Abstract
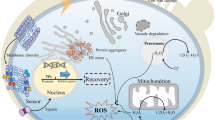
Inhibitory compounds liberated from lignocellulose pretreatment are representative toxic chemicals that repress microbial growth and metabolism. A tolerant strain of the industrial yeast Saccharomyces cerevisiae is able to detoxify a major class of toxic compounds while producing ethanol. Knowledge on the yeast tolerance was mostly obtained by gene expression analysis and limited protein expression evidence is yet available underlying the yeast adaptation. Here we report a comparative protein expression profiling study on Y-50049, a tolerant strain compared with its parental industrial type strain Y-12632. We found a distinctive protein expression of glucose-6-phosphate dehydrogenase (Zwf1) in Y-50049 but not in Y-12632, in the relatively conserved glycolysis and pentose phosphate pathway (PPP) in response to a combinational challenge of 2-furaldehyde (furfural) and 5-hydroxymethyl-2-furaldehyde (HMF). A group of proteins with aldehyde reduction activity was uniquely induced expressed in Y-50049 but not in Y-12632. Such evidence allowed fine-tuning a mechanism of the renovated in situ detoxification by Y-50049. As the key protein, Zwf1 drove the glucose metabolism in favor of the oxidative branch of the PPP facilitating in situ detoxification of the toxic chemicals by Y-50049. The activated expression of Zwf1 generated the essential cofactor nicotinamide adenine dinucleotide phosphate (NADPH) enabling reduction of furfural and HMF through a group of aldehyde reduction enzymes. In return, the activate aldehyde reductions released desirable feedbacks of NADP+ stimulating continued oxidative activity of Zwf1. Thus, a well-maintained cofactor regeneration cycle was established to restore the cofactor imbalance caused by furfural-HMF. Challenges and perspectives on adaptation of significantly differential expressions of ribosomal proteins and other unique proteins are also discussed.



Similar content being viewed by others
References
Allen SA, Clark W, McCAffery JM, Cai Z, Lanctot A, Slininger PJ, Liu ZL, Gorsich SW (2010) Furfural induces reactive oxygen species accumulation and cellular damage in Saccharomyces cerevisiae. Biotechnol Biofuels 3:2
Avery AM, Willetts SA, Avery SV (2004) Genetic dissection of the phospholipid hydroperoxidase activity of yeast gpx3 reveals its functional importance. J Bio Chem 279:46652–46658
Ban N, Beckmann R, Cate JHD, Dinman JD, Dragon F, Ellis SR, Lafontaine DLJ, Lindahl L, Liljas A, Lipton JM, McAlear MA, Moore PB, Noller HF, Ortega J, Panse VG, Ramakrishnan V, Spahn CMT, Steitz TA, Tchorzewski M, Tollervey D, Warren AJ, Williamson JR, Wilson D, Yonath A, Yusupov M (2014) A new system for naming ribosomal proteins. Curr Opin Struct Biol 24:165–169
Batth TS, Keasling JD, Petzold CJ (2012) Targeted proteomics for metabolic pathway optimization In: Keller N, Turner G, editors. Fungal secondary metabolism, methods in molecular biology (methods and protocols), Vol. 944, Humana Press Totowa. NJ 2012:237–249
Berger S, Kouzarides T, Shiekhattar R, Shilatifard A (2009) An operational definition of epigenetics. Genes Deve 23:781–783
Bowman MJ, Jordan DB, Vermillion KE, Braker JD, Moon J, Liu ZL (2010) Stereochemistry of furfural reduction by an aldehyde reductase from Saccharomyces cerevisiae that contributes to in situ furfural detoxification. Appl Environ Microbiol 76:4926–4932
Bradbury JE, Richards KD, Niederer HA, Lee SA, Dunbar PR, Gardner RC (2006) A homozygous diploid subset of commercial wine yeast strains. Antonie van Leeuwenhoek 89:27–37
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brooks AN, Turkarslan S, Beer KD, Lo FY, Baliga NS (2011) Adaptation of cells to new environments. Wiley Interdiscip Rev Syst Biol Med 3:544–561
Causton HC, Ren B, Koh SS, Harbison CT, Kanin E, Jennings EG, Lee TI, True HL, Lander ES, Young RA (2001) Remodeling of yeast genome expression in response to environmental changes. Mol Biol Cell 12:323–337
De la Cruz J, Karbstein K, Woodford JL Jr (2015) Functions of ribosomal proteins in assembly of eukaryotic ribosome in vivo. Annu Rev Biochem 84:93–129
Delaunay A, Pflieger D, Barrault MB, Vinh J, Toledano MB (2002) A thiol peroxidase is an H2O2 receptor and redox-transducer in gene activation. Cell 111:471–481
Fedoroff NV (2012) Transposable elements, epigenetics, and genome evolution. Sci 338:758–767
Fleischer TC, Weaver CM, McAfee KJ, Jennings JL, Link AJ (2006) Systematic identification and functional screens of uncharacterized proteins associated with eukaryotic ribosomal complexes. Genes Dev 20:1294–1307
Ford G, Ellis EM (2002) Characterization of Ypr1p from Saccharomyces cerevisiae as a 2-methylbutyraldehyde reductase. Yeast 19:1087–1096
Gorsich SW, Dien BS, Nichols NN, Slininger PJ, Liu ZL, Skory CD (2006) Tolerance to furfural-induced stress is associated with pentose phosphate pathway genes ZWF1, GND1, RPE1, and TKL1 in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 71:339–349
Grunstein M, Gasser S (2013) Epigenetics in Saccharomyces cerevisiae. Cold Spring Harb Perspect Biol 5:a017491
Hector RE, Bowman MJ, Skory CD, Cotta MA (2009) The Saccharomyces cerevisiae YMR315W gene encodes an NADP(H)-specific oxidoreductase regulated by the transcription factor Stb5p in response to NADPH limitation. New Biotechnol 26:171–180
Heer D, Heine D, Sauer U (2009) Resistance of Saccharomyces cerevisiae to high concentrations of furfural is based on NADPH-dependent reduction by at least two oxireductases. Appl Environ Microbiol 75:7631–7638
Huang X (2017) Data, sequence analysis, and evolution. In: Keith JM (ed) Bioinformatics, vol 1. Humana Press, Totowa, pp 35–45
Huang X, Adams MD, Zhou H, Kerlavage AR (1997) A tool for analyzing and annotating genomic sequences. Genomics 46:37–45
Huang X, Ye L, Chou HH, Yang IH, Chao KM (2004) Efficient combination of multiple word models for improved sequence comparison. Bioinformatics 20:2529–2533
Jordan D, Braker JD, Bowman MJ, Vermillion KE, Moon J, Liu ZL (2011) Kinetic mechanism of an aldehyde reductase of Saccharomyces cerevisiae that relieves toxicity of furfural and 5-hydroxymethylfurfural. Bioch Biophysica Acta 1814:1686–1694
Jung YH, Kim S, Yang J, Seo JH, Kim KH (2017) Intracellular metabolite profiling of Saccharomyces cerevisiae evolved under furfural. Microb Biotechnol 10:395–404
Klinke HB, Thomsen AB, Ahring BK (2004) Inhibition of ethanol-producing yeast and bacteria by degradation products produced during pre-treatment of biomass. Appl Microbiol Biotechnol 66:10–26
Langmead B, Salzberg S (2012) Fast gapped-read alignment with Bowtie 2. Nature Met 9:357–359
Lecompte O, Ripp R, Thierry JC, Moras D, Poch O (2002) Comparative analysis of ribosomal proteins in complete genomes: an example of reductive evolution at the domain scale. Nucleic Acids Res 30:24
Lin FM, Qiao B, Yuan YJ (2009a) Comparative proteomic analysis of tolerance and adaptation of ethanologenic Saccharomyces cerevisiae to furfural, a lignocellulosic inhibitory compound. Appl Environ Microbiol 75:3765–3776
Lin FM, Tan Y, Yuan YJ (2009b) Temporal quantitative proteomics of Saccharomyces cerevisiae in response to a nonlethal concentration of furfural. Proteomics 9:5471–5483
Liu ZL (2006) Genomic adaptation of ethanologenic yeast to biomass conversion inhibitors. Appl Microbiol Biotechnol 73:27–36
Liu ZL (2011) Molecular mechanisms of yeast tolerance and in situ detoxification of lignocellulose hydrolysates. Appl Microbiol Biotechnol 90:809–825
Liu ZL (2018) Understanding the tolerance of the industrial yeast Saccharomyces cerevisiae against a major class of toxic aldehyde compounds. Appl Microbiol Biotechnol 102:5369–5390
Liu ZL, Blaschek HP (2010) Biomass conversion inhibitors and in situ detoxification. In: Vertes A, Qureshi N, Yukawa H, Blaschek H (eds) Biomass to biofuels: strategies for global industries. Wiley, Chichester, pp 233–259
Liu ZL, Ma M, Song M (2009) Evolutionary engineered ethanologenic yeast detoxifies lignocellulosic biomass conversion inhibitors by reprogrammed pathways. Mol Genet Genomics 282:233–244
Liu ZL, Moon J (2009) A novel NADPH-dependent aldehyde reductase gene from Saccharomyces cerevisiae NRRL Y-12632 involved in the detoxification of aldehyde inhibitors derived from lignocellulosic biomass conversion. Gene 446:1–10
Liu ZL, Moon J, Andersh BJ, Slininger PJ, Weber SA (2008) Multiple gene-mediated NAD(P)H-dependent aldehyde reduction is a mechanism of in situ detoxification of furfural and 5-hydroxymethylfurfurl by Saccharomyces cerevisiae. Appl Microbiol Biotechnol 81:743–753
Liu ZL, Slininger PJ, Dien BS, Berhow MA, Kurtzman CP, Gorsich SW (2004) Adaptive response of yeasts to furfural and 5-hydroxymethylfurfural and new chemical evidence for HMF conversion to 2,5-bis-hydroxymethylfuran. J Ind Microbiol Biotechnol 31:345–352
Liu ZL, Slininger PJ, Gorsich SW (2005) Enhanced biotransformation of furfural and hydroxymethylfurfural by newly developed ethanologenic yeast strains. Appl Biochem Biotechnol 121-124:451–460
Liu ZL, Wang X, Webr SA (2018) Tolerant industrial yeast Saccharomyces cerevisiae possess a more robust cell wall integrity signaling pathway against 2-furaldehyde and 5-(hydroxymethyl)-2-furaldehyde. J Biotechnol 276-277:15–24
Llobell A, Lopez-Ruiz A, Peinado J, Lopez-Barea J (1988) Glutathione reductase directly mediates the stimulation of yeast glucose-6-phosphate dehydrogenase by GSSG. Biochem 249:293–296
Lu H, Zhu Y, Xiong J, Wang R, Jia Z (2015) Potential extra-ribosomal functions of ribosomal proteins in Saccharomyces cerevisiae. Microbiol Res 177:28–33
Luo C, Brink D, Blanch H (2002) Identification of potential fermentation inhibitors in conversion of hybrid poplar hydrolyzate to ethanol. Biomass Bioenergy 22:125–138
Ma LH, Takanishi CL, Wood MJ (2007) Molecular mechanism of oxidative stress perception by the Orp1 protein. J Biol Chem 282:1429–1436
Ma M, Liu ZL (2010) Comparative transcriptome profiling analyses during the lag phase uncover YAP1, PDR1, PDR3, RPN4 and HSF1 as key regulatory genes in genomic adaptation to the lignocellulose derived inhibitor HMF for Saccharomyces cerevisiae. BMC Genomics 11:660
Moon J, Liu ZL (2015) Direct enzyme assay evidence confirms aldehyde reductase function of Ydr541cp and Ygl039wp from Saccharomyces cerevisiae. Yeast 32:399–407
Moon J, Liu ZL (2012) Engineered NADH-dependent GRE2 from Saccharomyces cerevisiae by direct enzyme evolution enhances HMF reduction using additional cofactor NADPH. Enzyme Microb Technol 50:115–120
Nikolay R, van den Bruck D, Achenbach J, Nierhaus KH (2015) Ribosomal proteins: role in ribosomal functions. In: eLS. John Wiley & Sons Chichester http://www.els.net 2015.
Palmqvist E, Hahn-Hägerdal B (2000) Fermentation of lignocellulosic hydrolysates II: inhibitors and mechanisms of inhibition. Bioresour Technol 74:25–33
Reinders J, Zahedi RP, Pfanner N, Meisinger C, Sickmann A (2006) Toward the complete yeast mitochondrial proteome: multidimensional separation techniques for mitochondrial proteomics. J Proteome Res 5:1543–1554
Robinson JT, Thorvaldsdottir H, Winckler W, Guttman M, Lander ES, Getz G, Mesirov JP (2011) Integrative genomics viewer. Nature Biotechnol 29:24–26
Sehnem NT, Machado AS, FCB L, WdeB P, Morais MAM Jr, MAZ A (2013) 5-Hydoxymethylfurfural induced ADH7 and ARI1 expression in tolerant industrial Saccharomyces cerevisiae strain P6H9 during bioethanol production. Bioresour Technol 133:190–196
Steffen KK, McCormick MA, Pham KM, MacKay VL, Delaney JR, Murakami CJ, Kaeberlein M, Kennedy BK (2012) Ribosome deficiency protects against ER stress in Saccharomyces cerevisiae. Genetics 191:107–118
Warner JR, Mclntosh KB (2009) How common are extraribosomal functions of ribosomal proteins? Mol Cell 34:3–11
Woodford JL Jr, Baserga SJ (2013) Ribosome biogenesis in the yeast Saccharomyces cerevisiae. Genetics 195:643–681
Wool IG (1966) Extraribosomal functions of ribosomal proteins. Trends Biochem Sci 21:164–165
Yang J, Ding MZ, Li BZ, Liu ZL, Wang X, Yuan YJ (2012) Integrated phospholipidomics and transcriptomics analysis of Saccharomyces cerevisiae with enhanced tolerance to a mixture of acetic acid, furfural and phenol. OMICS J Integ Biol 16:374–386
Zhang Y, Liu ZL, Song M (2015) ChiNet uncovers rewired transcription subnetworks in tolerant yeast for advanced biofuels conversion. Nucleic Acids Res 43:4393–4407
Zhou Q, Liu ZL, Ning K, Wang A, Zeng X, Xu J (2014) Genomic and transcriptome analysis reveal that MAPK- and phosphatidylinositol-signaling pathways mediate tolerance to 5-hydroxymehyl-2-furaldehyde for industrial yeast Saccharomyces cerevisiae. Sci. Reports 4:6556
Zhou X, Liao WJ, Lia JM, Liao P, Lu H (2015) Ribosomal proteins: functions beyond the ribosome. J Mol Cell Biol 7:92–104
Acknowledgments
The authors are extremely grateful to the kind gift from Christopher Petzold at Joint BioEnergy Institute, US Department of Energy, for the protein detection applying their in-house developed LC-MS/MS technology. Without their generous support and the reproducible data obtained, this study would be impossible. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture. USDA is an equal opportunity provider and employer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 752 kb)
Rights and permissions
About this article
Cite this article
Liu, Z.L., Huang, X., Zhou, Q. et al. Protein expression analysis revealed a fine-tuned mechanism of in situ detoxification pathway for the tolerant industrial yeast Saccharomyces cerevisiae. Appl Microbiol Biotechnol 103, 5781–5796 (2019). https://doi.org/10.1007/s00253-019-09906-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-09906-9