Abstract
Introduction
Developments in anesthetic pharmacology have been aiming at minimizing physiological disturbance in addition to maintaining and improving titrateability, recovery profile, and patient experience. Remimazolam, a GABAAlpha receptor agonist, is a new intravenous anesthetic agent which has recently been approved for use. This analysis aimed to systematically compare the adverse drug events reported with the newly approved remimazolam in comparison to propofol for general anesthesia (GA) in patients undergoing surgery.
Methods
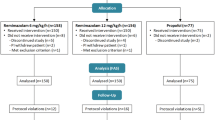
Electronic databases were searched from 15 May to 20 December 2023 for relevant publications which compared the outcomes reported with the newly approved remimazolam versus propofol in patients undergoing surgery. Relevant reported adverse drug events were the endpoints of this study. The statistical analysis was carried out using the latest version of the RevMan software. Data analysis was represented by risk ratio (RR) with 95% confidence intervals (CI).
Results
Sixteen studies with a total number of 1897 participants were included in this analysis; 1104 participants received remimazolam and 793 participants received propofol. The risks for hypotension (RR 0.50, 95% CI 0.43–0.58; P = 0.00001), hypoxemia (RR 0.43, 95% CI 0.19–0.99; P = 0.05), bradycardia (RR 0.53, 95% CI 0.36–0.78; P = 0.001), pain at injection site (RR 0.07, 95% CI 0.01–0.56; P = 0.01), and total adverse events (RR 0.33, 95% CI 0.24–0.47; P = 0.00001) were significantly lower with remimazolam. However, no significant differences were observed in terms of postoperative nausea and vomiting (RR 0.98, 95% CI 0.66–1.46; P = 0.93), dizziness (RR 0.42, 95% CI 0.11–1.57; P = 0.20), psychiatric symptoms (RR 1.09, 95% CI 0.45–2.67; P = 0.85), and respiratory depression (RR 0.81, 95% CI 0.24–2.76; P = 0.74).
Conclusion
Our current analysis showed that the newly approved remimazolam was apparently associated with significantly fewer adverse drug events in comparison to propofol for GA in patients undergoing surgery. Therefore, this new drug should be further studied and more research with larger population sizes should be carried out to confirm this hypothesis.





Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article. Furthermore, the data are available in all online databases including MEDLINE, EMBASE, Web of Science, and so on. References have been provided throughout the manuscript.
References
Poon KKS, Wong SHS. New and developing anesthesia drugs. Expert Opin Pharmacother. 2017;18(2):195–204.
Kim H, Park S-S, Shim J. Differences in midazolam premedication effects on recovery after short-duration ambulatory anesthesia with propofol or sevoflurane for gynecologic surgery in young patients: a randomized controlled trial. Medicine (Baltimore). 2020;99(47):e23194.
Ong LB, Plummer JL, Waldow WC, Owen H. Timing of midazolam and propofol administration for co-induction of anaesthesia. Anaesth Intensive Care. 2000;28(5):527–31.
Nordt SP, Clark RF. Midazolam: a review of therapeutic uses and toxicity. J Emerg Med. 1997;15(3):357–65.
Perel A. Non-anaesthesiologists should not be allowed to administer propofol for procedural sedation: a consensus statement of 21 European National Societies of Anaesthesia. Eur J Anaesthesiol. 2011;28(8):580–4.
Hemphill S, McMenamin L, Bellamy MC, Hopkins PM. Propofol infusion syndrome: a structured literature review and analysis of published case reports. Br J Anaesth. 2019;122(4):448–59.
Keam SJ. Remimazolam: first approval. Drugs. 2020;80(6):625–33.
Kilpatrick GJ. Remimazolam: non-clinical and clinical profile of a new sedative/anesthetic agent. Front Pharmacol. 2021;12:690875.
Goudra BG, Singh PM. Remimazolam: the future of its sedative potential. Saudi J Anaesth. 2014;8(3):388–91.
Noor N, Legendre R, Cloutet A, Chitneni A, Varrassi G, Kaye AD. A comprehensive review of remimazolam for sedation. Health Psychol Res. 2021;9(1):24514.
Lee A, Shirley M. Remimazolam: a review in procedural sedation. Drugs. 2021;81(10):1193–201.
Higgins JPT, Altman DG, Gøtzsche PC, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928.
Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25(9):603–5.
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700.
Choi JY, Lee HS, Kim JY, et al. Comparison of remimazolam-based and propofol-based total intravenous anesthesia on postoperative quality of recovery: a randomized non-inferiority trial. J Clin Anesth. 2022;82:110955.
Dai G, Pei L, Duan F, et al. Safety and efficacy of remimazolam compared with propofol in induction of general anesthesia. Minerva Anestesiol. 2021;87(10):1073–9.
Doi M, Morita K, Takeda J, et al. Efficacy and safety of remimazolam versus propofol for general anesthesia: a multicenter, single-blind, randomized, parallel-group, phase IIb/III trial. J Anesth. 2020;34(4):543–53.
Jeon Y-G, Kim S, Park J-H, et al. Incidence of intraoperative hypotension in older patients undergoing total intravenous anesthesia by remimazolam versus propofol: a randomized controlled trial. Medicine (Baltimore). 2023;102(49): e36440.
Kuang Q, Zhong N, Ye C, Zhu X, Wei F. Propofol versus remimazolam on cognitive function, hemodynamics, and oxygenation during one-lung ventilation in older patients undergoing pulmonary lobectomy: a randomized controlled trial. J Cardiothorac Vasc Anesth. 2023;37(10):1996–2005.
Liu T, Lai T, Chen J, et al. Effect of remimazolam induction on hemodynamics in patients undergoing valve replacement surgery: a randomized, double-blind, controlled trial. Pharmacol Res Perspect. 2021;9(5):e00851.
Sun WLM, Wan J, et al. Efficacy and safety of remimazolam tosilate versus propofol in patients undergoing day surgery: a prospective randomized controlled trial. BMC Anesthesiol. 2023;23(1):182.
Mao Y, Guo J, Yuan J, Zhao E, Yang J. Quality of recovery after general anesthesia with remimazolam in patients’ undergoing urologic surgery: a randomized controlled trial comparing remimazolam with propofol. Drug Des DevelTher. 2022;27(16):1199–209.
Matsumoto A, Satomi S, Kakuta N, et al. Remimazolam’s effects on postoperative nausea and vomiting are similar to those of propofol after laparoscopic gynecological surgery: a randomized controlled trial. J Clin Med. 2023;12(16):5402.
Oh EJ, Chung YJ, Lee J-H, et al. Comparison of propofol vs remimazolam on emergence profiles after general anesthesia: a randomized clinical trial. J Clin Anesth. 2023;90:111223.
Sato T, Nishiwaki K. Comparison of remimazolam and propofol in anesthetic management for awake craniotomy: a retrospective study. J Anesth. 2022;36(1):152–5.
Shi Fu, Chen Y, Li H, Zhang Y, Zhao T. Efficacy and safety of remimazolam tosilate versus propofol for general anesthesia in cirrhotic patients undergoing endoscopic variceal ligation. Int J Gen Med. 2022;13(15):583–91.
Tang F, Yi J-M, Gong H-Y, et al. Remimazolam benzenesulfonate anesthesia effectiveness in cardiac surgery patients under general anesthesia. World J Clin Cases. 2021;9(34):10595–603.
Tang L, Sun Y, Hao X, et al. Effect of general anaesthesia with remimazolam versus propofol on postoperative quality of recovery in patients undergoing ambulatory arthroscopic meniscus repair: a randomised clinical trial. BJA Open. 2023;28(8):100237.
Yue L, Ma X, Li Na, et al. Remimazolam versus propofol in combination with esketamine for surgical abortion: a double-blind randomized controlled trial. Clin Transl Sci. 2023;16(9):1606–16.
Zhang J, Wang X, Zhang Q, Wang Z, Zhu S. Application effects of remimazolam and propofol on elderly patients undergoing hip replacement. BMC Anesthesiol. 2022;22(1):118.
Zhu X, Wang H, Yuan S, et al. Efficacy and safety of remimazolam in endoscopic sedation-a systematic review and meta-analysis. Front Med (Lausanne). 2021;8:655042.
Xueyan Wu, Wang C, Gao H, et al. Comparison of remimazolam and propofol about safety outcome indicators during general anesthesia in surgical patients: a systematic review and meta-analysis. Minerva Anestesiol. 2023;89(6):553–64.
Bo-JyunJhuang B-H, Huang Y-T, Lai P-C. Efficacy and safety of remimazolam for procedural sedation: a meta-analysis of randomized controlled trials with trial sequential analysis. Front Med (Lausanne). 2021;27(8):641866.
Zhang S, Wang J, Ran R, Peng Y, Xiao Y. Efficacy and safety of remimazolam tosylate in hysteroscopy: a randomized, single-blind, parallel controlled trial. J Clin Pharm Ther. 2022;47(1):55–60.
Zhang X, Li S, Liu J. Efficacy and safety of remimazolam besylate versus propofol during hysteroscopy: single-centre randomized controlled trial. BMC Anesthesiol. 2021;21(1):156.
Liu X, Ding B, Shi F, et al. The efficacy and safety of remimazolam tosilate versus etomidate-propofol in elderly outpatients undergoing colonoscopy: a prospective, randomized, single-blind, non-inferiority trial. Drug Des Devel Ther. 2021;15:4675–85.
Chen S, Wang J, Xu X, et al. The efficacy and safety of remimazolam tosylate versus propofol in patients undergoing colonoscopy: a multicentered, randomized, positive-controlled, phase III clinical trial. Am J Transl Res. 2020;12(8):4594–603.
Toyota Y, Kondo T, Oshita K, et al. Remimazolam-based anesthesia with flumazenil allows faster emergence than propofol-based anesthesia in older patients undergoing spinal surgery: a randomized controlled trial. Medicine (Baltimore). 2023;102(46):e36081.
Zhang J, Zhang J, Wang Y, et al. Effect of remimazolam vs propofol on emergence from general anesthesia in patients undergoing cerebral endovascular procedures: a randomized controlled, non-inferiority trial. J Clin Anesth. 2023;5(93):111356.
Acknowledgements
We would like to thank the participants of this study.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Funding
No funding or sponsorship was received for this study or publication of this article.
Author information
Authors and Affiliations
Contributions
The authors Lidan Huang, Hong Liu, Xue Zou, Jiawang Ding, and Song Tao were responsible for the conception and design, as well as drafting the manuscript at its initial stage and revising it critically for important intellectual content. Lidan Huang and Hong Liu wrote the final draft. Approval was given by all the authors for submission and publication of this manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors Lidan Huang, Hong Liu, Xue Zou, Jiawang Ding, and Song Tao declare that they have no potential conflicts of interest.
Ethical Approval
This meta-analysis is based on previously published studies and does not contain any studies with human participants or animals performed by any of the authors. Therefore, no ethical approval was required for this meta-analysis.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, L., Liu, H., Zou, X. et al. Adverse Drug Events Observed with the Newly Approved Remimazolam in Comparison to Propofol for General Anesthesia in Patients Undergoing Surgery: A Meta-analysis. Adv Ther 41, 1896–1910 (2024). https://doi.org/10.1007/s12325-024-02820-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-024-02820-1