Abstract
Introduction
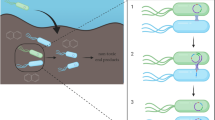
Naphthalene catabolic strain Pseudomonas putida ND6 harbors two megaplasmids pND6–1 and pND6–2; naphthalene-degrading associated genes are located on pND6–1, while plasmid pND6–2 possesses potential coding genes for conjugative transfer.
Methods
In this study, the recombinant ND6 in which pND6–1 and pND6–2 were labeled with gentamicin and kanamycin-encoding genes, respectively, was constructed to investigate the conjugative transfer behavior of the two plasmids in distinct aqueous matrices.
Results
The results indicated that both pND6–1 and pND6–2 plasmids could transfer from donor strain ND6 to a recipient strain P. putida KT2440, while the transfer frequency of pND6–2 (1.90 × 10− 2) was significantly higher than that of pND6–1 (3.12 × 10− 9). Furthermore, the concomitant transfer of pND6–1 and pND6–2 was confirmed at a lower frequency of 10− 9 colonies per recipient similar with that of pND6–1. The conjugative transfer efficiency was obviously affected by the initial inoculum and the stability of microbes. Moreover, more than 90% of the transconjugants lost the plasmids after 20 generations cultivation without resistance pressure, suggesting the importance of selective pressure to maintain the plasmid stability. Finally, the naphthalene degradation analysis by mixed ND6 and KT2440 revealed that conjugative transfer of catabolic plasmids contributed to the rapid dispersion of the degradation genes on plasmids and enhanced the naphthalene removal.




Similar content being viewed by others
References
Barkay T, Kroer N, Rasmussen LD, Sørensen SJ (2010) Conjugal transfer at natural population densities in a microcosm simulating an estuarine environment. FEMS Microbiol Ecol 16(1):43–54. https://doi.org/10.1111/j.1574-6941.1995.tb00267.x
Bellanger X, Guilloteau H, Bonot S, Merlin C (2014) Demonstrating plasmid-based horizontal gene transfer in complex environmental matrices: a practical approach for a critical review. Sci Total Environ 493C(493C):872–882. https://doi.org/10.1016/j.scitotenv.2014.06.070
Bishnoi K, Kumar R, Bishnoi NR (2008) Biodegradation of polycyclic aromatic hydrocarbons by white rot fungi Phanerochaete chrysosporium in sterile and unsterile soil. J Sci Ind Res India 67(7):538–542 https://doi.org/10.1061/(ASCE)1052-3928(2008)134:3(315)
Dennis JJ, Zylstra GJ (2004) Complete sequence and genetic organization of pDTG1, the 83 kilobase naphthalene degradation plasmid from Pseudomonas putida strain NCIB 9816-4. J Mol Biol 341(3):753–768. https://doi.org/10.1016/j.jmb.2004.06.034
Fang F, Sarah F, Yin L, Claesson MJ, Jan-Peter VP, Collins JK, Douwe VS, O’Toole PW (2008) Characterization of endogenous plasmids from Lactobacillus salivarius UCC118. Appl Environ Microbiol 74(10):3216–3228. https://doi.org/10.1128/AEM.02631-07
Gama JA, Zilhao R, Dionisio F (2017) Co-resident plasmids travel together. Plasmid 93:24–29. https://doi.org/10.1016/j.plasmid.2017.08.004
Garbisu C, Garaiyurrebaso O, Epelde L, Grohmann E, Alkorta I (2017) Plasmid-mediated bioaugmentation for the bioremediation of contaminated soils. Front Microbiol 8:1966. https://doi.org/10.3389/fmicb.2017.01966
Hall EA, Sarkar MR, Bell SG (2017) The selective oxidation of substituted aromatic hydrocarbons and the observation of uncoupling via redox cycling during naphthalene oxidation by the CYP101B1 system. Catal Sci Technol 7(7):1537–1548. https://doi.org/10.1039/c7cy00088j
Itaya M, Sakaya N, Matsunaga S, Fujita K, Kaneko S (2014) Conjugational transfer kinetics of pLS20 between Bacillus subtilisin liquid medium. Biosci Biotechnol Biochem 70(3):740–742. https://doi.org/10.1271/bbb.70.740
Jain R, Rivera MC, Moore JE, Lake JA (2003) Horizontal gene transfer accelerates genome innovation and evolution. Mol Biol Evol 20(10):1598. https://doi.org/10.1093/molbev/msg154
Jiao Y-N, Chen H, Gao R-X, Zhu Y-G, Rensing C (2017) Organic compounds stimulate horizontal transfer of antibiotic resistance genes in mixed wastewater treatment systems. Chemosphere 184:53–61. https://doi.org/10.1016/j.chemosphere.2017.05.149
Jiménez JI, Miñambres B, Garcia JL, Diaz E (2002) Genomic analysis of the aromatic catabolic pathways from Pseudomonas putida KT2440. Environ Microbiol 4(12):824–841. https://doi.org/10.1046/j.1462-2920.2002.00370.x
Khan NA, Engle M, Dungan B, Holguin FO, Xu P, Carroll KC (2016) Volatile-organic molecular characterization of shale-oil produced water from the Permian Basin. Chemosphere 148:126–136. https://doi.org/10.1016/j.chemosphere.2015.12.116
Kishida K, Inoue K, Ohtsubo Y, Nagata Y, Tsuda M (2017) Host range of the conjugative transfer system of IncP-9 naphthalene-catabolic plasmid NAH7 and characterization of its oriT region and relaxase. Appl Environ Microbiol 83(1):AEM.02359–AEM.02316. https://doi.org/10.1128/AEM.02359-16
Kovach ME, Elzer PH, Hill DS, Robertson GT, Farris MA, Roop RM, Peterson KM (1995) Four new derivatives of the broad-host-range cloning vector pBBR1MCS, carrying different antibiotic-resistance cassettes. Gene 166 (1):175–176. https://doi.org/10.1016/0378-1119(95)00584-1
Lee S, Lim WA, Thorn KS (2013) Improved blue, green, and red fluorescent protein tagging vectors for S. cerevisiae. PLoS One 8(7):e67902. https://doi.org/10.1371/journal.pone.0067902
Lerminiaux NA, Cameron ADS (2019) Horizontal transfer of antibiotic resistance genes in clinical environments. Can J Microbiol 65(1):34–44. https://doi.org/10.1139/cjm-2018-0275
Lessard JC (2013) Molecular cloning. In: Lorsch J (ed) laboratory methods in enzymology: DNA. Methods Enzymol 529:85–98. https://doi.org/10.1016/B978-0-12-418687-3.00007-0
Li W, Shi JD, Wang XG, Han YN, Tong W, Ma L, Liu B, Cai BL (2004) Complete nucleotide sequence and organization of the naphthalene catabolic plasmid pND6-1 from Pseudomonas sp. strain ND6. Gene 336(2):231–240. https://doi.org/10.1016/j.gene.2004.03.027
Li SS, Li X, Zhao HB, Cai BL (2011) Physiological role of the novel salicylaldehyde dehydrogenase NahV in mineralization of naphthalene by Pseudomonas putida ND6. Microbiol Res 166(8):643–653. https://doi.org/10.1016/j.micres.2011.01.003
Li S, Zhao H, Li Y, Niu S, Cai B (2012) Complete genome sequence of the naphthalene-degrading Pseudomonas putida strain ND6. J Bacteriol 194(18):5154–5155. https://doi.org/10.1128/jb.01190-12
Li S, Zhao H, Li Y, Niu S, Cai B (2013) Complete nucleotide sequence of plasmid pND6-2 from Pseudomonas putida ND6 and characterization of conjugative genes. Gene 512(1):148–156. https://doi.org/10.1016/j.gene.2012.09.065
Long H, Feng Y, Ma K, Liu L, McNally A, Zong Z (2019) The co-transfer of plasmid-borne colistin-resistant genes mcr-1 and mcr-3.5, the carbapenemase gene bla (NDM-5) and the 16S methylase gene rmtB from Escherichia coli. Sci Rep 9:696. https://doi.org/10.1038/s41598-018-37125-1
Masaki S, Hirokazu Y, Hiroshi H, Toshio O, Hisakazu Y, Masataka T, Hideaki N (2006) Characterization of the replication, maintenance, and transfer features of the IncP-7 plasmid pCAR1, which carries genes involved in carbazole and dioxin degradation. Appl Environ Microbiol 72(5):3206–3216. https://doi.org/10.1128/AEM.02359-16
Miyazaki R, Ohtsubo Y, Nagata Y, Tsuda M (2008) Characterization of the traD operon of naphthalene-catabolic plasmid NAH7: a host-range modifier in conjugative transfer. J Bacteriol 190(19):6281–6289. https://doi.org/10.1128/jb.00709-08
Nakamura N, Tanaka C, Takeuchi-Kaneko Y (2019) Transmission of antibiotic-resistance markers by hyphal fusion suggests partial presence of parasexuality in the root endophytic fungus Glutinomyces brunneus. Mycol Prog 18(3):453–462. https://doi.org/10.1007/s11557-018-1455-9
Nelson KE (2002) The complete genome sequence of Pseudomonas putida KT2440 is finally available. Environ Microbiol 4 (12):777–778. https://doi.org/10.1046/j.1462-2920.2002.00367.x
Obeng EM, Brossette T, Ongkudon CM, Budiman C, Maas R, Jose J (2018) The workability of Escherichia coli BL21 (DE3) and Pseudomonas putida KT2440 expression platforms with autodisplayed cellulases: a comparison. Appl Microbiol Biotechnol 102(11):1–13. https://doi.org/10.1007/s00253-018-8987-4
Pei R, Gunsch CK (2009) Plasmid conjugation in an activated sludge microbial community. Environ Eng Sci 26(4):825–831. https://doi.org/10.1089/ees.2008.0236
Peng P, Yang H, Li L, Jia R (2013) Biodegradation of dioxin by a newly isolated Rhodococcus sp. with the involvement of self-transmissible plasmids. Appl Microbiol Biotechnol 97(12):5585–5595. https://doi.org/10.1007/s00253-012-4363-y
Pozzi EA, Schwall LR, Jiménez R, Weber JM (2012) Pressure-induced changes in the fluorescence behavior of red fluorescent proteins. J Phys Chem B 116(34):10311–10316. https://doi.org/10.1021/jp306093h
Shang R-F, Wang G-H, Xu X-M, Liu S-J, Zhang C, Yi Y-P, Liang J-P, Liu Y (2014) Synthesis and biological evaluation of new pleuromutilin derivatives as antibacterial agents. Molecules 19(11):19050–19065. https://doi.org/10.3390/molecules191119050
Shintani M, Fukushima N, Tezuka M, Yamane H, Nojiri H (2008) Conjugative transfer of the IncP-7 carbazole degradative plasmid, pCAR1, in river water samples. Biotechnol Lett 30(1):117–122. https://doi.org/10.1007/s10529-007-9519-y
Shintani M, Takahashi Y, Yamane H, Nojiri H (2010) The behavior and significance of degradative plasmids belonging to Inc groups in Pseudomonas within natural environments and microcosms. Microbes Environ 25(4):253–265. https://doi.org/10.1264/jsme2.ME10155
Simon R, Priefer U, Pühler A (1983) A broad host range mobilization system for in vivo genetic engineering: transposon mutagenesis in gram negative bacteria. Nat Biotechnol 1 (9):784–791. https://doi.org/10.1038/nbt1183-784
Smets BF, Barkay T (2005) Horizontal gene transfer: perspectives at a crossroads of scientific disciplines. Nat Rev Microbiol 3(9):675–678. https://doi.org/10.1038/nrmicro1253
Sota M, Yano H, Ono A, Miyazaki R, Ishii H, Genka H, Top EM, Tsuda M (2006) Genomic and functional analysis of the IncP-9 naphthalene-catabolic plasmid NAH7 and its transposon Tn4655 suggests catabolic gene spread by a tyrosine recombinase. J Bacteriol 188(11):4057–4067. https://doi.org/10.1128/jb.00185-06
Summers DK, Sherratt DJ (1985) Bacterial plasmid stability. Bioessays 2(5):209–211. https://doi.org/10.1002/bies.950020506
Sun J, Qiu Y, Ding P, Peng P, Yang H, Li L (2017) Conjugative transfer of dioxin–catabolic megaplasmids and bioaugmentation prospects of a Rhodococcus sp. Environ Sci Technol 51(11):6298–6307. https://doi.org/10.1021/acs.est.7b00188
Walter WG (1961) Standard methods for the examination of water and wastewater (11th ed.). Am Pub Health Ass 51(6):940. https://doi.org/10.2105/AJPH.56.4.684-a
Watanabe T, Takeda K (2017) Dynamic promotion and suppression model for plasmid conjugal transfer under a flow condition. Recent Global Research and Education: Technological Challenges. Springer International Publishing, p 203–209
Yang Y, Wang J, Liao J, Xie S, Huang Y (2014) Distribution of naphthalene dioxygenase genes in crude oil-contaminated soils. Microb Ecol 68(4):785. https://doi.org/10.1007/s00248-014-0457-7
Yano H, Miyakoshi M, Ohshima K, Tabata M, Nagata Y, Hattori M, Tsuda M (2010) Complete nucleotide sequence of TOL plasmid pDK1 provides evidence for evolutionary history of IncP-7 catabolic plasmids. J Bacteriol 192(17):4337–4347. https://doi.org/10.1128/jb.00359-10
Yano T, Sanders C, Catalano J, Daldal F (2005) sacB-5-Fluoroorotic acid-pyrE-based bidirectional selection for integration of unmarked alleles into the chromosome of Rhodobacter capsulatus. Appl Environ Microbiol 71(6):3014–3024 https://doi.org/10.1128/AEM.71.6.3014-3024.2005
Zhao HB, Chen DF, Li YJ, Cai BL (2005) Overexpression, purification and characterization of a new salicylate hydroxylase from naphthalene-degrading Pseudomonas sp. strain ND6. Microbiol Res 160(3):307–313. https://doi.org/10.1016/j.micres.2005.02.004
Funding
This work was supported by the National Natural Science Foundation of China (Grant NO. 31670512), Natural Science Basic Research Plan in Shaanxi Province of China (Program No. 2018JM3039).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 202 kb)
Rights and permissions
About this article
Cite this article
Wang, S., Li, S., Du, D. et al. Conjugative transfer of Megaplasmids pND6–1 and pND6–2 enhancing naphthalene degradation in aqueous environment: characterization and bioaugmentation prospects. Appl Microbiol Biotechnol 104, 861–871 (2020). https://doi.org/10.1007/s00253-019-10273-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-10273-8