Abstract
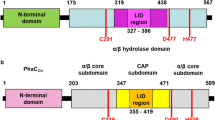
Poly[(R)-3-hydroxybutyrate] (PHB) depolymerase from Ralstonia pickettii T1 (PhaZRpiT1) consists of three functional domains to effectively degrade solid PHB materials, and its catalytic domain catalyzes the ester bond cleavage of the substrate. We performed the directed evolution of PhaZRpiT1 targeted at the catalytic domain in combination with the cell surface display method to effectively screen for mutants with improved p-nitrophenyl butyrate (pNPC4) activity. Mutated PhaZRpiT1 genes generated by error-prone PCR were fused to the oprI gene to display them as fusion proteins on Escherichia coli cell surface. Some cells displaying the mutant enzymes showed a two- to fourfold increase in pNPC4 hydrolysis activity relative to cells displaying wild-type enzyme. These mutant genes were recombined by a staggered extension process and the recombined enzymes were displayed to result in a five- to eightfold higher pNPC4 hydrolysis activity than the wild type. To further evaluate the mutation effects, unfused and undisplayed enzymes were prepared and applied to the hydrolysis of p-nitrophenyl esters having different chain lengths (pNPCn; n = 2–6) and PHB degradation. One specific second-generation mutant showed an approximately tenfold increase in maximum rate for pNPC3 hydrolysis, although its PHB degradation efficiency at 1 μg/mL of enzyme concentration was approximately 3.5-fold lower than that of the wild type. Gene analysis showed that N285D or N285Y mutations were found in six of the seven improved second-generation mutants, indicating that Asn285 probably participates in the regulation of substrate recognition and may be more favorable for PHB degradation process than other amino acid residues.








Similar content being viewed by others
References
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modeling. Bioinformatics 22:195–201
Bachmann BM, Seebach D (1999) Investigation of the enzymatic cleavage of diastereomeric oligo(3-hydroxybutanoates) containing two to eight HB units. A model for the stereoselectivity of PHB depolymerase from Alcaligenes faecalis T1. Macromolecules 32:1777–1784
Benkert P, Biasini M, Schwede T (2011) Toward the estimation of the absolute quality of individual protein structure models. Bioinformatics 27:343–350
Berg OG, Cajal Y, Butterfoss GL, Grey RL, Alsina MA, Yu BZ, Jain MK (1998) Interfacial activation of triglyceride lipase from Thermomyces (Humicola) lanuginosa: kinetic parameters and a basis for control of the lid. Biochemistry 37:6615–6627
Bloom JD, Meyer MM, Meinhold P, Otey CR, Macmillan D, Arnold FH (2005) Evolving strategies for enzyme engineering. Curr Opin Struc Biol 15:447–452
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Chen GQ (2009) A microbial polyhydroxyalkanoates (PHA) based bio- and materials industry. Chem Soc Rev 38:2434–2446
Fan Y, Nishida H, Mori T, Shirai Y, Endo T (2004) Thermal degradation of poly(l-lactide): effect of alkali earth metal oxides for selective l,l-lactide formation. Polymer 45:1197–1205
Fan Y, Fang W, Xiao Y, Yang X, Zhang Y, Bidochka MJ, Pei Y (2007) Directed evolution for increased chitinase activity. Appl Microbiol Biotechnol 76:135–139
Hiraishi T, Taguchi S (2009) Enzyme-catalyzed synthesis and degradation of biopolymers. Mini-Rev Org Chem 6:44–54
Hiraishi T, Ohura T, Ito S, Kasuya K, Doi Y (2000) Function of the catalytic domain of poly(3-hydroxybutyrate) depolymerase from Pseudomonas stutzeri. Biomacromolecules 1:320–324
Hiraishi T, Hirahara Y, Doi Y, Maeda M, Taguchi S (2006) Effects of mutations in the substrate-binding domain of poly[(R)-3-hydroxybutyrate] (PHB) depolymerase from Ralstonia pickettii T1 on PHB degradation. Appl Environ Microbiol 72:7331–7338
Hiraishi T, Komiya N, Maeda M (2010a) Y443F mutation in the substrate-binding domain of extracellular PHB depolymerase enhances its PHB adsorption and disruption abilities. Polym Degrad Stabil 95:1370–1374
Hiraishi T, Komiya N, Matsumoto N, Abe H, Fujita M, Maeda M (2010b) Degradation and adsorption characteristics of PHB depolymerase as revealed by kinetics of mutant enzymes with amino acid substitution in substrate-binding domain. Biomacromolecules 11:113–119
Hiraishi T, Yamashita K, Sakono M, Nakanishi J, Tan LT, Sudesh K, Abe H, Maeda M (2012) Display of functionally active PHB depolymerase on Escherichia coli cell surface. Macromol Biosci 12:218–224
Hisano T, Kasuya K, Tezuka Y, Ishii N, Kobayashi T, Shiraki M, Oroudjev E, Hansma H, Iwata T, Doi Y, Saito T, Miki K (2006) The crystal structure of polyhydroxybutyrate depolymerase from Penicillium funiculosum provides insights into the recognition and degradation of biopolyesters. J Mol Biol 356:993–1004
Jendrossek D, Handrick R (2002) Microbial degradation of polyhydroxyalkanoates. Annu Rev Microbiol 56:403–432
Jendrossek D, Schirmer A, Schlegel HG (1996) Biodegradation of polyhydroxyalkanoic acids. Appl Microbiol Biotechnol 46:451–463
Johannes TW, Zhao H (2006) Directed evolution of enzymes and biosynthetic pathways. Curr Opin Microbiol 9:261–267
Kiefer F, Arnold K, Künzli M, Bordoli L, Schwede T (2009) The SWISS-MODEL repository and associated resources. Nucl Acids Res 37:387–392
Kumar A, Gross RA, Jendrossek D (2000) Poly(3-hydroxybutyrate) depolymerase from Pseudomonas lemoignei: catalysis of esterification in organic media. J Org Chem 65:7800–7806
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lin L, Meng X, Liu P, Hong Y, Wu G, Huang X, Li C, Dong J, Xiao L, Liu Z (2009) Improved catalytic efficiency of endo-β-1,4-glucanase from Bacillus subtilis BME-15 by directed evolution. Appl Microbiol Biotechnol 82:671–679
Mukai K, Yamada K, Doi Y (1993) Kinetics and mechanism of heterogeneous hydrolysis of poly[(R)-3-hydroxybutyrate] film by PHA depolymerases. Int J Biol Macromol 15:361–366
Olsen M, Iverson B, Georgiou G (2000) High-throughput screening of enzyme libraries. Curr Opin Biotechnol 11:331–337
Otten LG, Quax WJ (2005) Directed evolution: selecting today’s biocatalysts. Biomol Eng 22:1–9
Papageorgiou AC, Hermawan S, Singh CB, Jendrossek D (2008) Structural basis of poly(3-hydroxybutyrate) hydrolysis by PhaZ7 depolymerase from Paucimonas lemoignei. J Mol Biol 382:1184–1194
Peitsch MC (1995) Protein modeling by e-mail bio/technology. Nat Biotechnol 13:658–660
Samuelson P, Gunneriusson E, Nygren PA, Stahl S (2002) Display of proteins on bacteria. J Biotechnol 96:129–154
Scandola M, Focarete ML, Frisoni G (1998) Simple kinetic model for the heterogeneous enzymatic hydrolysis of natural poly(3-hydroxybutyrate). Macromolecules 31:3846–3851
Suzuki Y, Ohura T, Kasuya K, Toshima K, Doi Y, Matsumura S (2000) Ring-opening polymerization of β-butyrolactone using PHB depolymerase. Chem Lett 4:318–319
Suzuki Y, Taguchi S, Saito T, Toshima K, Matsumura S, Doi Y (2001) Involvement of catalytic amino acid residues in enzyme-catalyzed polymerization for the synthesis of polyesters. Biomacromolecules 2:541–544
Tokiwa Y, Jarerat A (2004) Biodegradation of poly(l-lactide). Biotechnol Lett 26:771–777
Verlinden RAJ, Hill DJ, Kenward MA, Williams CD, Radecka IJ (2007) Bacterial synthesis of biodegradable polyhydroxyalkanoates. Appl Microbiol 102:1437–1449
Zhao H, Zha W (2006) In vitro ‘sexual’ evolution through the PCR-based staggered extension process (StEP). Nat Protoc 1:1865–1871
Zhao H, Giver L, Shao Z, Affholter JA, Arnold FH (1998) Molecular evolution by staggered extension process (StEP) in vitro recombination. Nat Biotechnol 16:258–261
Zhao H, Chockalingam K, Chen Z (2002) Directed evolution of enzymes and pathways for industrial biocatalysis. Curr Opin Biotechnol 13:104–110
Acknowledgements
We are grateful to the NBRP (NIG, Japan): E. coli for providing pVUB3 vector used in this work. Special thanks go to the RIKEN BSI Research Resource Centre for the DNA sequencing services. We are also greatly indebted to Dr. Hisano for his invaluable advice on the modeled structure of PhaZRpiT1. This manuscript has been submitted for an English language review. This research was supported by grants for Ecomolecular Science Research and Biomass Engineering Program from RIKEN Institute. L.T. Tan is grateful to RIKEN IPA and USM Fellowship for the financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 344 kb)
Rights and permissions
About this article
Cite this article
Tan, LT., Hiraishi, T., Sudesh, K. et al. Directed evolution of poly[(R)-3-hydroxybutyrate] depolymerase using cell surface display system: functional importance of asparagine at position 285. Appl Microbiol Biotechnol 97, 4859–4871 (2013). https://doi.org/10.1007/s00253-012-4366-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4366-8