Abstract
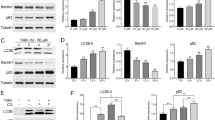
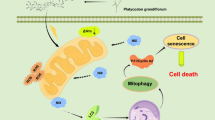
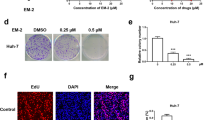
Hepatocellular carcinoma (HCC) is one of the most fatal malignancies with high mortality worldwide. Here the underlying antitumor mechanism of gallotannin was elucidated in HCC cells. Gallotannin suppressed viability and colony formation, increased subG1 portion and also induced senescence via upregulation of p21, G0/G1 arrest and higher SA-β-gal activity in HepG2 and SK-Hep1 cells. However, pan-caspase inhibitor Z-VAD-FMK reversed the ability of gallotannin to activate caspase 3 at 48 h after treatment in two HCC cells. Of note, gallotannin also induced autophagic features by increasing LC3 punctae, LC3B-II conversion, autophagic vacuoles and decreasing the expression of Beclin1 in two HCC cells. Furthermore, autophagy flux assay using GFP–mRFP–LC3 plasmid revealed increased yellowish color and late autophagy inhibitor CQ or NH4Cl enhanced cytotoxicity, LC3B-II conversion, and LC3 punctae in gallotannin-treated HepG2 and SK-Hep1 cells compared to early autophagy inhibitor 3-MA or wortmannin. Interestingly, gallotannin attenuated the expression of SIRT1 and mTOR and activated phosphorylation of AMPK in two HCC cells. Furthermore, AMPK activator AICAR significantly enhanced SA-β-gal activity and antiproliferation induced by gallotannin, while AMPK inhibitor compound C did not in two HCC cells. Consistently, LC3B-II conversion by gallotannin was not shown in AMPKα1 −/− MEF cells compared to WT AMPK +/+ MEF cells. Consistently, gallotannin reduced in vivo growth of HepG2 cells implanted in NCr nude mice along with decreased expression of PCNA and SIRT1 and increased AMPKα1 and TUNEL. Overall, these findings highlight evidence that regulation of SIRT1/AMPK is critically involved in gallotannin-induced senescence and impaired autophagy leading to cell death in HCC cells.






Similar content being viewed by others
Change history
16 July 2018
This article contains an error.
Abbreviations
- p62/SQSTM1:
-
p62 sequestosome 1
- Gal:
-
Gallotannin
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- HCC:
-
Human hepatocellular carcinoma
- HepG2:
-
Human epithelial hepatocellular carcinoma
- SK-Hep1:
-
Human epithelial hepatic adenocarcinoma
- MEF:
-
Mouse embryo fibroblast
- CQ:
-
Chloroquine
- NH4Cl:
-
Ammoniun chloride
- AICAR:
-
5-Aminoimidazole-4-carboxamide ribonucleotide
- Com. C:
-
Compound C
- LC3:
-
Microtubule-associated protein 1 light chain 3
- LC3-I:
-
Soluble unlipidated form of LC3
- LC3-II:
-
LC3–phospholipid conjugate
- AMPK:
-
AMP-activated protein kinase
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- FITC:
-
Fluorescein isothiocyanate
- GFP/RFP:
-
Green fluorescent protein/red fluorescent protein
- SIRT1:
-
NAD-dependent deacetylase sirtuin-1
- Caspase:
-
Cysteine aspartyl-specific protease
- 3-MA:
-
3-Methyladenine
- SA-β-gal:
-
Senescence-associated β-galactosidase
- TEM:
-
Transmission electron microscopy
- TUNEL:
-
Terminal deoxynucleotidyl transferase dUTP nick end labeling
References
Al-Ayyoubi S, Gali-Muhtasib H (2007) Differential apoptosis by gallotannin in human colon cancer cells with distinct p53 status. Mol Carcinog 46(3):176–186. doi:10.1002/mc.20252
Alcorta DA, Xiong Y, Phelps D, Hannon G, Beach D, Barrett JC (1996) Involvement of the cyclin-dependent kinase inhibitor p16 (INK4a) in replicative senescence of normal human fibroblasts. Proc Natl Acad Sci USA 93(24):13742–13747
Al-Halabi R, Abou Merhi R, Chakilam S et al (2015) Gallotannin is a DNA damaging compound that induces senescence independently of p53 and p21 in human colon cancer cells. Mol Carcinog 54(10):1037–1050. doi:10.1002/mc.22172
Althubiti M, Lezina L, Carrera S et al (2014) Characterization of novel markers of senescence and their prognostic potential in cancer. Cell Death Dis 5:e1528. doi:10.1038/cddis.2014.489
Asfour OM, el-Fiky HM, Nassar A (1990) Lamellar keratoplasty combined with extracapsular cataract extraction. Refract Corneal Surg 6(2):125–126
Bae HJ, Chang YG, Noh JH et al (2012) DBC1 does not function as a negative regulator of SIRT1 in liver cancer. Oncol Lett 4(5):873–877. doi:10.3892/ol.2012.875
Bae HJ, Noh JH, Kim JK et al (2014) MicroRNA-29c functions as a tumor suppressor by direct targeting oncogenic SIRT1 in hepatocellular carcinoma. Oncogene 33(20):2557–2567. doi:10.1038/onc.2013.216
Baffy G, Brunt EM, Caldwell SH (2012) Hepatocellular carcinoma in non-alcoholic fatty liver disease: an emerging menace. J Hepatol 56(6):1384–1391. doi:10.1016/j.jhep.2011.10.027
Baker DJ, Weaver RL, van Deursen JM (2013) p21 both attenuates and drives senescence and aging in BubR1 progeroid mice. Cell Rep 3(4):1164–1174. doi:10.1016/j.celrep.2013.03.028
Barth S, Glick D, Macleod KF (2010) Autophagy: assays and artifacts. J Pathol 221(2):117–124. doi:10.1002/path.2694
Berk V, Kaplan MA, Tonyali O et al (2013) Efficiency and side effects of sorafenib therapy for advanced hepatocellular carcinoma: a retrospective study by the anatolian society of medical oncology. Asian Pacific J Cancer Prev APJCP 14(12):7367–7369
Campisi J (2000) Cancer, aging and cellular senescence. In vivo 14(1):183–188 (Athens, Greece)
Canto C, Jiang LQ, Deshmukh AS et al (2010) Interdependence of AMPK and SIRT1 for metabolic adaptation to fasting and exercise in skeletal muscle. Cell Metab 11(3):213–219. doi:10.1016/j.cmet.2010.02.006
Casella ML, Parody JP, Ceballos MP et al (2014) Quercetin prevents liver carcinogenesis by inducing cell cycle arrest, decreasing cell proliferation and enhancing apoptosis. Mol Nutr Food Res 58(2):289–300. doi:10.1002/mnfr.201300362
Chi X, Zhang R, Shen N et al (2015) Sulforaphane reduces apoptosis and oncosis along with protecting liver injury-induced ischemic reperfusion by activating the Nrf2/ARE pathway. Hep Intl 9(2):321–329. doi:10.1007/s12072-014-9604-y
Dai W, Wang F, He L et al (2015) Genistein inhibits hepatocellular carcinoma cell migration by reversing the epithelial-mesenchymal transition: partial mediation by the transcription factor NFAT1. Mol Carcinog 54(4):301–311. doi:10.1002/mc.22100
Ding S, Hu A, Hu Y, Ma J, Weng P, Dai J (2014) Anti-hepatoma cells function of luteolin through inducing apoptosis and cell cycle arrest. Tumour Biol J Int Soc Oncodev Biol Med 35(4):3053–3060. doi:10.1007/s13277-013-1396-5
Dong Y, Yin S, Jiang C et al (2014) Involvement of autophagy induction in penta-1,2,3,4,6-O-galloyl-beta-d-glucose-induced senescence-like growth arrest in human cancer cells. Autophagy 10(2):296–310. doi:10.4161/auto.27210
Dupont N, Orhon I, Bauvy C, Codogno P (2014) Autophagy and autophagic flux in tumor cells. Methods Enzymol 543:73–88. doi:10.1016/B978-0-12-801329-8.00004-0
Feng Y, Ke C, Tang Q et al (2014) Metformin promotes autophagy and apoptosis in esophageal squamous cell carcinoma by downregulating Stat3 signaling. Cell Death Dis 5:e1088. doi:10.1038/cddis.2014.59
Flores A, Marrero JA (2014) Emerging trends in hepatocellular carcinoma: focus on diagnosis and therapeutics. Clin Med Insights Oncol 8:71–76. doi:10.4137/CMO.S9926
Gali-Muhtasib H, Hmadi R, Kareh M, Tohme R, Darwiche N (2015) Cell death mechanisms of plant-derived anticancer drugs: beyond apoptosis. Apoptosis 20(12):1531–1562. doi:10.1007/s10495-015-1169-2
Gewirtz DA (2013) Autophagy and senescence: a partnership in search of definition. Autophagy 9(5):808–812. doi:10.4161/auto.23922
Gewirtz DA (2014) Autophagy and senescence in cancer therapy. J Cell Physiol 229(1):6–9. doi:10.1002/jcp.24420
Ghosh S, Zhou Z (2015) SIRTain regulators of premature senescence and accelerated aging. Protein Cell 6(5):322–333. doi:10.1007/s13238-015-0149-1
Guo L, Xie B, Mao Z (2012) Autophagy in premature senescent cells is activated via AMPK pathway. Int J Mol Sci 13(3):3563–3582. doi:10.3390/ijms13033563
Hou X, Xu S, Maitland-Toolan KA et al (2008) SIRT1 regulates hepatocyte lipid metabolism through activating AMP-activated protein kinase. J Biol Chem 283(29):20015–20026. doi:10.1074/jbc.M802187200
Huh JE, Lee EO, Kim MS et al (2005) Penta-O-galloyl-beta-d-glucose suppresses tumor growth via inhibition of angiogenesis and stimulation of apoptosis: roles of cyclooxygenase-2 and mitogen-activated protein kinase pathways. Carcinogenesis 26(8):1436–1445. doi:10.1093/carcin/bgi097
Jung DB, Yun M, Kim EO et al (2015) The heparan sulfate mimetic PG545 interferes with Wnt/beta-catenin signaling and significantly suppresses pancreatic tumorigenesis alone and in combination with gemcitabine. Oncotarget 6(7):4992–5004. doi:10.18632/oncotarget.3214
Kant R, Yen CH, Lu CK, Lin YC, Li JH, Chen YM (2016) Identification of 1,2,3,4,6-penta-O-galloyl-β-d-glucopyranoside as a glycine N-methyltransferase enhancer by high-throughput screening of natural products inhibits hepatocellular carcinoma. Int J Mol Sci 17(5):669. doi:10.3390/ijms17050669
Kim J, Yun M, Kim EO et al (2016) Decursin enhances TRAIL-induced apoptosis through oxidative stress mediated- endoplasmic reticulum stress signalling in non-small cell lung cancers. Br J Pharmacol 173(6):1033–1044. doi:10.1111/bph.13408
Kiss AK, Filipek A, Zyzynska-Granica B, Naruszewicz M (2013) Effects of penta-O-galloyl-beta-d-glucose on human neutrophil function: significant down-regulation of l-selectin expression. Phytother Res PTR 27(7):986–992. doi:10.1002/ptr.4822
Klionsky DJ, Abdalla FC, Abeliovich H et al (2012) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 8(4):445–544
Klose J, Stankov MV, Kleine M et al (2014) Inhibition of autophagic flux by salinomycin results in anti-cancer effect in hepatocellular carcinoma cells. PLoS One 9(5):e95970. doi:10.1371/journal.pone.0095970
Kwon TR, Jeong SJ, Lee HJ et al (2012) Reactive oxygen species-mediated activation of JNK and down-regulation of DAXX are critically involved in penta-O-galloyl-beta-d-glucose-induced apoptosis in chronic myeloid leukemia K562 cells. Biochem Biophys Res Commun 424(3):530–537. doi:10.1016/j.bbrc.2012.06.150
Lao Y, Wan G, Liu Z et al (2014) The natural compound oblongifolin C inhibits autophagic flux and enhances antitumor efficacy of nutrient deprivation. Autophagy 10(5):736–749. doi:10.4161/auto.28034
Lau AW, Liu P, Inuzuka H, Gao D (2014) SIRT1 phosphorylation by AMP-activated protein kinase regulates p53 acetylation. Am J Cancer Res 4(3):245–255
Lee HJ, Seo NJ, Jeong SJ et al (2011) Oral administration of penta-O-galloyl-beta-d-glucose suppresses triple-negative breast cancer xenograft growth and metastasis in strong association with JAK1-STAT3 inhibition. Carcinogenesis 32(6):804–811. doi:10.1093/carcin/bgr015
Lee CW, Wong LL, Tse EY et al (2012) AMPK promotes p53 acetylation via phosphorylation and inactivation of SIRT1 in liver cancer cells. Cancer Res 72(17):4394–4404. doi:10.1158/0008-5472.CAN-12-0429
Marino G, Niso-Santano M, Baehrecke EH, Kroemer G (2014) Self-consumption: the interplay of autophagy and apoptosis. Nat Rev Mol Cell Biol 15(2):81–94. doi:10.1038/nrm3735
Marquardt JU, Gomez-Quiroz L, Arreguin Camacho LO et al (2015) Curcumin effectively inhibits oncogenic NF-kappaB signaling and restrains stemness features in liver cancer. J Hepatol 63(3):661–669. doi:10.1016/j.jhep.2015.04.018
Mizushima N, Yoshimori T, Levine B (2010) Methods in mammalian autophagy research. Cell 140(3):313–326. doi:10.1016/j.cell.2010.01.028
Oh GS, Pae HO, Oh H et al (2001) In vitro anti-proliferative effect of 1,2,3,4,6-penta-O-galloyl-beta-d-glucose on human hepatocellular carcinoma cell line, SK-HEP-1 cells. Cancer Lett 174(1):17–24
Park KY, Lee HJ, Jeong SJ et al (2010) 1,2,3,4,6-Penta-O-galloyl-beta-d-glucose suppresses hypoxia-induced accumulation of hypoxia-inducible factor-1alpha and signaling in LNCaP prostate cancer cells. Biol Pharm Bull 33(11):1835–1840
Park IJ, Yang WK, Nam SH et al (2014) Cryptotanshinone induces G1 cell cycle arrest and autophagic cell death by activating the AMP-activated protein kinase signal pathway in HepG2 hepatoma. Apoptosis Int J Program cell Death 19(4):615–628. doi:10.1007/s10495-013-0929-0
Park E, Kwon HY, Jung JH et al (2015) Inhibition of myeloid cell leukemia 1 and activation of caspases are critically involved in gallotannin-induced apoptosis in prostate cancer cells. Phytother Res 29(8):1225–1236. doi:10.1002/ptr.5371
Pietrocola F, Izzo V, Niso-Santano M et al (2013) Regulation of autophagy by stress-responsive transcription factors. Semin Cancer Biol 23(5):310–322. doi:10.1016/j.semcancer.2013.05.008
Puissant A, Robert G, Fenouille N et al (2010) Resveratrol promotes autophagic cell death in chronic myelogenous leukemia cells via JNK-mediated p62/SQSTM1 expression and AMPK activation. Cancer Res 70(3):1042–1052. doi:10.1158/0008-5472.CAN-09-3537
Raghuram GV, Mishra PK (2014) Stress induced premature senescence: a new culprit in ovarian tumorigenesis? Indian J Med Res 140(Suppl):S120–S129
Romanov VS, Abramova MV, Svetlikova SB et al (2010) p21(Waf1) is required for cellular senescence but not for cell cycle arrest induced by the HDAC inhibitor sodium butyrate (Georgetown, Tex). Cell Cycle 9(19):3945–3955. doi:10.4161/cc.9.19.13160
Sabin RJ, Anderson RM (2011) Cellular senescence—its role in cancer and the response to ionizing radiation. Genome Integr 2(1):7. doi:10.1186/2041-9414-2-7
Shaikh QU, Yang M, Memon KH et al (2016) 1,2,3,4,6-Pentakis[-O-(3,4,5-trihydroxybenzoyl)]-alpha, beta-d-glucopyranose (PGG) analogs: design, synthesis, anti-tumor and anti-oxidant activities. Carbohydr Res 430:72–81. doi:10.1016/j.carres.2016.04.021
Sharma K, Goehe R, Beckta JM, Valerie K, Gewirtz DA (2014) Autophagy and radiosensitization in cancer. EXCLI J 13:178–191
Singh K, Sharma A, Mir MC et al (2014) Autophagic flux determines cell death and survival in response to Apo2L/TRAIL (dulanermin). Mol Cancer 13:70. doi:10.1186/1476-4598-13-70
Song X, Yin S, Huo Y et al (2015) Glycycoumarin ameliorates alcohol-induced hepatotoxicity via activation of Nrf2 and autophagy. Free Radic Biol Med 89:135–146. doi:10.1016/j.freeradbiomed.2015.07.006
Tang Q, Li G, Wei X et al (2013) Resveratrol-induced apoptosis is enhanced by inhibition of autophagy in esophageal squamous cell carcinoma. Cancer Lett 336(2):325–337. doi:10.1016/j.canlet.2013.03.023
Vucicevic L, Misirkic M, Janjetovic K et al (2011) Compound C induces protective autophagy in cancer cells through AMPK inhibition-independent blockade of Akt/mTOR pathway. Autophagy 7(1):40–50
Wang Y, Liang Y, Vanhoutte PM (2011) SIRT1 and AMPK in regulating mammalian senescence: a critical review and a working model. FEBS Lett 585(7):986–994. doi:10.1016/j.febslet.2010.11.047
Wang L, Gao C, Yao S, Xie B (2013) Blocking autophagic flux enhances matrine-induced apoptosis in human hepatoma cells. Int J Mol Sci 14(12):23212–23230. doi:10.3390/ijms141223212
Wei L, Huang N, Yang L et al (2009) [Sorafenib reverses multidrug resistance of hepatoma cells in vitro]. Nan fang yi ke da xue xue bao = J South Med Univ 29(5):1016–1019 (1023)
Wu Y, Li X, Zhu JX et al (2011) Resveratrol-activated AMPK/SIRT1/autophagy in cellular models of Parkinson’s disease. Neurosignals 19(3):163–174. doi:10.1159/000328516
Yang D, Li L, Liu H et al (2013) Induction of autophagy and senescence by knockdown of ROC1 E3 ubiquitin ligase to suppress the growth of liver cancer cells. Cell Death Differ 20(2):235–247. doi:10.1038/cdd.2012.113
Yip NK, Ho WS (2013) Berberine induces apoptosis via the mitochondrial pathway in liver cancer cells. Oncol Rep 30(3):1107–1112. doi:10.3892/or.2013.2543
Zhang J, Li L, Kim SH, Hagerman AE, Lu J (2009) Anti-cancer, anti-diabetic and other pharmacologic and biological activities of penta-galloyl-glucose. Pharm Res 26(9):2066–2080. doi:10.1007/s11095-009-9932-0
Zhang J, Nkhata K, Shaik AA et al (2011) Mouse prostate proteome changes induced by oral pentagalloylglucose treatment suggest targets for cancer chemoprevention. Curr Cancer Drug Targets 11(7):787–798
Zhang XJ, Chen S, Huang KX, Le WD (2013) Why should autophagic flux be assessed? Acta Pharmacol Sin 34(5):595–599. doi:10.1038/aps.2012.184
Zhao H, Darzynkiewicz Z (2014) Attenuation of replication stress-induced premature cellular senescence to assess anti-aging modalities. Curr Protoc Cytom 69:9.47.1–9.47.10. doi:10.1002/0471142956.cy0947s69
Zhao C, Yin S, Dong Y et al (2013) Autophagy-dependent EIF2AK3 activation compromises ursolic acid-induced apoptosis through upregulation of MCL1 in MCF-7 human breast cancer cells. Autophagy 9(2):196–207. doi:10.4161/auto.22805
Zhao T, Sun Q, del Rincon SV, Lovato A, Marques M, Witcher M (2014) Gallotannin imposes S phase arrest in breast cancer cells and suppresses the growth of triple-negative tumors in vivo. PLoS One 9(3):e92853. doi:10.1371/journal.pone.0092853
Zhou J, Hu SE, Tan SH et al (2012) Andrographolide sensitizes cisplatin-induced apoptosis via suppression of autophagosome-lysosome fusion in human cancer cells. Autophagy 8(3):338–349. doi:10.4161/auto.18721
Zhou ZW, Li XX, He ZX et al (2015) Induction of apoptosis and autophagy via sirtuin1- and PI3K/Akt/mTOR-mediated pathways by plumbagin in human prostate cancer cells. Drug Des Devel Ther 9:1511–1554. doi:10.2147/DDDT.S75976
Zu Y, Liu L, Lee MY et al (2010) SIRT1 promotes proliferation and prevents senescence through targeting LKB1 in primary porcine aortic endothelial cells. Circ Res 106(8):1384–1393. doi:10.1161/CIRCRESAHA.109.215483
Acknowledgements
This work was supported by the Korea Science and Engineering Foundation (KOSEF) grant funded by the Korea government (MEST) (2014R1A2A10052872).
Author information
Authors and Affiliations
Contributions
HY Kwon designed this project, performed most of the experiments, and drafted the manuscript; JH Kim contributed to animal study; SK. Srivastava and BL Kim advised us of the experiment design and helped proofreading; SH Kim supervised all experiments and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
204_2017_2021_MOESM1_ESM.jpg
Supplementary Figure 1. Gallotannin induced weak apoptosis in HCC cells in a time and concentration dependent fashion. a Cell cycle analysis of gallotannin treated HepG2 and SK-Hep1 cells. Cells were treated with gallotannin indicated concentrations and for different times. Then the cell cycle analysis was performed using FACs Calibur, and the cell cycle was analyzed using CellQuest Software. b SubG1 portion was increased dependent on time and concentration in gallotannin treated HepG2 and SK-Hep1 cells. The results of cell cycle analysis were represented as relative percentage ± SD. SubG1 portion of each treatment was represented (means ± SD of three independent experiments, **p < 0.01 and *** p < 0.001 vs. control by One-Way ANOVA). (JPEG 3334 kb)
Rights and permissions
About this article
Cite this article
Kwon, H.Y., Kim, JH., Kim, B. et al. Regulation of SIRT1/AMPK axis is critically involved in gallotannin-induced senescence and impaired autophagy leading to cell death in hepatocellular carcinoma cells. Arch Toxicol 92, 241–257 (2018). https://doi.org/10.1007/s00204-017-2021-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-017-2021-y