Abstract
Background
The number of non-intubated general anesthesia outside the operating room is growing as the increasing demand for comfort treatment. Non-intubated general anesthesia outside the operating room requires rapid onset of anesthesia, smoothness, quick recovery, and few postoperative complications. Traditional anesthetic regimens (propofol alone or propofol and opioids/dezocine/midazolam, etc.) have severe respiratory and circulatory depression and many systemic adverse effects. In this paper, we compare the effectiveness and safety of propofol and subclinical doses of esketamine with other traditional regimens applied to non-intubated general anesthesia through a systematic review and meta-analysis.
Methods
We searched PubMed, Embase, Cochrane Library, Web of Science, CNKI, Wanfang, VIP, and Sinomed databases for the period from January 2000 to October 2022. We rigorously screened the literature according to predefined inclusion and exclusion criteria, while risk assessment of the studies was performed using The Cochrane Collaboration’s tool, and statistical analysis of the data was performed using RevMan 5.4 software. The main outcome indicators we evaluated were the various hemodynamic parameters and incidence of various adverse effects between the experimental and control groups after induction of anesthesia.
Results
After a rigorous screening process, a total of 14 papers were included in the final meta-analysis. After risk bias assessment, three of the papers were judged as low risk and the others were judged as having moderate to high risk. Forest plots were drawn for a total of 16 indicators. Meta-analysis showed statistically significant differences in HR’ WMD 3.27 (0.66, 5.87), MAP’ WMD 9.68 (6.13, 13.24), SBP’ WMD 5.42 (2.11, 8.73), DBP’ WMD 4.02 (1.15, 6.88), propofol dose’ SMD -1.39 (-2.45, -0.33), hypotension’ RR 0.30 (0.20, 0.45), bradycardia’ RR 0.33 (0.14, 0.77), hypoxemia or apnea’ RR 0.45 (0.23, 0.89), injection pain’ RR 0.28 (0.13, 0.60), intraoperative choking’ RR 0.62 (0.50, 0.77), intraoperative body movements’ RR 0.48 (0.29, 0.81) and overall incidence of adverse reactions’ RR 0.52 (0.39, 0.70).The indicators that were not statistically different were time to wake up’ WMD − 0.55 (-1.29, 0.19), nausea and vomiting 0.84’ RR (0.43, 1.67), headache and dizziness’ RR 1.57 (0.98, 2.50) and neuropsychiatric reaction’ RR 1.05 (0.28, 3.93). The funnel plot showed that the vast majority of studies fell within the funnel interval, but the symmetry was relatively poor.
Conclusion
In non-intubated general anesthesia, the combination of subclinical doses of esketamine and propofol did reduce circulatory and respiratory depression, injection pain, and other adverse effects, while the incidence of esketamine’s own side effects such as neuropsychiatric reactions did not increase, and the combination of the two did not cause the occurrence of new and more serious adverse reactions, and the combination of the two was safe and effective.
Trial registration
PROSPREO registration number: CRD 42022368966.
Similar content being viewed by others
Introduction
With the increasing popularity of comfort medicine, many people are choosing general anesthesia when they receive treatment, such as painless gastroenteroscopy, painless bronchoscopy, painless abortion, etc [1]. Many procedures that could previously be performed under local anesthesia are now being performed under general anesthesia for a better medical experience. As a result, the proportion of non-intubated general anesthesia outside the operating room is increasing dramatically. The risks of non-intubated general anesthesia outside the operating room cannot be underestimated due to the limitations of the operating site, equipment conditions, and anesthesia procedures. Non-intubated general anesthesia outside the operating room requires rapid onset of anesthesia, smoothness, rapid recovery, and few postoperative complications, which requires a very safe and effective anesthetic drug regimen.
Propofol is now basically one of the essential drugs for non-intubated general anesthesia outside the operating room, with rapid onset of action, smooth induction, rapid metabolism, and no side effects such as involuntary muscle tremors, etc [2]. Although propofol has many advantages that cannot be compared with other intravenous anesthetics, it still has some adverse effects that cannot be ignored, such as respiratory and circulatory depression, injection pain, etc [3]. Therefore, this creates the need to use other drugs in combination with propofol to minimize these side effects of propofol. But there is no consensus about which drug and propofol is the best pairing options [4]. Currently, the commonly used drugs in clinical practice are opioids (fentanyl, sufentanil, etc.), benzodiazepines (midazolam, etc.), and lidocaine, but because opioids and benzodiazepines themselves have strong side effects such as respiratory and circulatory depression, combining with propofol greatly increases the difficulty of respiratory and circulatory management, we considered whether there are more proper drug would possess better efficacy when paired with propofol [5,6,7,8]. Given the unique pharmacological characteristics of esketamine, it came into our consideration.
Esketamine, as the right-hand molecular structure of ketamine, acts like ketamine mainly by antagonizing the N-methyl-D-aspartate receptor [9]. However, compared to ketamine, esketamine has about twice the analgesic potency, so it can achieve the same effect as ketamine through a smaller dose, which can greatly reduce the occurrence of side effects such as neuropsychiatric reactions and secretion production during the awakening period [10]. Because of its sympathomimetic effect, we considered esketamine in combination with propofol, which can buffer the violent fluctuations of the circulation [11]. At the same time, esketamine has less effect on respiration and has analgesic effect, which can effectively compensate for the respiratory depression and injection pain caused by propofol [12]. Based on their pharmacological characteristics, we conclude that the combination of the two is very complementary in non-intubated general anesthesia procedures outside the operating room. The current clinical dose of esketamine alone for induction of anesthesia is usually 0.5-1 mg/kg, but the incidence of neuropsychiatric and other adverse effects caused by esketamine in this dose range is still high, while some studies have shown that subclinical doses (less than 0.5 mg/kg) of esketamine can produce analgesia with relatively few neuropsychiatric and other side effects [10, 13]. Many randomized controlled trials have been conducted to examine the efficacy of subclinical doses of esketamine and propofol together, and our study compares the efficacy and safety of propofol and subclinical doses of esketamine with other conventional regimens (propofol alone or propofol and opioids/dezocine/midazolam, etc.) applied to non-intubated general anesthesia through a systematic review and meta-analysis.
Methods
Search strategy
A computer-based search of databases including PubMed, Embase, Cochrane Library, Web of Science, CNKI, Wanfang, VIP, Sinomed. The search strategy was as follows:(“Propofol”) AND (“Esketamine”). The date parameter for the search was set from January 2000 to October 2022.
Inclusion and exclusion criteria
The inclusion criteria are as follows: (a) randomized controlled trials on humans, (b) non-intubated general anesthesia without any muscle relaxants, (c) only two anesthetics, propofol and esketamine, were used in the experimental group (d) subclinical doses (0.5 mg/kg less) of esketamine, and (e) adults over 18 years old.
The exclusion criteria are as follows: (a) data missing and (b) repeatedly published articles.
Quality assessment
All included articles were assessed independently by the review authors using The Cochrane Collaboration’s tool for assessing risk of bias. Disagreements and discrepancies were solved through a consensus discussion with correspondence author. The risk of bias was graded as high, uncertain or low according to the following seven critical domains with respect to The Cochrane Collaboration’s tool for assessing risk of bias: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting and other bias. The Cochrane Collaboration’s tool for assessing risk of bias proposes an approach for summarizing the risk of bias according to the seven domains, which was used judiciously. A study was judged to have low risk of bias when there was low risk of bias for all key domains and plausible bias was unlikely to seriously alter the results. Unclear risk of bias for one or more key domains resulted in an overall unclear risk of bias for the study, while high risk of bias for any domain resulted in an overall high risk of bias for the study.
Data extraction
To clarify the efficacy and incidence of adverse effects of different drug regimens in non-intubated anesthesia, we extracted basic information for each article (including author and year of publication), number of subjects and age groups, trial design protocol (dosing regimen for experimental and control groups), and results on the incidence of various adverse effects (including, but not limited to, e.g., hemodynamic parameters, respiratory depression, injection pain, etc.)
Statistical analysis
Statistical analysis of the data was performed using RevMan 5.4 (Cochrane Collaboration). According to the heterogeneity test results, the effect model is determined. I2 ≥ 50% indicates greater heterogeneity, and the random effect model is selected; I2 < 50% indicates that the heterogeneity is within the acceptable range, and the fixed effect model is selected. When P < 0.05, it was considered that there were significant differences in the changes of each outcome indices. The weighted mean difference (WMD) is used to represent the results of measurements using the same unit of measurement; otherwise, the standardized mean difference (SMD) is used to represent the results. All results were expressed with 95% confidence intervals (CI). Sensitivity analysis was used to assess the reliability and stability of the results. Funnel plots were drawn, and the publication bias was evaluated by the symmetry of the funnel plot and concentration of literature to the midline.
Results
Literature search results and profile analysis
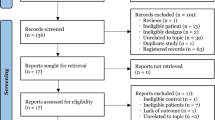
Pubmed searched 35 articles, Embase searched 135 articles, Cochrane Library searched 82 articles, web of science searched 32 articles, CNKI searched 74 articles, Wanfang searched 60 articles, VIP searched 37 articles, and Sinomed searched 56 articles, and a total of 14 articles were included in this review after being screened strictly according to the inclusion and exclusion criteria, and the screening process of these articles is shown in Fig. 1. The grouping of each study with outcome indicators and other information are summarized in Table 1.
Bias risk assessment of included literatures
The bias risk assessment tool recommended by the Cochrane Handbook for Systematic Reviews of Interventions was used to evaluate the quality of the included literatures, and the results are shown in Figs. 2 and 3. Articles Chen J 2022, Eberl S 2019 and Zhan Y 2022 were rated as having a low risk, and the remaining studies were rated as having a moderate to high risk.
Pooled analysis
The results of the meta-analysis are summarized in Table 2. Forest plots for each indicator are shown in detail in Figs. 4, 5, 6 and 7. The indicators that showed statistical differences were HR’ WMD 3.27 (0.66, 5.87), MAP’ WMD 9.68 (6.13, 13.24), SBP’ WMD 5.42 (2.11, 8.73), DBP’ WMD 4.02 (1.15, 6.88), propofol dose’ SMD -1.39 (-2.45, -0.33), hypotension’ RR 0.30 (0.20, 0.45), bradycardia’ RR 0.33 (0.14, 0.77), hypoxemia or apnea’ RR 0.45 (0.23, 0.89), injection pain’ RR 0.28 (0.13, 0.60), intraoperative choking’ RR 0.62 (0.50, 0.77), intraoperative body movements’ RR 0.48 (0.29, 0.81) and overall incidence of adverse reactions’ RR 0.52 (0.39, 0.70), while the indicators that did not show statistical differences were time to wake up’ WMD − 0.55 (-1.29, 0.19), nausea and vomiting 0.84’ RR (0.43, 1.67), headache and dizziness’ RR 1.57 (0.98, 2.50) and neuropsychiatric reaction’ RR 1.05 (0.28, 3.93). Meta-analysis’ results confirmed that subclinical doses of esketamine and propofol in combination slowed the dramatic fluctuations of hemodynamic parameters (HR, MAP, SBP, DBP), reduced the dose of propofol, and reduced the incidence of hypotension, bradycardia, hypoxemia and apnea, injection pain, intraoperative choking, intraoperative body movements, and overall adverse effects compared with propofol and other drugs.
Publication bias analysis
We will analyze publication bias by plotting funnel plots using fixed-effect models with statistically significant meta-analysis’s results, including hypotension, bradycardia, intraoperative cough, and neuropsychiatric response, and the results are shown in Fig. 8. The data in all four plots are relatively concentrated. However, the symmetry of the two funnel plots, hypotension and intraoperative choking, was not particularly good, indicating a possible publication bias.
Discussion
Based on the results of the meta-analysis, we can conclude that the combination of propofol and subclinical doses of esketamine in non-intubated general anesthesia does result in smoother hemodynamic (heart rate and blood pressure) fluctuations and a reduced incidence of hypotension and bradycardia in patients, as we previously hypothesized. Propofol has a significant depressant effect on the cardiovascular system, resulting in a decrease in cardiac output, cardiac index, per-beat index and total peripheral resistance during induction of anesthesia, due to the dual effect of peripheral vasodilation and direct cardiac depression [14]. The sympathomimetic effect of esketamine excites the sympathetic nerve center and increases the release of endogenous catecholamines, and also inhibits the reuptake of norepinephrine, which can reduce the inhibition of the cardiovascular system by propofol, an advantage that other drugs do not have in combination with propofol [15]. However, it should be noted that the sympathomimetic effect of ketamine is positive only in patients with normal sympathetic nervous system activity; otherwise, a flip-flop effect may occur [12]. Respiratory depression and apnea induced by clinical doses of propofol anesthesia is another major problem that cannot be ignored [16]. The combination of esketamine and propofol will definitely reduce the amount of propofol used, which will help to mitigate this side effect. At the same time, compared with opioids, the induction dose of clinical ketamine intravenously has only mild respiratory depression, but it can be recovered quickly, and the subclinical dose of esketamine may not worry about this problem, so the combination of the two drugs can significantly reduce the occurrence of respiratory depression and apnea compared with other drugs, which is also confirmed by the results of this meta-analysis [17]. In addition, esketamine can inhibit the production of pro-inflammatory factors and selectively block nociceptive signals from the reticular tract of the spinal cord, blocking pain transmission to the thalamus and cortical areas and producing analgesic effects [18]. It has also been reported that esketamine is able to agonize opioid receptors and produce analgesic effects, so esketamine should be effective in reducing the injection pain of propofol when it is injected before propofol, and the results of this meta-analysis verified this point [12]. Therefore, the use of esketamine significantly alleviates and compensates for the major side effects of propofol.
One of the non-negligible side effects of clinical induction doses of esketamine is the neuropsychiatric reaction during the awakening period, which is due to the ability of esketamine to activate prefrontal glutamate neurotransmission [19]. The use of subclinical doses definitely alleviates this side effect, and the results of the meta-analysis confirm that the combination of subclinical doses of esketamine and propofol does not increase the incidence of neuropsychiatric reactions compared to other matching regimens. In addition, esketamine increases cerebral blood flow and cerebral metabolic rate, and intracranial pressure increases with cerebral blood flow, while propofol has the effect of decreasing cerebral blood flow, cerebral oxygen metabolic rate and intracranial pressure, so propofol is able to reduce this side effect of esketamine [15]. In addition, there are some studies showing that esketamine has a rapid antidepressant effect, which may have a very positive meaning for some patients [20, 21]. Therefore, we can find that the use of propofol also greatly alleviates and compensates for the major side effects of esketamine.
Based on the results of the meta-analysis, we also found that the combination of propofol and subclinical doses of esketamine attenuated the incidence of intraoperative choking and intraoperative somatic movements, but the combination did not show differences in time to awakening, nausea and vomiting, and headache and dizziness compared with other pairing regimens. The combination of propofol and esketamine does not theoretically conflict with each other because they act on different receptor pathways, and their pharmacological properties suggest that they should compensate for each other’s deficiencies. The results of the meta-analysis confirmed that the overall incidence of adverse reactions was lower with subclinical doses of esketamine and propofol than with other combinations, and that no more serious side effects occurred.
In clinical practice, propofol is used in a wide range, and the dose has to be adjusted according to the patient’s specific situation, and the actual clinical dose range is about 1-2.5 mg/kg [22]. our specific dose of propofol use is oriented to the depth of patient sedation, so subclinical doses of esketamine and propofol combined, we cannot strictly limit the dose of propofol use, which is the reason why we did not use the dose of propofol as a variable for the screening of the literature when we performed the meta-analysis. We suggest that, in clinical practice, esketamine is injected first, followed by propofol. First, the injection of esketamine first can significantly reduce the injection pain of propofol, and second, because we mainly achieve our desired depth of sedation with propofol, and the use of esketamine is mainly to alleviate some side effects of propofol, we can limit the dose of esketamine, thus providing an effect that can alleviate the side effects of propofol without causing the side effects that esketamine itself. The optimal dose of subclinical esketamine is currently considered by most studies to be 0.3 mg/kg, which may require more randomized controlled trials to verify [23].
We found through our search that although there have been many randomized controlled trials that have confirmed that subclinical doses of esketamine and propofol combined significantly reduce the incidence of various adverse reactions compared with the combination of propofol and other drugs, no higher level of evidence has been found to validate this conclusion. Therefore, this meta-analysis is groundbreaking, although it still has many imperfections. We look forward to the emergence of more randomized controlled trials in the future to further update and improve the issues analyzed in this meta.
Our study is not without limitations. First, most of the studies we included were from China, which may lead to poor extrapolation of the findings and potentially large publication bias. Second, we included studies with a wide range of subjects’ ages and did not strictly distinguish between younger and older adults, which may have affected the results to some extent, so we excluded one study with all subjects in the older age group and found that the meta results for each outcome indicator containing this study did not reverse after excluding this study, suggesting that this study did not seriously affect the results of the meta. We would have liked to perform a subgroup analysis because the drugs used in the control group and propofol were different in each study, but we abandoned the subgroup analysis because of the small sample size of each subgroup.
Conclusion
In non-intubated general anesthesia, the combination of subclinical doses of esketamine and propofol did reduce circulatory and respiratory depression, injection pain, and other adverse effects, while the incidence of esketamine’s own side effects such as neuropsychiatric reactions did not increase, and the combination of the two did not cause the occurrence of new and more serious adverse reactions, and the combination of the two was safe and effective.
Data Availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Change history
31 October 2023
A Correction to this paper has been published: https://doi.org/10.1186/s12871-023-02326-3
References
Zhou S, Zhu Z, Dai W, Qi S, Tian W, Zhang Y, et al. National survey on sedation for gastrointestinal endoscopy in 2758 chinese hospitals. Br J Anaesth. 2021;127(1):56–64.
Nishizawa T, Suzuki H. Propofol for gastrointestinal endoscopy. United Eur Gastroenterol J. 2018;6(6):801–5.
Desousa KA. Pain on propofol injection: causes and remedies. Indian J Pharmacol. 2016;48(6):617–23.
Xu Y, Zheng Y, Tang T, Chen L, Zhang Y, Zhang Z. The effectiveness of esketamine and propofol versus dezocine and propofol sedation during gastroscopy: a randomized controlled study. J Clin Pharm Ther. 2022;47(9):1402–8.
Gao W, Sha B, Zhao Y, Fan Z, Liu L, Shen X. Comparison of simultaneous and sequential administration of fentanyl-propofol for surgical abortion: a randomized single-blinded controlled trial. Artif cells Nanomed Biotechnol. 2017;45(5):1045–50.
Rudner R, Jalowiecki P, Kawecki P, Gonciarz M, Mularczyk A, Petelenz M. Conscious analgesia/sedation with remifentanil and propofol versus total intravenous anesthesia with fentanyl, midazolam, and propofol for outpatient colonoscopy. Gastrointest Endosc. 2003;57(6):657–63.
Reimann FM, Samson U, Derad I, Fuchs M, Schiefer B, Stange EF. Synergistic sedation with low-dose midazolam and propofol for colonoscopies. Endoscopy. 2000;32(3):239–44.
Zhang L, Li C, Zhao C, Zhao Z, Feng Y. Analgesic comparison of dezocine plus propofol versus fentanyl plus propofol for gastrointestinal endoscopy: a meta-analysis. Medicine. 2021;100(15):e25531.
Li J, Wang Z, Wang A, Wang Z. Clinical effects of low-dose esketamine for anaesthesia induction in the elderly: a randomized controlled trial. J Clin Pharm Ther. 2022;47(6):759–66.
Wang J, Huang J, Yang S, Cui C, Ye L, Wang SY, et al. Pharmacokinetics and safety of esketamine in chinese patients undergoing painless gastroscopy in comparison with ketamine: a randomized, open-label clinical study. Drug Des Devel Ther. 2019;13:4135–44.
Geisslinger G, Hering W, Thomann P, Knoll R, Kamp HD, Brune K. Pharmacokinetics and pharmacodynamics of ketamine enantiomers in surgical patients using a stereoselective analytical method. Br J Anaesth. 1993;70(6):666–71.
Zanos P, Moaddel R, Morris PJ, Riggs LM, Highland JN, Georgiou P, et al. Ketamine and ketamine metabolite pharmacology: insights into therapeutic mechanisms. Pharmacol Rev. 2018;70(3):621–60.
Backonja M, Arndt G, Gombar KA, Check B, Zimmermann M. Response of chronic neuropathic pain syndromes to ketamine: a preliminary study. Pain. 1994;56(1):51–7.
Short CE, Bufalari A. Propofol anesthesia. Veterinary Clin North Am Small Anim Pract. 1999;29(3):747–78.
Annetta MG, Iemma D, Garisto C, Tafani C, Proietti R. Ketamine: new indications for an old drug. Curr Drug Targets. 2005;6(7):789–94.
Ren J, Lenal F, Yang M, Ding X, Greer JJ. Coadministration of the AMPAKINE CX717 with propofol reduces respiratory depression and fatal apneas. Anesthesiology. 2013;118(6):1437–45.
Gorlin AW, Rosenfeld DM, Ramakrishna H. Intravenous sub-anesthetic ketamine for perioperative analgesia. J Anaesthesiol Clin Pharmacol. 2016;32(2):160–7.
Niesters M, Martini C, Dahan A. Ketamine for chronic pain: risks and benefits. Br J Clin Pharmacol. 2014;77(2):357–67.
Abdallah CG, De Feyter HM, Averill LA, Jiang L, Averill CL, Chowdhury GMI, et al. The effects of ketamine on prefrontal glutamate neurotransmission in healthy and depressed subjects. Neuropsychopharmacology: official publication of the American College of Neuropsychopharmacology. 2018;43(10):2154–60.
Ng J, Rosenblat JD, Lui LMW, Teopiz KM, Lee Y, Lipsitz O, et al. Efficacy of ketamine and esketamine on functional outcomes in treatment-resistant depression: a systematic review. J Affect Disord. 2021;293:285–94.
Daly EJ, Singh JB, Fedgchin M, Cooper K, Lim P, Shelton RC, et al. Efficacy and safety of Intranasal Esketamine adjunctive to oral antidepressant therapy in treatment-resistant depression: a Randomized Clinical Trial. JAMA psychiatry. 2018;75(2):139–48.
Larijani GE, Gratz I, Afshar M, Jacobi AG. Clinical pharmacology of propofol: an intravenous anesthetic agent. DICP: the annals of pharmacotherapy. 1989;23(10):743–9.
Zheng Y, Xu Y, Huang B, Mai Y, Zhang Y, Zhang Z. Effective dose of propofol combined with a low-dose esketamine for gastroscopy in elderly patients: a dose finding study using dixon’s up-and-down method. Front Pharmacol. 2022;13:956392.
Chen J, Zou X, Hu B, Yang Y, Wang F, Zhou Q, et al. Effect of different doses of esketamine compared with fentanyl combined with propofol on hypotension in patients undergoing painless abortion surgery: a prospective, randomized, double-blind controlled clinical trial. BMC Anesthesiol. 2022;22(1):305.
Eberl S, Koers L, van Hooft J, de Jong E, Hermanides J, Hollmann MW, et al. The effectiveness of a low-dose esketamine versus an alfentanil adjunct to propofol sedation during endoscopic retrograde cholangiopancreatography: a randomised controlled multicentre trial. Eur J Anaesthesiol. 2020;37(5):394–401.
Li CL, Shen Y, Xu YF, Zhang ZQ. Clinical observation of different doses of esketamine combined with propofol in gastroscopy. Chin J New Drugs. 2022;31(8):773–7.
Zhan Y, Liang S, Yang Z, Luo Q, Li S, Li J, et al. Efficacy and safety of subanesthetic doses of esketamine combined with propofol in painless gastrointestinal endoscopy: a prospective, double-blind, randomized controlled trial. BMC Gastroenterol. 2022;22(1):391.
Chen SL, Zhu X, Zhao JF, Zhai XQ, Li J, Chang PA. The effect of esketamine on peri-examination stress response in patients undergoing painless gastrointestinal endoscopy (in chinese). China &Foreign Medical Treatment. 2022;41(14):14–8.
Gao CS, Huang FX, Li YL. The efficacy observation of intravenous injection of esketamine in abortion procedure (in chinese). Shandong Med J. 2022;62(11):56–8.
Ren YL, Zhang W. Application of esketamine combined with propofol in the minimally invasive surgery of benign breast tumors (in chinese). Clin Medication J. 2022;20(6):63–6.
Sheng K, Hu XW, Wu Y, Weng LJ. A clinical study of low-dose esketamine in combination with propofol in painless gastroscopy (in chinese). Chin J Med. 2022;57(3):341–4.
Shi YH. Effect of subclinical dose of esketamine combined with propofol in painless gastrointestinal endoscopy (in chinese). Fertility & Health. 2021(7):189–90.
Song ZQ, Li DD. The anesthesia effect of esketamine in the diagnosis and treatment of painless gastrointestinal endoscopy in elderly patients (in chinese). Acta Academiae Medicinae Weifang. 2021;43(6):475–7.
Sun YM, Yang J. Efficacy of esketamine combined with propofol in anesthesia for painless bronchoscopy (in chinese). Regimen. 2022(12):76–8.
Wan X, Yang QQ, Fan D, Feng FM, Ji MH. Effect of subanesthestic dose of esketamine combined with propofol on painless gastroenterological endoscopy (in chinese). J Clin Anesthesiology. 2022;38(2):144–8.
Cheng XL, Wang QQ, Ying ZQ. A study of propofol combined with low-dose esketamine in painless abortion. (in Chinese). 2021;19(15):119–21.
Acknowledgements
Not applicable.
Funding
This work is supported by the Science and Technology Plan Project of Sichuan Province, China (2020YJ0452, 2022YFS0439, 2023YFS0137, 2022NSFSC1567), Medical Research Subject of Chengdu Health Commission (2022037), National Natural Science Foundation of China (82170634).
Author information
Authors and Affiliations
Contributions
CHM and DXZ designed the study, DXZ and XGL conducted database searches, screened literatures, extracted study data and prepared Figs. 1, 2, 3, 4, 5, 6, 7 and 8, CHM performed the data analysis, prepared supplementary materials, Tables 1 and 2, and was the major contributor to the manuscript writing, LP and FQ provided critical review and revision to the manuscript. XL and LQ revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they do not have any conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: an error was found in affiliation 1.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Chen, H., Ding, X., Xiang, G. et al. Analysis of the efficacy of subclinical doses of esketamine in combination with propofol in non-intubated general anesthesia procedures - a systematic review and meta-analysis. BMC Anesthesiol 23, 245 (2023). https://doi.org/10.1186/s12871-023-02135-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12871-023-02135-8