Abstract
The emergence of immune checkpoint inhibitors (ICIs), mainly including anti-programmed cell death protein 1/programmed cell death ligand 1 (PD-1/PD-L1) and anti-cytotoxic T lymphocyte-associated antigen-4 (CTLA-4) monoclonal antibodies (mAbs), has shaped therapeutic landscape of some type of cancers. Despite some ICIs have manifested compelling clinical effectiveness in certain tumor types, the majority of patients still showed de novo or adaptive resistance. At present, the overall efficiency of immune checkpoint therapy remains unsatisfactory. Exploring additional immune checkpoint molecules is a hot research topic. Recent studies have identified several new immune checkpoint targets, like lymphocyte activation gene-3 (LAG-3), T cell immunoglobulin and mucin-domain containing-3 (TIM-3), T cell immunoglobulin and ITIM domain (TIGIT), V-domain Ig suppressor of T cell activation (VISTA), and so on. The investigations about these molecules have generated promising results in preclinical studies and/or clinical trials. In this review, we discussed the structure and expression of these newly-characterized immune checkpoints molecules, presented the current progress and understanding of them. Moreover, we summarized the clinical data pertinent to these recent immune checkpoint molecules as well as their application prospects.
Similar content being viewed by others
Background
The past decade has witnessed the rapid development of immunotherapy. Now, it has been recognized as a key strategy to control the progression of malignant tumors. Among these immunotherapies, immune checkpoint inhibitors (ICIs) [1], chimeric antigen receptor T cell [2] and bispecific antibodies [3] are the most promising immunotherapy strategies. Encouragingly, the 2018 Nobel Prize in Physiology and Medicine was awarded to Drs. James Allison and Tasuku Honjo, who discovered programmed cell death protein-1 (PD-1) and cytotoxic T lymphocyte-associated antigen-4 (CTLA-4), to honor their outstanding work for the development of immunotherapy [4]. In addition, Dr. Lieping Chen did seminal contributions to the discovery of PD-L1 (CD274, B7-H1) [5]. Now, these immune checkpoint targets have realized the transformation from the laboratory to the clinical application.
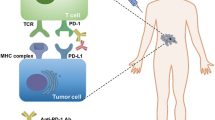
CTLA-4 is a cell-surface receptor related to CD28, binding to the ligands CD80 (B7–1) and CD86 (B7–2) [6]. The binding of CTLA-4 to CD80/CD86 delivers a negative signal to T cells activation by making CD80/CD86 less available to CD28 [7]. In the early 1996, Leach and his colleagues found that injecting anti-CTLA-4 antibody in mice with pre-established tumors significantly reduced tumor growth [8]. Ipilimumab, as the first anti-CTLA-4 monoclonal antibody (mAb) reaching the clinic [9], garnered the approval for the treatment of patients with malignant melanoma in 2011 [10]. PD-1, also known as CD279, belongs to CD28 superfamily [11]. The binding of PD-1 to its ligands PD-L1 and PD-L2 (CD273, B7-DC) serves as a “rheostat” of immunological regulation, suppressing the activation and function of T cells to downregulate the immune response [12, 13]. Pembrolizumab, the first humanized mAb against PD-1, gained its first global approval for patients with unresectable or metastatic melanoma by United States Food and Drug Administration (FDA) in 2014 [14]. Subsequently, its indication was expended to head and neck squamous cell carcinoma (HNSCC) [15], non-small cell lung cancer (NSCLC) [16], metastatic urothelial carcinoma [17], cervical carcinoma [18] among others in a list that continues to grow. By the end of 2018, as many as 7 types of ICIs have been approved by FDA for the treatment of cancers and all of them were immune checkpoint blockers against PD-1/PD-L1 or CTLA-4 [19].
Nevertheless, the response rate of anti-PD-1/PD-L1 mAb and anti-CTLA-4 mAb in overall patients is far from satisfactory. Most patients show primary or acquired resistance to these ICIs [9]. Thus, intensive researches aimed at finding novel immune checkpoint targets have been ongoing. The next generation immune checkpoints such as lymphocyte activation gene-3 (LAG-3) [20], T cell immunoglobulin and mucin-domain containing-3 (TIM-3) [21], T cell immunoglobulin and ITIM domain (TIGIT) [22], V-domain Ig suppressor of T cell activation (VISTA) [23], B7 homolog 3 protein (B7-H3) [24] and B and T cell lymphocyte attenuator (BTLA) [25] demonstrate as promising therapeutic targets with the chance to realize clinical application. In this review, we will emphasize these newly-characterized immune checkpoint molecules and their clinical studies that suggest the promising future for the clinical application.
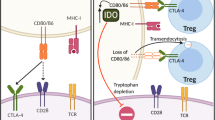
Immune checkpoints
The full activation of T cell dependents on two different signals, signal one is derived from the interaction between antigenic peptide/major histocompatibility complex (MHC) on the surface of APCs and the T cell receptor (TCR), and signal two requires an antigen-independent co-signaling molecules [26]. Note worthily, T cell activation is tightly regulated by co-stimulators or co-inhibitors known as immune checkpoints [7]. If antigen/MHC and TCR binding is accompanied by the engagement of costimulatory receptors, such as CD28, it allows T cell to proliferate and to migrate toward specific antigen. On the contrary, if antigen/MHC and TCR binding is accompanied by the engagement of coinhibitory receptors, such as CTLA-4, it will suppress T cell activation [27, 28]. CTLA-4 is not detectable in naïve T cell but is rapidly induced upon T cell activation and it primarily regulates the amplitude of T cell during the early priming phase in lymphoid organs [29, 30]. The binding of CTLA-4 to B7 proteins competes CD28 costimulatory signals and eventually acts to impede excessive immunity [31]. The aim of this co-inhibitor is to minimize damage to normal tissues and prevent unwanted autoimmunity [31, 32]. In contrast to CTLA-4, PD-1 plays a major role in the maintenance of peripheral tolerance [33]. The engagement of PD-1 by its ligands results in the recruitment of Src homology 2 (SH2) domain containing phosphatases 1/2 (SHP1/2) and then inhibits T cell proliferation and cytokine secretion mediated by TCR [34]. Some cancer cells possess the ability to generate inhibitory ligands which can bind co-inhibitory receptor molecules. This engagement limits normal anti-tumor immune responses thus assisting in immune escape [35]. Therefore, the blockades of these immune checkpoints are capable to invoke patient’s own anti-tumor immune response [32]. Immune checkpoint therapies do not kill cancer cells directly, instead they harness the power of the host’s immune system to re-enhance endogenous anti-tumor activity [36].
Newly emerging immune checkpoints
Apart from CTLA-4 and PD-1, novel immune checkpoint molecules on T cells have been discovered continuously. So far, all these emerging immune checkpoints targets are either in the clinical trial or under active development. Those delineated below are the most promising immune checkpoint targets for which blocking antibodies are available in clinical trials (Table 1). A substantial body of evidence accumulates to indicate the synergistic effect of combinatorial blockade among these new immune checkpoints and anti-PD-1/L1 and/or anti-CTLA-4 mAbs. Huang and his colleague utilized a murine model to explore the effect of combinatorial blockade of LAG-3 and PD-1 pathways in ovarian cancer [37]. Their results showed that dual blockade of LAG-3 and PD-1 synergistically enhanced anti-tumor immunity and suppressed tumor growth by enhancing CD8+ tumor infiltrating T cells (TILs) and decreasing regulatory T cells (Tregs) in the tumor microenvironment (TME) [37]. The same group further detected the level of other inhibitory receptors when PD-1 or LAG-3 was blocked. When the mice were treated with anti-PD-1 mAb, the level of LAG-3 and CTLA-4 were increased. Interesting, treatment with anti-LAG-3 mAb upregulated the level of PD-1 [38]. Their experiments indicated that the blockade of a single immune checkpoint targets may lead to compensatory upregulation of other checkpoint receptors in TME. The similar compensatory mechanism between TIM-3 and PD-1 was observed in lung cancer [39] and melanoma [40]. It seems that the compensatory mechanism is common across different types of cancer. These preclinical results pave the way for the combinatorial blockade strategies in clinical trials.
Lymphocyte activation Gene-3 (LAG-3)
The biology of LAG-3
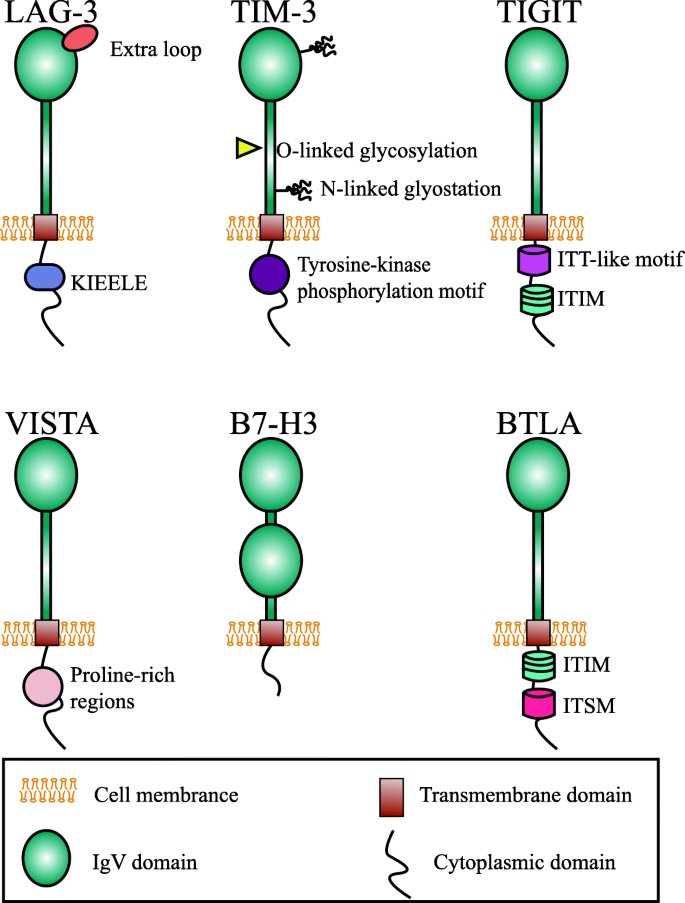
LAG-3 (CD223) was discovered by Triebel and colleagues as early as 1990 [41]. The LAG-3 gene encompass 8 exons and the corresponding cDNA can encode a 498-amino acid type I membrane protein [41]. LAG-3 gene is located adjacent to CD4 gene on chromosome 12, and further analysis of amino acid sequence reveals an approximately 20% identical to CD4 (Table 2) [41]. Mature LAG-3 protein includes four parts, hydrophobic leader, extracellular region, transmembrane region, and cytoplasmic region (Fig.1). The extracellular region is consisted of four immunoglobulin (Ig) superfamily-like domains (D1-D4) [42]. The membrane-distal D1 domain contains a unique short amino acid sequence, the so-called “extra loop” [43]. The cytoplasmic domain of LAG-3 has three conserved regions: a serine-phosphorylation site, a KIEELE motif, and a glutamic acid-proline repeats, of which the KIEELE motif is essential for LAG-3 to exert inhibitory function [44]. Metalloproteases can cleave LAG-3 within the connecting peptide between the D4 transmembrane domain and the transmembrane domain, generating a soluble LAG-3 (sLAG-3) [45]. Some researches demonstrated that sLAG-3 could limit the magnitude of the T cell immune responses [46]. LAG-3 is usually expressed on activated CD4+ and CD8+ T cells [41], Tregs [47], a subpopulation natural killer (NK) cells [48], B cells [49], plasmacytoid dendritic cells (pDCs) as well [50]. Ample of evidence have indicated that LAG-3 signaling play a negative regulatory role in T helper 1 (Th1) cell activation, proliferation and cytokine secretion [51,52,53]. During tumorigenesis and cancer progression, tumor cells exploit this pathway to escape from immune surveillance.
Based on the structural similarity between LAG-3 and CD4, MHC-II is reasonable considered as ligand for LAG-3. In fact, the binding affinity between LAG-3 and MHC-II is 100-fold higher than CD4 [20]. Now, MHC-II [54], galectin-3 [55], LSECtin [56], and a-synuclein [57] have been described to interact with LAG-3, with the MHC-II as a canonical ligand (Fig. 2). More recently, Wang and his co-workers elucidated that fibrinogen-like protein 1 (FGL1) was a novel high-affinity ligand for LAG-3 independent from MHC-II [58].
Current and emerging immune checkpoint receptors and their respective ligands. Various immune checkpoint molecules expressed on T cells were shown with their ligands. Immune checkpoints such as PD-1, CTLA-4, LAG-3, TIM-3, TIGIT bound with their respective ligands on APCs and/or tumor cells, triggering a negative or positive signal to T cells response
Clinical trials on LAG-3
Based on the experimental results that administration of recombinant sLAG-3 molecule with irradiated tumor cells can dampen the growth of established tumors, early clinical work centered on developing a sLAG-3-Ig [59]. IMP321 (Eftilagimod alpha) was initially developed as a vaccine immunological adjuvant [60]. It is a soluble recombinant protein by fusing the four extracellular Ig domains of LAG-3 to the Fc portion of human IgG1 [61]. The first-in-man phase I trial was conducted in patients with metastatic renal cell carcinoma (mRCC) (NCT00351949) [62]. The results showed that adverse events (AEs) related to IMP321 were minimal and IMP321 increased the subset of circulating activated CD8+ T cells which was correlated to tumor growth reduction. Although no objective response was observed in this trial, 7 of 8 patients experienced stable disease in higher IMP321 dose group (> 6 mg) while only 3 of 11 in the lower dose group [62]. IMP321 monotherapy showed a modest efficacy in cancer therapy, thus it was rational to combine the agent with other chemotherapeutic drugs. Subsequently, another two clinical trials were lunched in metastatic breast cancer (MBC) (NCT00349934) [63] and advanced pancreatic cancer (NCT00732082) [64]. Ultimately, the former phase I/II trial showed a 50% objective response rate (ORR) at the end of treatment point, which compared favorably to a historical response rate of approximately 25% [63]. Furthermore, the authors found an absolute and proportional increase in MHC class II-expressing APCs, NK cells and CD8+ T cell populations and these subsets were known to connect with antitumor activity [63]. This encouraging result has prompted a further phase IIb multicenter clinical trial that is currently recruiting patients with MBC (NCT02614833) [65]. Unfortunately, the later phase I clinical trial intended to assess the role of IMP321 and gemcitabine as a front-line therapy in patients with pancreatic cancer showed no meaningful objective response [64]. The role of IMP321 in combination with other immunotherapies (e.g., anti-PD-1 mAb, NCT03625323) or as an adjuvant for cancer vaccines are being explored (NCT00324623, NCT01308294) [66, 67].
Relatlimab, also named BMS-986016, is the first commercially mAb directed against LAG-3 [68]. Many preclinical mouse models have showed that PD-1/PD-L1 blockade upregulated LAG-3 or other immune checkpoints as a compensatory mechanism [37, 38, 69]. These data evoked the further exploration of combination therapy strategies. For example, the first phase I clinical trial about Relatlimab was opened in 2013 (NCT01968109) [38]. The aim of this clinical trial was to evaluate the efficacy of Relatlimab as a monotherapy or in combination with Nivolumab (anti-PD-1 antibody) in patients with various advanced malignancies including melanoma, NSCLC, and RCC [20]. At ESMO 2017, researcher announced the updated efficacy and safety results in a cohort of 68 melanoma patients who had received prior immunotherapy [70]. The ORR was 11.5% in 61 patients who were able to assess efficacy, including one patient achieved complete response and 6 were partial response (PR). Noticeably, the ORR was higher in patients with the expression of LAG-3 ≥ 1% and the AEs were tolerable [70]. As of July 15, 2019, at least 18 clinical trials on Relatlimab had been registered on the ClinicalTrials.gov. All of them were phase I or II but none of them are completed.
LAG525 is another humanized anti-LAG-3 mAb. It is a high-affinity IgG4 antibody which blocks the binding of MHC-II to LAG-3. Currently, LAG525 is undergoing a series of phase I or II testing in combination with anti-PD-1 antibody for patients with advanced cancers. For example, LAG525’s first clinical trial was launched in 2015 (NCT02460224). It was a phase I trial to determine the efficacy and safety of LAG525 plus PDR001 (anti-PD-1 mAb) in advanced malignancies [36]. Other LAG-3 inhibitory antibodies MK-4280, REGN3767 [71], TSR-033 [72], BI754111 [73], and Sym022 [74] have also been investigated at various stages of clinical development. To capitalize on synergistic effects of co-blockade PD-1 and LAG-3 pathways in preclinical models [75], some bispecific anti-LAG-3/PD-(L)1 antagonistic mAbs have also been developed, such as FS118 [76] and MGD013 [77]. To date, at least 10 kinds of LAG-3 blockade agents have been developed and studied in clinical trials, yet their results are not available now.
T cell immunoglobulin and mucin-domain containing-3 (TIM-3)
The biology of TIM-3
TIM-3, also called hepatitis A virus cellular receptor 2 (HAVCR2), presents several unique features making it another intriguing immune checkpoint [78]. It was first identified as a protein selectively expressed on CD4+ Th1 and CD8+ T cytotoxic 1 (Tc1) cells as early as 2002 [21]. But now it is commonly classified as immune checkpoint molecule similar to CTLA-4 and PD-1. The genomic analysis shows that the TIM gene family is composed of three genes, namely TIM-1, TIM-3, TIM-4, located on human chromosome 5q33.2 [79]. Human TIM-3 protein comprises of 302 amino acids, while mouse homolog includes 281 amino acids residues with 63% identity to human TIM-3 [21]. It belongs to Ig superfamily (IgSF) with an N-terminal Ig variable region (IgV)-like domain, a membrane-proximal mucin-like domain containing sites for O-linked glycosylation (glycosylated mucin domain), a single transmembrane region and a C-terminal cytoplasmic tail. There are also sites for N-linked glycosylation between the mucin and transmembrane [79]. The TIM-3 cytoplasmic tail does not have the classical inhibitory signaling motif, like immune receptor tyrosine based inhibitory motif (ITIM) or immune receptor tyrosine-based switch motif (ITSM), but contains five conserved tyrosine residues, two of which (Y265 and 272) can be phosphorylated by Src kinases or interleukin inducible T cell kinase and are crucial for downstream signaling [35, 80]. The expression of TIM-3 was not limited on T cell, it was known to express on different types of immune cells, including B cells, Tregs, NK cells, DCs, monocytes, and macrophages [81]. Lately, the expression of TIM-3 has been identified in leukemic stem cells and tumor-associated endothelium [82, 83].
Hitherto, four distinct ligands have been reported to bind to the IgV domain of TIM-3, including galectin-9, high-mobility group protein B1 (HMGB1), carcinoembryonic antigen cell adhesion molecule 1 (Ceacam-1), and phosphatidyl serine (PtdSer) [84]. It is noteworthy that galectin-9 and HMGB1are soluble ligands, while Ceacam-1 and PtdSer belong to surface ligands. The engagement of TIM-3 with galectin-9 triggered intracellular calcium flux of Th1 cells, inducing cell death [85]. Furthermore, a study by Kang et al. showed that galectin-9 also induced apoptosis of TIM-3+CD8+T cell in colon cancer [86]. The interaction between HMGB1 and TIM-3 mainly had an impact on innate immune response. In tumor, TIM-3 was highly expressed on tumor infiltrating DCs and can compete with nucleic acid binding to HMGB1, therefore dampening anti-tumor immunity mediated by nucleic acids [87]. Ceacam-1 was a molecule involved in T cell inhibition. Huang and his colleague elucidated that TIM-3 and Ceacam-1 can form a heterodimer in both cis and trans which acts as a negative regulator of T cell responses [88]. The interaction of PtdSer with TIM-3 has been showed to connect with the clearance of apoptotic bodies and also improve the antigen cross-presentation [89]. More importantly, higher expression of TIM-3 was associated with a poor prognosis in solid malignant [90] and accumulating preclinical models have verified the therapeutic benefit of TIM-3 blockade by regulating TME and restricting tumor growth especially in combination with PD-1 blockade [91].
Clinical trials on TIM-3
To date, at least eight TIM-3 antagonistic mAbs have been registered on ClinicalTrials.gov. TSR-022 (Cobolimab), a novel IgG4 anti-TIM-3 mAb developed by Tesaro entered the first phase I clinical trial in 2016 (NCT02817633) [92]. This multicenter, open-label study intended to evaluate the safety and efficacy of TSR-022 as a monotherapy or in combination with TSR-042 (anti-PD-1 mAb) in patients with advanced solid tumor. The results have been released in 2018 Annual Meeting of the Society for Immunotherapy of Cancer (SITC) Conference [93]. Clinical benefits have been observed in the combination group, especially at a high dose of TSR-022 (300 mg) with a 15% ORR (3/20) and 40% stable disease (8/20) [93]. Another two clinical trials including TSR-022 are still recruiting patients with no clinical results available (NCT03307785, NCT03680508). MBG453 is another anti-TIM-3 mAb produced by Novartis. Similar to TSR-022, the first clinical trial aimed to assess the safety and efficacy of MBG453 as single agent or in combination with PDR001 (an anti-PD-1 mAb) in advanced malignancies patients (NCT02608268). Another clinical trial was conducted in patients with acute myelocytic leukemia or high-risk myelodysplastic syndromes (NCT03066648). Sym023, is a recombinant, fully human antibody that bound TIM-3 [94]. A phase I trial evaluating the safety, tolerability, and dose-limiting toxicities of sym023 is recruiting at present (NCT03489343). Other TIM-3 inhibitors INCAGN2390, LY3321367, BMS-986258 and SHR1702 are also being tested in phase I trial alone (INCAGN02390 NCT03652077) or in combination with anti-PD-1/PD-L1 mAb (LY3321367 NCT03099109; BMS-986258 NCT03446040; SHR1702 NCT03871855) in the metastatic setting [84, 95]. RO7121661 is a bispecific antibody targeting PD-1 and TIM-3 simultaneously. It was developed by Roche and a phase I dose escalation and expansion study has been ongoing on advanced solid tumors (NCT03708328).
T cell immunoglobulin and ITIM domain (TIGIT)
The biology of TIGIT
TIGIT was first identified by Yu and his colleagues as an immune checkpoint rheostat that suppress the activation of T cells in 2009 [22]. Subsequently, it was described by other groups with each group giving a different name including WUCAM [96], Vstm3 [97], and Vsig9 [98]. TIGIT gene is located on human chromosome 3q13.31 and encodes a 244-amino acid transmembrane glycoprotein. The protein includes an extracellular IgV region, a transmembrane domain, and a cytoplasmic tail that harbors a canonical ITIM and an immunoglobulin tail tyrosine (ITT)-like phosphorylation motif [22]. The expression of TIGIT was demonstrated to be tightly restricted to lymphocytes, mainly on T cell subsets (including Tregs and memory T cells) and NK cells [22, 99]. TIGIT binds two ligands, namely CD155 (PVR or Necl-5) and CD112 (nectin-2, also known as PRR2 or PVRL2) with different affinity. Whether nectin-3 is another ligand for TIGIT is still in question [99]. TIGIT exerts its immunosuppressive effects by competing with other counterparts, CD266 (DNAM-1) or CD96 [100]. CD226 delivered a positive co-stimulatory signal, while TIGIT delivered inhibitory signals. This group of interacting proteins formed a co-stimulatory axis that are similar to the CTLA-4/B7/CD28 pathway [101].
As TIGIT was initially identified by a genomic search for structures shared a conserved ITIM motif, its immunosuppressive effects were delineated as expected. The initial research believed that TIGIT suppressed T cell activation in an indirect way. Specifically, the engagement of TIGIT with CD155 on DCs induced phosphorylation of CD155 and Erk, increased the secretion of IL-10, thus inhibiting T cell responses indirectly [22]. Subsequent studies demonstrated that TIGIT could also directly suppress T cell function by competing with CD226 [97, 102]. The role of TIGIT molecule in NK cells has been well studied. Stanietsky et al. indicated that ligation of TIGIT could lead to the inhibition of NK cells cytotoxicity through its cytoplasmic ITIM domain both in human and mouse [99, 103]. Furthermore, the major role of ITT-like motif in negatively modulating NK cells has been proved by two independent studies [104, 105]. Work from the Kurtulus group showed that the expression of TIGIT on Tregs was critically involved in Treg suppressive function [106]. Interestingly, Gur and his co-works discovered that TIGIT could directly bind to the Fap2 protein derived from Fusobacterium nucleatum, triggering a negative signal to suppress the activities of NK cells and T cells, and hence mediating a tumor-immune evasion mechanism [107]. Many groups generated agonistic anti-TIGIT mAb to verify the effect of TIGIT, indeed, they consistently reported a direct inhibitory effect on T cell proliferation [97, 102, 108]. Recently, some groups have reported that co-blockade of TIGIT with other checkpoint receptors, such as PD-1 and TIM-3, can exert synergistic effects in regulating antitumor responses [106, 109, 110].
Clinical trials on TIGIT
Based on the promising preclinical results, targeting TIGIT as a strategy for cancer treatment attracts the attention of many pharmaceutical companies, especially combined with ant-PD-1/PD-L1 mAb. There are at least six major agents targeting TIGIT now, focusing on three products. MK-7684, a candidate anti-TIGIT drug developed by Merck entered into a phase I clinical trial to analysis the safety, efficacy, and pharmacokinetics of MK-7684 as monotherapy and in combination with pembrolizumab in metastatic solid tumors (NCT02964013) [111]. The early phase I data was announced at the SITC’s 3rd Annual Meeting in 2018. Sixty eight individuals were enrolled with 34 patients in the monotherapy and 34 patients in combination arms. Finally, one PR and eight PR were observed in these two groups, and the disease control rates were 35 and 47%, respectively [112]. Etigilimab (OMP-313 M32) is a humanized mAb that developed to block TIGIT from binding CD155. It was developed by OncoMed/Celgene and entered the first phase I clinical trial in April 2017 (NCT03119428). This open-label research was designed to evaluate the safety and tolerability of Etigilimab as a single agent or in combination with an anti-PD-1 mAb in patients with advanced malignancies. At the 2017 American Association for Cancer Research Annual Meeting, OncoMed presented some positive results from several preclinical trials, thus its clinic performance was worth pursuing [111]. Another anti-TIGIT candidate drugs made by Genentech was named Tiragolumab (MTIG7192A, RG-6058). It’s also a fully human mAb designed to engage to TIGIT and hinder its interaction with CD155. There were two clinical trials about Tiragolumab registered on ClinicalTrials. gov (NCT02794571, NCT03563716). Other drugs, such as BMS-986207 made by Bristol-Myers Squibb, AB-154 made by Arcus biosciences and ASP-8374 made by Potenza also have initiated their phase I clinical trial with no clinical results reported [111].
VISTA, B7-H3, BTLA, and Siglec-15
Apart from these three new immune checkpoints mentioned above, many other immune checkpoint co-inhibitors are also attractive targets, with a few drugs step into clinical trials. VISTA, is also known as PD-1 homolog (PD-1H), DD1α, Gi24, differentiation of embryonic stem cells 1 (Dies1), and B7-H5 [113]. It was first described as an IgSF ligand which can negatively regulate T cell responses in mouse [23]. Subsequently, the same laboratory presented the characteristic of human VISTA [114]. It is a type I transmembrane protein with an extracellular IgV domain, a stalk region, a transmembrane segment, and a cytoplasmic tail. Structural analysis shows that the IgV domain of VISTA shares a sequence homology both to CD28 and B7 families, while the full-length VISTA harbors a highest identity with PD-1 [115]. But unlike PD-1, VISTA don’t include a classical ITIM or ITSM motif in the cytoplasmic domain, the intracellular tail contains two potential protein kinase C binding sites and a proline rich motif that may function as docking sites, suggesting that VISTA has the potentially function as both a receptor and a ligand [115]. Up to now the counter structures for VISTA has not been well identified [116], and VSIG-3 was reported as a novel ligand for VISTA a short time ago [117]. VISTA was highly expressed on myeloid cells and a lesser extent on T cells, but not on tumor cells within the TME [118]. The preclinical studies on multiple murine models showed that VISTA blockade improved the infiltration, proliferation, and effector function of tumor-infiltrating T cells within the TME, thus altered the suppressive character of the TME [118]. JNJ-61610588 is a fully human IgG1 anti-VISTA mAb made by Johnson & Johnson. The phase I clinical trial was intended to evaluate safety and pharmacokinetics of JNJ-61610588 in patients with advanced cancers (NCT02671955) [119]. Another candidate, CA-170, is an oral inhibitor which can selectively target both PD-L1 and VISTA. The results from preclinical models showed remarkable anti-tumor effects with well-tolerance and the phase I clinical trial in patients with advanced solid tumor and lymphomas is currently recruiting (NCT02812875) [119]. Based on the newest result published by Blando et al., VISTA was regard as a promising target for patients with pancreatic cancer [120] and the HMBD-002, a novel anti-VISTA antibody developed by Hummingbird Bioscience, have received a financial assistance from Cancer Prevention and Research Institute of Texas (CPRIT) with the plan to initiate clinical trials in 2020.
B7-H3, also named CD276, is a type I transmembrane glycoprotein that is encoded on human chromosome 15 [24]. It was discovered as early as 2001 [24]. The initial study described it as a positive co-stimulator for it can stimulate the T cell response and IFN-γ production [24]. But recently studies reported that B7-H3 was involved in the inhibition of T cells [121, 122]. The receptor for B7-H3 has not yet been identified and it may explain the intricate immunomodulatory activity of B7-H3 for it may have more than one binding partner with distinct function [123]. The expression of B7-H3 protein can be detected on activated immune cells such as T cells, NK cells, and APCs. More importantly, it was overexpressed on a wide spectrum of tumor tissue and linked to disease states and prognosis [124]. Recently, Enoblituzumab (MGA271), an engineered Fc humanized IgG1 mAb against B7-H3, has been developed and brought to clinic trials [125]. Among the five clinical trials about Enoblituzumab, one of which had been completed but did not reported the final results. Another agents MGD009, is a bispecific mAb designed to bind both CD3 on T cells and B7-H3 on tumor cells [36]. It is being studied on two phase I clinical studies in patients with B7-H3 expression (NCT02628535, NCT03406949) [36]. Furthermore, 8H9 (omburtamab) is an antibody specific to B7-H3 [126]. It has showed a positive clinical efficacy as an antibody drug conjugate after it was labeled with radioactive iodine-131 (131I) and administrated to patients with metastatic central nervous system (CNS) neuroblastoma [127]. Currently, clinical trials with radiolabeled 8H9 have been evaluated on peritoneal cancers, gliomas, and CNS (NCT01099644, NCT01502917, NCT00089245 et al.) The newest result of NCT01502917 supported the further study in expanded cohort [128].
BTLA (CD272) is identified as another inhibitory receptor that belongs to CD28 superfamily [113]. It is located on human chromosome 3 in q13.2 and encodes a 289-amino acid type I glycosylated transmembrane protein [25]. Similar to PD-1 and CTLA-4, the protein structure of BTLA includes a single extracellular region, a transmembrane domain and cytoplasmic domain. The ITIM and ITSM within the cytoplasmic tail mediate a negative signaling to T cells by recruiting the SHP-1 and SHP-2 [129]. BTLA was expressed on mature lymphocytes (such as B cells, T cells, and Tregs), macrophages, and mature bone marrow-derived DCs [130]. Herpesvirus entry mediator (HVEM), a member of the tumor necrosis factor receptor superfamily (TNFRSF), was identified as the unique BTLA ligand in 2005 [131]. But BTLA was not the unique binding partner for HVEM, it competed with other two TNF family members, LIGHT and lymphotoxin-α, as well as IgSF member CD160 for binding to HVEM [132]. CD160 is another negative regulator of T cell while LIGHT is a costimulatory molecule [133, 134]. The ligation of BTLA with HVEM triggered the inhibition of T cell proliferation and cytokine production [131]. At present, there are no clinical trials opened for BTLA. But, in the past April, Junshi Biosciences announced that the world’s first anti-BTLA mAb, TAB004/JS004, have been approved for clinical trial by FDA [135].
A recent publication reported a new immune suppressor, sialic acid-binding immunoglobulin-like lectin 15 (Siglec-15) [136]. The team of Dr. Lieping Chen elegantly demonstrated the expression of Siglect-15 (mainly on cancer cells, macrophages, and myeloid cells) and the inhibitory role of Siglect-15 in regulation of T cell responses. More importantly, they revealed that both genetic ablation and antagonize antibody of Siglec-15 suppressed the growth of tumor in murine models [136]. Right now, a clinical trial lead by Chen’s group is recruiting to test the efficacy of NC318 (an anti-Siglec-15 mAb) in solid tumors (NCT03665285) [136, 137].
Conclusion
The success of CTLA-4 or PD-1/PD-L1 blockade catalyzed the enthusiasm for a new class of antibody that block negative immune checkpoint regulators for cancer therapy. The past two decades have witnessed the significant progress in identifying alternative targets and developing novel specific agents in treating cancer. As we described above, considerable immune checkpoints have been explored with some been chosen as novel therapeutic targets by pharmaceutical companies. Although the number of clinical trials about these emerging immune modulators, such as anti-LAG-3 antibody and anti-TIM-3 antibody, registered on ClinialTrial.gov has grown exponentially, no drugs entered the clinic up to date. There are still some puzzles to be solved, like identification of ligand for VISTA and B7-H3 which may be the key to fully understand their therapeutic potential. In addition, while the results of monotherapy treatments are compelling, more attempts should be made to design rational combinations of immune-therapeutics that target non-redundant pathways to achieve synergistic effects in inhibiting tumor growth. We are still in the early stage of understanding these new immune systems with the aim that more candidates’ agents can transform to clinical fields and achieve even greater success than that initially observed with CTLA-4 or PD-1 blockade.
Availability of data and materials
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Abbreviations
- AEs:
-
Adverse events
- B7-H3:
-
B7 homolog 3 protein
- BTLA:
-
B and T cell lymphocyte attenuator
- Ceacam-1:
-
Carcinoembryonic antigen cell adhesion molecule 1
- CNS:
-
Central nervous system
- CTLA-4:
-
Cytotoxic T lymphocyte-associated antigen-4
- DC:
-
Dendritic cells
- FDA:
-
United States Food and Drug Administration
- HMGB1:
-
High-mobility group protein B1
- HVEM:
-
Herpesvirus entry mediator
- ICI:
-
Immune checkpoint inhibitor
- IgSF:
-
Ig superfamily
- IgV:
-
Ig variable region
- ITIM:
-
Immune receptor tyrosine based inhibitory motif
- ITSM:
-
Immune receptor tyrosine-based switch motif
- ITT:
-
Immunoglobulin tail tyrosine
- LAG-3:
-
Lymphocyte activation gene-3
- mAb:
-
Monoclonal antibody
- MBC:
-
Metastatic breast cancer
- MHC:
-
Major histocompatibility complex
- NK:
-
Natural killer
- NSCLC:
-
Non-small cell lung cancer
- ORR:
-
Objective response rate
- PD-1:
-
Programmed cell death protein-1
- PD-L1:
-
Programmed cell death ligand 1
- PR:
-
Partial response
- PtdSer:
-
Phosphatidyl serine
- RCC:
-
Renal cell carcinoma
- SHP1/2:
-
Src homology 2 domain containing phosphatases 1/2
- Siglec-15:
-
Sialic acid-binding immunoglobulin-like lectin 15
- SITC:
-
Society for Immunotherapy of Cancer
- TCR:
-
T cell receptor
- TIGIT:
-
T cell immunoglobulin and ITIM domain
- TIM-3:
-
T cell immunoglobulin and mucin-domain containing-3
- TME:
-
Tumor microenvironment
- Tregs:
-
Regulatory T cells
- VISTA:
-
V-domain Ig suppressor of T cell activation
References
Yi M, Jiao D, Xu H, Liu Q, Zhao W, Han X, et al. Biomarkers for predicting efficacy of PD-1/PD-L1 inhibitors. Mol Cancer. 2018;17:129.
Yu S, Li A, Liu Q, Li T, Yuan X, Han X, et al. Chimeric antigen receptor T cells: a novel therapy for solid tumors. J Hematol Oncol. 2017;10:78.
Yu S, Li A, Liu Q, Yuan X, Xu H, Jiao D, et al. Recent advances of bispecific antibodies in solid tumors. J Hematol Oncol. 2017;10:155.
Ballas ZK. The 2018 Nobel prize in physiology or medicine: an exemplar of bench to bedside in immunology. J Allergy Clin Immunol. 2018;142:1752–3.
Wang J, Yuan R, Song W, Sun J, Liu D, Li Z. PD-1, PD-L1 (B7-H1) and tumor-site immune modulation therapy: the historical perspective. J Hematol Oncol. 2017;10:34.
Krummel MF, Allison JP. CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. J Exp Med. 1995;182:459–65.
Chen L. Co-inhibitory molecules of the B7-CD28 family in the control of T-cell immunity. Nat Rev Immunol. 2004;4:336–47.
Leach D, Krummel M, Allison J. Enhancement of antitumor immunity by CTLA-4 blockade. Science (New York, NY). 1996;271:1734–6.
Yi M, Yu S, Qin S, Liu Q, Xu H, Zhao W, et al. Gut microbiome modulates efficacy of immune checkpoint inhibitors. J Hematol Oncol. 2018;11:47.
Cameron F, Whiteside G, Perry C. Ipilimumab: first global approval. Drugs. 2011;71:1093–104.
Okazaki T, Honjo T. PD-1 and PD-1 ligands: from discovery to clinical application. Int Immunol. 2007;19:813–24.
Zhu B, Tang L, Chen S, Yin C, Peng S, Li X, et al. Targeting the upstream transcriptional activator of PD-L1 as an alternative strategy in melanoma therapy. Oncogene. 2018;37:4941–54.
Berger KN, Pu JJ. PD-1 pathway and its clinical application: a 20year journey after discovery of the complete human PD-1 gene. Gene. 2018;638:20–5.
Kwok G, Yau TC, Chiu JW, Tse E, Kwong YL. Pembrolizumab (Keytruda). Hum Vaccin Immunother. 2016;12:2777–89.
Fuereder T. Immunotherapy for head and neck squamous cell carcinoma. Memo. 2016;9:66–9.
Peters S, Kerr KM, Stahel R. PD-1 blockade in advanced NSCLC: a focus on pembrolizumab. Cancer Treat Rev. 2018;62:39–49.
Bellmunt J, de Wit R, Vaughn DJ, Fradet Y, Lee JL, Fong L, et al. Pembrolizumab as second-line therapy for advanced urothelial carcinoma. New Engl J Med. 2017;376:1015–26.
Frenel JS, Le Tourneau C, O'Neil B, Ott PA, Piha-Paul SA, Gomez-Roca C, et al. Safety and efficacy of Pembrolizumab in advanced, programmed death ligand 1-positive cervical cancer: results from the phase Ib KEYNOTE-028 trial. J Clin Oncol. 2017;35:4035–41.
Li Z, Song W, Rubinstein M, Liu D. Recent updates in cancer immunotherapy: a comprehensive review and perspective of the 2018 China cancer immunotherapy workshop in Beijing. J Hematol Oncol. 2018;11:142.
Andrews LP, Marciscano AE, Drake CG, Vignali DA. LAG3 (CD223) as a cancer immunotherapy target. Immunol Rev. 2017;276:80–96.
Monney L, Sabatos CA, Gaglia JL, Ryu A, Waldner H, Chernova T, et al. Th1-specific cell surface protein Tim-3 regulates macrophage activation and severity of an autoimmune disease. Nature. 2002;415:536–41.
Yu X, Harden K, Gonzalez LC, Francesco M, Chiang E, Irving B, et al. The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat Immunol. 2009;10:48–57.
Wang L, Rubinstein R, Lines JL, Wasiuk A, Ahonen C, Guo Y, et al. VISTA, a novel mouse Ig superfamily ligand that negatively regulates T cell responses. J Exp Med. 2011;208:577–92.
Chapoval AI, Ni J, Lau JS, Wilcox RA, Flies DB, Liu D, et al. B7-H3: a costimulatory molecule for T cell activation and IFN-gamma production. Nat Immunol. 2001;2:269–74.
Yu X, Zheng Y, Mao R, Su Z, Zhang J. BTLA/HVEM signaling: milestones in research and role in chronic hepatitis B virus infection. Front Immunol. 2019;10:617.
Sharpe AH. Mechanisms of costimulation. Immunol Rev. 2009;229:5–11.
La-Beck NM, Jean GW, Huynh C, Alzghari SK, Lowe DB. Immune checkpoint inhibitors: new insights and current place in cancer therapy. Pharmacotherapy. 2015;35:963–76.
Lizee G, Overwijk WW, Radvanyi L, Gao J, Sharma P, Hwu P. Harnessing the power of the immune system to target cancer. Annu Rev Med. 2013;64:71–90.
Chambers CA, Kuhns MS, Egen JG, Allison JP. CTLA-4-mediated inhibition in regulation of T cell responses: mechanisms and manipulation in tumor immunotherapy. Annu Rev Immunol. 2001;19:565–94.
Rudd CE, Taylor A, Schneider H. CD28 and CTLA-4 coreceptor expression and signal transduction. Immunol Rev. 2009;229:12–26.
Sharma P, Wagner K, Wolchok JD, Allison JP. Novel cancer immunotherapy agents with survival benefit: recent successes and next steps. Nat Rev Cancer. 2011;11:805–12.
Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252–64.
Boussiotis VA. Molecular and biochemical aspects of the PD-1 checkpoint pathway. N Engl J Med. 2016;375:1767–78.
Toor SM, Sasidharan Nair V, Decock J, Elkord E. Immune checkpoints in the tumor microenvironment. Semin Cancer Biol. 2019. https://doi.org/10.1016/j.semcancer.2019.06.021.
Das M, Zhu C, Kuchroo VK. Tim-3 and its role in regulating anti-tumor immunity. Immunol Rev. 2017;276:97–111.
Marin-Acevedo JA, Dholaria B, Soyano AE, Knutson KL, Chumsri S, Lou Y. Next generation of immune checkpoint therapy in cancer: new developments and challenges. J Hematol Oncol. 2018;11:39.
Huang RY, Eppolito C, Lele S, Shrikant P, Matsuzaki J, Odunsi K. LAG3 and PD1 co-inhibitory molecules collaborate to limit CD8+ T cell signaling and dampen antitumor immunity in a murine ovarian cancer model. Oncotarget. 2015;6:27359–77.
Huang RY, Francois A, McGray AR, Miliotto A, Odunsi K. Compensatory upregulation of PD-1, LAG-3, and CTLA-4 limits the efficacy of single-agent checkpoint blockade in metastatic ovarian cancer. Oncoimmunology. 2017;6:e1249561.
Koyama S, Akbay EA, Li YY, Herter-Sprie GS, Buczkowski KA, Richards WG, et al. Adaptive resistance to therapeutic PD-1 blockade is associated with upregulation of alternative immune checkpoints. Nat Commun. 2016;7:10501.
Fourcade J, Sun Z, Benallaoua M, Guillaume P, Luescher IF, Sander C, et al. Upregulation of Tim-3 and PD-1 expression is associated with tumor antigen-specific CD8+ T cell dysfunction in melanoma patients. J Exp Med. 2010;207:2175–86.
Triebel F, Jitsukawa S, Baixeras E, Roman-Roman S, Genevee C, Viegas-Pequignot E, et al. LAG-3, a novel lymphocyte activation gene closely related to CD4. J Exp Med. 1990;171:1393–405.
Huard B, Mastrangeli R, Prigent P, Bruniquel D, Donini S, El-Tayar N, et al. Characterization of the major histocompatibility complex class II binding site on LAG-3 protein. Proc Natl Acad Sci U S A. 1997;94:5744–9.
Goldberg MV, Drake CG. LAG-3 in cancer immunotherapy. Curr Top Microbiol Immunol. 2011;344:269–78.
Workman CJ, Vignali DA. The CD4-related molecule, LAG-3 (CD223), regulates the expansion of activated T cells. Eur J Immunol. 2003;33:970–9.
Li N, Workman CJ, Martin SM, Vignali DA. Biochemical analysis of the regulatory T cell protein lymphocyte activation gene-3 (LAG-3; CD223). J Immunol. 2004;173:6806–12.
Buisson S, Triebel F. LAG-3 (CD223) reduces macrophage and dendritic cell differentiation from monocyte precursors. Immunology. 2005;114:369–74.
Huang CT, Workman CJ, Flies D, Pan X, Marson AL, Zhou G, et al. Role of LAG-3 in regulatory T cells. Immunity. 2004;21:503–13.
Huard B, Tournier M, Triebel F. LAG-3 does not define a specific mode of natural killing in human. Immunol Lett. 1998;61:109–12.
Kisielow M, Kisielow J, Capoferri-Sollami G, Karjalainen K. Expression of lymphocyte activation gene 3 (LAG-3) on B cells is induced by T cells. Eur J Immunol. 2005;35:2081–8.
Andreae S, Piras F, Burdin N, Triebel F. Maturation and activation of dendritic cells induced by lymphocyte activation gene-3 (CD223). J Immunol. 2002;168:3874–80.
Huard B, Prigent P, Tournier M, Bruniquel D, Triebel F. CD4/major histocompatibility complex class II interaction analyzed with CD4- and lymphocyte activation gene-3 (LAG-3)-Ig fusion proteins. Eur J Immunol. 1995;25:2718–21.
Macon-Lemaitre L, Triebel F. The negative regulatory function of the lymphocyte-activation gene-3 co-receptor (CD223) on human T cells. Immunology. 2005;115:170–8.
Workman CJ, Dugger KJ, Vignali DA. Cutting edge: molecular analysis of the negative regulatory function of lymphocyte activation gene-3. J Immunol. 2002;169:5392–5.
Baixeras E, Huard B, Miossec C, Jitsukawa S, Martin M, Hercend T, et al. Characterization of the lymphocyte activation gene 3-encoded protein. A new ligand for human leukocyte antigen class II antigens. J Exp Med. 1992;176:327–37.
Kouo T, Huang L, Pucsek AB, Cao M, Solt S, Armstrong T, et al. Galectin-3 shapes antitumor immune responses by suppressing CD8+ T cells via LAG-3 and inhibiting expansion of Plasmacytoid dendritic cells. Cancer Immunol Res. 2015;3:412–23.
Xu F, Liu J, Liu D, Liu B, Wang M, Hu Z, et al. LSECtin expressed on melanoma cells promotes tumor progression by inhibiting antitumor T-cell responses. Cancer Res. 2014;74:3418–28.
Mao X, Ou MT, Karuppagounder SS, Kam TI, Yin X, Xiong Y, et al. Pathological alpha-synuclein transmission initiated by binding lymphocyte-activation gene 3. Science (New York, NY). 2016;353:aah3374
Wang J, Sanmamed MF, Datar I, Su TT, Ji L, Sun J, et al. Fibrinogen-like protein 1 is a major immune inhibitory ligand of LAG-3. Cell. 2019;176:334–347.e12.
Prigent P, El Mir S, Dreano M, Triebel F. Lymphocyte activation gene-3 induces tumor regression and antitumor immune responses. Eur J Immunol. 1999;29:3867–76.
Fougeray S, Brignone C, Triebel F. A soluble LAG-3 protein as an immunopotentiator for therapeutic vaccines: preclinical evaluation of IMP321. Vaccine. 2006;24:5426–33.
Huard B, Prigent P, Pages F, Bruniquel D, Triebel F. T cell major histocompatibility complex class II molecules down-regulate CD4+ T cell clone responses following LAG-3 binding. Eur J Immunol. 1996;26:1180–6.
Brignone C, Escudier B, Grygar C, Marcu M, Triebel F. A phase I pharmacokinetic and biological correlative study of IMP321, a novel MHC class II agonist, in patients with advanced renal cell carcinoma. Clin Cancer Res. 2009;15:6225–31.
Brignone C, Gutierrez M, Mefti F, Brain E, Jarcau R, Cvitkovic F, et al. First-line chemoimmunotherapy in metastatic breast carcinoma: combination of paclitaxel and IMP321 (LAG-3Ig) enhances immune responses and antitumor activity. J Transl Med. 2010;8:71.
Wang-Gillam A, Plambeck-Suess S, Goedegebuure P, Simon PO, Mitchem JB, Hornick JR, et al. A phase I study of IMP321 and gemcitabine as the front-line therapy in patients with advanced pancreatic adenocarcinoma. Invest New Drugs. 2013;31:707–13.
Dirix L, Triebel F. AIPAC: a phase IIb study of eftilagimod alpha (IMP321 or LAG-3Ig) added to weekly paclitaxel in patients with metastatic breast cancer. Future Oncol. 2019;15:1963–73.
Romano E, Michielin O, Voelter V, Laurent J, Bichat H, Stravodimou A, et al. MART-1 peptide vaccination plus IMP321 (LAG-3Ig fusion protein) in patients receiving autologous PBMCs after lymphodepletion: results of a phase I trial. J Transl Med. 2014;12:97.
Legat A, Maby-El Hajjami H, Baumgaertner P, Cagnon L, Abed Maillard S, Geldhof C, et al. Vaccination with LAG-3Ig (IMP321) and peptides induces specific CD4 and CD8 T-cell responses in metastatic melanoma patients--report of a phase I/IIa clinical trial. Clin Cancer Res. 2016;22:1330–40.
Yu X, Huang X, Chen X, Liu J, Wu C, Pu Q, et al. Characterization of a novel anti-human lymphocyte activation gene 3 (LAG-3) antibody for cancer immunotherapy. MAbs. 2019;11:1139–48.
Wierz M, Pierson S, Guyonnet L, Viry E, Lequeux A, Oudin A, et al. Dual PD1/LAG3 immune checkpoint blockade limits tumor development in a murine model of chronic lymphocytic leukemia. Blood. 2018;131:1617–21.
Kwiatkowska D, Kluska P, Reich A. Beyond PD-1 immunotherapy in malignant melanoma. Dermatol Ther. 2019;9:243–57.
Papadopoulos KP, Lakhani NJ, Johnson ML, Park H, Wang D,Yap TA. A study of REGN3767, an anti-LAG-3 antibody, alone and in combination with cemiplimab (REGN2810), an anti-PD1 antibody, in advanced cancers. J Clin Oncol. 2018;36(suppl):TPS3127.
Ghosh S, Sharma G, Travers J, Kumar S, Choi J, Jun HT, et al. TSR-033, a novel therapeutic antibody targeting LAG-3, enhances T-cell function and the activity of PD-1 blockade in vitro and in vivo. Mol Cancer Ther. 2019;18:632–41.
Zettl M, Wurm M, Schaaf O, Tirapu I, Mostbock S, Reschke M, et al. Characterization of the LAG-3 targeting antibody BI 754111 in monotherapy and in combination with the anti-PD-1 antibody BI 754091. Cancer Res. 2018;78(suppl):4547.
Grandal MM, Melander MC, Bhatia VK, Gjetting T, Lindsted T, Frohlich C, et al. Preclinical characterization of Sym022, a novel anti-LAG3 antibody. Cancer Res. 2018;78(suppl):5626.
Woo SR, Turnis ME, Goldberg MV, Bankoti J, Selby M, Nirschl CJ, et al. Immune inhibitory molecules LAG-3 and PD-1 synergistically regulate T-cell function to promote tumoral immune escape. Cancer Res. 2012;72:917–27.
Kraman M, Fosh N, Kmiecik K, Everett K, Zimarino C, Faroudi M, et al. Dual blockade of PD-L1 and LAG-3 with FS118, a unique bispecific antibody, induces CD8+T-cell activation and modulates the tumor microenvironment to promote antitumor immune responses. Cancer Res. 2018;78(suppl):2719.
La Motte-Mohs R, Shah K, Brown JG, Smith D, Gorlatov S, Ciccarone V, et al. Preclinical characterization of MGD013, a PD-1 x LAG-3 bispecific DART (R) molecule. J Immunother Cancer. 2017;5(suppl 2):P337.
He Y, Cao J, Zhao C, Li X, Zhou C, Hirsch FR. TIM-3, a promising target for cancer immunotherapy. OncoTargets Ther. 2018;11:7005–9.
Meyers JH, Sabatos CA, Chakravarti S, Kuchroo VK. The TIM gene family regulates autoimmune and allergic diseases. Trends Mol Med. 2005;11:362–9.
van de Weyer PS, Muehlfeit M, Klose C, Bonventre JV, Walz G, Kuehn EW. A highly conserved tyrosine of Tim-3 is phosphorylated upon stimulation by its ligand galectin-9. Biochem Biophys Res Commun. 2006;351:571–6.
Anderson AC. Tim-3, a negative regulator of anti-tumor immunity. Curr Opin Immunol. 2012;24:213–6.
Jan M, Chao MP, Cha AC, Alizadeh AA, Gentles AJ, Weissman IL, et al. Prospective separation of normal and leukemic stem cells based on differential expression of TIM3, a human acute myeloid leukemia stem cell marker. Proc Natl Acad Sci U S A. 2011;108:5009–14.
Huang X, Bai X, Cao Y, Wu J, Huang M, Tang D, et al. Lymphoma endothelium preferentially expresses Tim-3 and facilitates the progression of lymphoma by mediating immune evasion. J Exp Med. 2010;207:505–20.
Liu F, Liu Y, Chen Z. Tim-3 expression and its role in hepatocellular carcinoma. J Hematol Oncol. 2018;11:126.
Zhu C, Anderson AC, Schubart A, Xiong H, Imitola J, Khoury SJ, et al. The Tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat Immunol. 2005;6:1245–52.
Kang CW, Dutta A, Chang LY, Mahalingam J, Lin YC, Chiang JM, et al. Apoptosis of tumor infiltrating effector TIM-3+CD8+ T cells in colon cancer. Sci Rep. 2015;5:15659.
Chiba S, Baghdadi M, Akiba H, Yoshiyama H, Kinoshita I, Dosaka-Akita H, et al. Tumor-infiltrating DCs suppress nucleic acid-mediated innate immune responses through interactions between the receptor TIM-3 and the alarmin HMGB1. Nat Immunol. 2012;13:832–42.
Huang YH, Zhu C, Kondo Y, Anderson AC, Gandhi A, Russell A, et al. CEACAM1 regulates TIM-3-mediated tolerance and exhaustion. Nature. 2015;517:386–90.
Nakayama M, Akiba H, Takeda K, Kojima Y, Hashiguchi M, Azuma M, et al. Tim-3 mediates phagocytosis of apoptotic cells and cross-presentation. Blood. 2009;113:3821–30.
Zhang Y, Cai P, Liang T, Wang L, Hu L. TIM-3 is a potential prognostic marker for patients with solid tumors: a systematic review and meta-analysis. Oncotarget. 2017;8:31705–13.
Hahn AW, Gill DM, Pal SK, Agarwal N. The future of immune checkpoint cancer therapy after PD-1 and CTLA-4. Immunotherapy. 2017;9:681–92.
Murtaza A, Laken H, Correia JDS, McNeeley P, Altobell L, Zhang J, et al. Discovery of TSR-022, a novel, potent anti-human TIM-3 therapeutic antibody. Eur J Cancer. 2016;69(Suppl 1):S102.
Chen X, Song X, Li K, Zhang T. FcgammaR-binding is an important functional attribute for immune checkpoint antibodies in cancer immunotherapy. Front Immunol. 2019;10:292.
Lindsted T, Gad M, Grandal MV, Frolich C, Bhatia VK, Gjetting T, et al. Preclinical characterization of Sym023 a human anti-TIM3 antibody with a novel mechanism of action. Cancer Res. 2018;78(Suppl):5629.
Mollica V, Di Nunno V, Gatto L, Santoni M, Cimadamore A, Cheng L, et al. Novel therapeutic approaches and targets currently under evaluation for renal cell carcinoma: waiting for the revolution. Clin Drug Investig. 2019;39:503–19.
Boles KS, Vermi W, Facchetti F, Fuchs A, Wilson TJ, Diacovo TG, et al. A novel molecular interaction for the adhesion of follicular CD4 T cells to follicular DC. Eur J Immunol. 2009;39:695–703.
Levin SD, Taft DW, Brandt CS, Bucher C, Howard ED, Chadwick EM, et al. Vstm3 is a member of the CD28 family and an important modulator of T-cell function. Eur J Immunol. 2011;41:902–15.
Anderson AC, Joller N, Kuchroo VK. Lag-3, Tim-3, and TIGIT: co-inhibitory receptors with specialized functions in immune regulation. Immunity. 2016;44:989–1004.
Stanietsky N, Simic H, Arapovic J, Toporik A, Levy O, Novik A, et al. The interaction of TIGIT with PVR and PVRL2 inhibits human NK cell cytotoxicity. Proc Natl Acad Sci U S A. 2009;106:17858–63.
Dougall WC, Kurtulus S, Smyth MJ, Anderson AC. TIGIT and CD96: new checkpoint receptor targets for cancer immunotherapy. Immunol Rev. 2017;276:112–20.
Le Mercier I, Lines JL, Noelle RJ. Beyond CTLA-4 and PD-1, the generation Z of negative checkpoint regulators. Front Immunol. 2015;6:418.
Lozano E, Dominguez-Villar M, Kuchroo V, Hafler DA. The TIGIT/CD226 axis regulates human T cell function. J Immunol. 2012;188:3869–75.
Stanietsky N, Rovis TL, Glasner A, Seidel E, Tsukerman P, Yamin R, et al. Mouse TIGIT inhibits NK-cell cytotoxicity upon interaction with PVR. Eur J Immunol. 2013;43:2138–50.
Liu S, Zhang H, Li M, Hu D, Li C, Ge B, et al. Recruitment of Grb2 and SHIP1 by the ITT-like motif of TIGIT suppresses granule polarization and cytotoxicity of NK cells. Cell Death Differ. 2013;20:456–64.
Li M, Xia P, Du Y, Liu S, Huang G, Chen J, et al. T-cell immunoglobulin and ITIM domain (TIGIT) receptor/poliovirus receptor (PVR) ligand engagement suppresses interferon-gamma production of natural killer cells via beta-arrestin 2-mediated negative signaling. J Biol Chem. 2014;289:17647–57.
Kurtulus S, Sakuishi K, Ngiow SF, Joller N, Tan DJ, Teng MW, et al. TIGIT predominantly regulates the immune response via regulatory T cells. J Clin Invest. 2015;125:4053–62.
Gur C, Ibrahim Y, Isaacson B, Yamin R, Abed J, Gamliel M, et al. Binding of the Fap2 protein of fusobacterium nucleatum to human inhibitory receptor TIGIT protects tumors from immune cell attack. Immunity. 2015;42:344–55.
Joller N, Hafler JP, Brynedal B, Kassam N, Spoerl S, Levin SD, et al. Cutting edge: TIGIT has T cell-intrinsic inhibitory functions. J Immunol. 2011;186:1338–42.
Johnston RJ, Comps-Agrar L, Hackney J, Yu X, Huseni M, Yang Y, et al. The immunoreceptor TIGIT regulates antitumor and antiviral CD8(+) T cell effector function. Cancer Cell. 2014;26:923–37.
Zhang Q, Bi J, Zheng X, Chen Y, Wang H, Wu W, et al. Blockade of the checkpoint receptor TIGIT prevents NK cell exhaustion and elicits potent anti-tumor immunity. Nat Immunol. 2018;19:723–32.
Solomon BL, Garrido-Laguna I. TIGIT: a novel immunotherapy target moving from bench to bedside. Cancer Immunol Immunother. 2018;67:1659–67.
SITC18: Merck highlights new LAG-3 and TIGIT data. https://www.fiercebiotech.com/biotech/sitc-merck-highlights-new-lag-3-and-tigit-data. Accessed 21 Sept 2019.
Ceeraz S, Nowak EC, Noelle RJ. B7 family checkpoint regulators in immune regulation and disease. Trends Immunol. 2013;34:556–63.
Lines JL, Pantazi E, Mak J, Sempere LF, Wang L, O'Connell S, et al. VISTA is an immune checkpoint molecule for human T cells. Cancer Res. 2014;74:1924–32.
Flies DB, Wang S, Xu H, Chen L. Cutting edge: a monoclonal antibody specific for the programmed death-1 homolog prevents graft-versus-host disease in mouse models. J Immunol. 2011;187:1537–41.
Nowak EC, Lines JL, Varn FS, Deng J, Sarde A, Mabaera R, et al. Immunoregulatory functions of VISTA. Immunol Rev. 2017;276:66–79.
Wang JH, Wu GP, Manick B, Hernandez V, Renelt M, Erickson C, et al. VSIG-3 as a ligand of VISTA inhibits human T-cell function. Immunology. 2019;156:74–85.
Le Mercier I, Chen W, Lines JL, Day M, Li J, Sergent P, et al. VISTA regulates the development of protective antitumor immunity. Cancer Res. 2014;74:1933–44.
Dempke WCM, Fenchel K, Uciechowski P, Dale SP. Second- and third-generation drugs for immuno-oncology treatment-the more the better? Eur Cancer (Oxford, England : 1990). 2017;74:55–72.
Blando J, Sharma A, Higa MG, Zhao H, Vence L, Yadav SS, et al. Comparison of immune infiltrates in melanoma and pancreatic cancer highlights VISTA as a potential target in pancreatic cancer. Proc Natl Acad Sci U S A. 2019;116:1692–7.
Prasad DV, Nguyen T, Li Z, Yang Y, Duong J, Wang Y, et al. Murine B7-H3 is a negative regulator of T cells. J Immunol. 2004;173:2500–6.
Suh WK, Gajewska BU, Okada H, Gronski MA, Bertram EM, Dawicki W, et al. The B7 family member B7-H3 preferentially down-regulates T helper type 1-mediated immune responses. Nat Immunol. 2003;4:899–906.
Janakiram M, Shah UA, Liu W, Zhao A, Schoenberg MP, Zang X. The third group of the B7-CD28 immune checkpoint family: HHLA2, TMIGD2, B7x, and B7-H3. Immunol Rev. 2017;276:26–39.
Castellanos JR, Purvis IJ, Labak CM, Guda MR, Tsung AJ, Velpula KK, et al. B7-H3 role in the immune landscape of cancer. Am J Clin Exp Immunol. 2017;6:66–75.
Benzon B, Zhao SG, Haffner MC, Takhar M, Erho N, Yousefi K, et al. Correlation of B7-H3 with androgen receptor, immune pathways and poor outcome in prostate cancer: an expression-based analysis. Prostate Cancer Prostatic Dis. 2017;20:28–35.
Ahmed M, Cheng M, Zhao Q, Goldgur Y, Cheal SM, Guo HF, et al. Humanized affinity-matured monoclonal antibody 8H9 has potent antitumor activity and binds to FG loop of tumor antigen B7-H3. J Biol Chem. 2015;290:30018–29.
Kramer K, Kushner BH, Modak S, Pandit-Taskar N, Smith-Jones P, Zanzonico P, et al. Compartmental intrathecal radioimmunotherapy: results for treatment for metastatic CNS neuroblastoma. J Neurooncol. 2010;97:409–18.
Souweidane MM, Kramer K, Pandit-Taskar N, Zhou Z, Haque S, Zanzonico P, et al. Convection-enhanced delivery for diffuse intrinsic pontine glioma: a single-Centre, dose-escalation, phase 1 trial. Lancet Oncol. 2018;19:1040–50.
Gavrieli M, Watanabe N, Loftin SK, Murphy TL, Murphy KM. Characterization of phosphotyrosine binding motifs in the cytoplasmic domain of B and T lymphocyte attenuator required for association with protein tyrosine phosphatases SHP-1 and SHP-2. Biochem Biophys Res Commun. 2003;312:1236–43.
Han P, Goularte OD, Rufner K, Wilkinson B, Kaye J. An inhibitory Ig superfamily protein expressed by lymphocytes and APCs is also an early marker of thymocyte positive selection. J Immunol. 2004;172:5931–9.
Sedy JR, Gavrieli M, Potter KG, Hurchla MA, Lindsley RC, Hildner K, et al. B and T lymphocyte attenuator regulates T cell activation through interaction with herpesvirus entry mediator. Nat Immunol. 2005;6:90–8.
Steinberg MW, Cheung TC, Ware CF. The signaling networks of the herpesvirus entry mediator (TNFRSF14) in immune regulation. Immunol Rev. 2011;244:169–87.
Murphy KM, Nelson CA, Sedy JR. Balancing co-stimulation and inhibition with BTLA and HVEM. Nat Rev Immunol. 2006;6:671–81.
Cai G, Anumanthan A, Brown JA, Greenfield EA, Zhu B, Freeman GJ. CD160 inhibits activation of human CD4+ T cells through interaction with herpesvirus entry mediator. Nat Immunol. 2008;9:176–85.
IND for Junshi Biosciences' FIH anti-BTLA antibody approved by FDA. http://www.junshipharma.com/upload/201904/26/201904260917018006.pdf. Accessed 21 Sept 2019.
Wang J, Sun JW, Liu LN, Flies DB, Nie XX, Toki M, et al. Siglec-15 as an immune suppressor and potential target for normalization cancer immunotherapy. Nat Med. 2019;25:656–66.
Cao G, Xiao Z, Yin Z. Normalization cancer immunotherapy: blocking Siglec-15! Signal Transduct Target Ther. 2019;4:10.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81572608, 81874120) and Wuhan Science and Technology Bureau (No. 2017060201010170).
Author information
Authors and Affiliations
Contributions
SQ performed the selection of literature, drafted the manuscript, and prepared the Figs. LX, MY, and SY helped to collect literatures, participate in the discussion and revise the manuscript. SL and KW carried out the design of this review and revised the manuscript. All authors contributed to this manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Qin, S., Xu, L., Yi, M. et al. Novel immune checkpoint targets: moving beyond PD-1 and CTLA-4. Mol Cancer 18, 155 (2019). https://doi.org/10.1186/s12943-019-1091-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12943-019-1091-2