya G (1998) Production of ethanol from various pure and natural cellulosic biomass by Clostridium thermocellum strains SS21 and SS22. Process Biochem 33(4):435–440. https://doi.org/10.1016/s0032-9592(97)00095-2
Suo Y, Fu H, Ren M, Yang X, Liao Z, Wang J (2018) Butyric acid production from lignocellulosic biomass hydrolysates by engineered Clostridium tyrobutyricum overexpressing class I heat shock protein GroESL. Bioresour Technol 250:691–698. https://doi.org/10.1016/j.biortech.2017.11.059
Sveinsdottir M, Baldursson SRB, Orlygsson J (2009) Ethanol production from monosugars and lignocellulosic biomass by thermophilic bacteria isolated from Icelandic hot springs. Icel Agric Sci 22(62):45–58
Taylor MP, Eley KL, Martin S, Tuffin MI, Burton SG, Cowan DA (2009) Thermophilic ethanologenesis: future prospects for second-generation bioethanol production. Trends Biotechnol 27(7):398–405. https://doi.org/10.1016/j.tibtech.2009.03.006
Weimer PJ, Zeikus JG (1977) Fermentation of cellulose and cellobiose by Clostridium thermocellum in the absence of Methanobacterium thermoautotrophicum. Appl Environ Microbiol 33(2):289–297
Wietzke M, Bahl H (2012) The redox-sensing protein Rex, a transcriptional regulator of solventogenesis in Clostridium acetobutylicum. Appl Microbiol Biotechnol 96(3):749–761. https://doi.org/10.1007/s00253-012-4112-2
Zhang L, Nie X, Ravcheev DA, Rodionov DA, Sheng J, Gu Y, Yang S, Jiang W, Yang C (2014) Redox-responsive repressor Rex modulates alcohol production and oxidative stress tolerance in Clostridium acetobutylicum. J Bacteriol 196(22):3949–3963. https://doi.org/10.1128/jb.02037-14
Zheng Y, Ko T-P, Sun H, Huang C-H, Pei J, Qiu R, Wang AHJ, Wiegel J, Shao W, Guo R-T (2014) Distinct structural features of Rex-family repressors to sense redox levels in anaerobes and aerobes. J Struct Biol 188(3):195–204. https://doi.org/10.1016/j.jsb.2014.11.001
Zheng T, Olson DG, Murphy SJ, Shao X, Liang T, Lynd LR (2017) Both adhE and a separate NADPH-dependent alcohol dehydrogenase gene, adhA, are necessary for high ethanol production in Thermoanaerobacterium saccharolyticum. J Bacteriol 199(3). https://doi.org/10.1128/JB.00542-16
Zheng T, Lanahan AA, Lynd LR, Olson DG (2018) The redox-sensing protein Rex modulates ethanol production in Thermoanaerobacterium saccharolyticum. PLoS One 13(4):e0195143. https://doi.org/10.1371/journal.pone.0195143
Zhou J, Ouyang J, Xu Q, Zheng Z (2016) Cost-effective simultaneous saccharification and fermentation of l-lactic acid from bagasse sulfite pulp by Bacillus coagulans CC17. Bioresour Technol 222:431–438. https://doi.org/10.1016/j.biortech.2016.09.119
Zhou JJ, Shen JT, Wang XL, Sun YQ, Xiu ZL (2018) Stability and oscillatory behavior of microbial consortium in continuous conversion of crude glycerol to 1,3-propanediol. Appl Microbiol Biotechnol 102(19):8291–8305. https://doi.org/10.1007/s00253-018-9244-6
Zhu M, Lu Y, Wang J, Li S, Wang X (2015) Carbon catabolite repression and the related genes of ccpA, ptsH and hprK in Thermoanaerobacterium aotearoense. PLoS One 10(11):e0142121. https://doi.org/10.1371/journal.pone.0142121
Funding
This work was supported by the National Natural Science Foundation of China (21878103, 21676098) and the Natural Science Foundation of Guangdong Province of China (2018A030310368).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 4955 kb)
Rights and permissions
About this article
Cite this article
Qu, C., Chen, L., Li, Y. et al. The redox-sensing transcriptional repressor Rex is important for regulating the products distribution in Thermoanaerobacterium aotearoense SCUT27. Appl Microbiol Biotechnol 104, 5605–5617 (2020). https://doi.org/10.1007/s00253-020-10554-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10554-7