Abstract
Extracellular vesicles (EVs) are nanoscale vesicles derived from cells that mediate intercellular communication by transporting bioactive molecules. They play significant roles in various physiological and pathological conditions. EVs hold great potential as novel biomarkers of diseases, therapeutic agents, and drug delivery vehicles. Furthermore, EVs as novel drug delivery vehicles have demonstrated significant advantages in preclinical settings. In this review, we discussed the biogenesis and characteristics of EVs and their functions in cancer. We summarize the therapeutic applications of EVs as a natural delivery vehicles in cancer therapy. We highlight the existing challenges, illuminate vital questions, and propose recommendations to effectively address them effectively.
Similar content being viewed by others
Introduction
In the past decade, there has been rapid growth in our understanding of the types, characteristics, and physiological and pathological roles of extracellular vesicles (EVs). The International Society for Extracellular Vesicles (ISEV) recognizes EVs as general term for particles naturally released from cells, which are enveloped by a lipid bilayer and cannot replicate, and do not contain functional nucleus [1]. EVs can be broadly classified into three main subpopulations based on their biogenesis and size, including apoptotic bodies, microvesicles, exosomes and others [2,3,4]. EVs can be characterized based on their size and homogeneity (Table 1). Apoptotic bodies (1000—5000 nm) are the largest subpopulation of EVs released by cells undergoing apoptosis [5]. Microvesicles (also called ectosomes, 100 –1000 nm) are formed through the outward budding and fission of the plasma membrane and released into the extracellular space [6]. Exosome (40—160 nm) is the product of fusion between multivesicular bodies containing intraluminal vesicles and plasma membranes [7,8,9,10]. However, there is still a lack of confidence in identifying EV subtypes for a variety of reasons. In 2018, ISEV recommends using operational terminology to describe EVs subtypes when subcellular origin markers cannot be reliably established. This involves categorizing them based on their physical properties, biochemical composition, and describing their conditions or cellular origins. It is advised to avoid using historically vague, contradictory, and uncertainly generated terms like “exosome” and “microvesicle” [11].
EVs are secreted by both prokaryotic and eukaryotic cells. Most mammalian cell types, including neurons, endothelial cells, mesenchymal stem cells, and epithelial cells, have been shown to release EVs [11,12,13,14,15,16]. Furthermore, EVs have been detected in various biological fluids, including blood, urine, ascites, synovial fluid, and saliva [17,18,19]. It is noteworthy that certain subpopulations of EVs and particles have slightly different size ranges, biophysical characteristics, morphological characteristics, and protein marker expression, as well as the limitations of commonly used EVs purification methods, many studies have relied on analyzing mixed EVs populations consisting of exosomes, microvesicles, and non-membranous particles.
EVs play a critical role in intercellular communication and regulation, orchestrating diverse biological processes [20,21,22,23,24]. Internally, they harbor a repertoire of bioactive molecules including a blend of RNA, double-stranded DNA, proteins, lipids, glycoconjugates, and metabolites [25, 26]. This protective feature enables EVs to undergo long-range transfer across different tissues through blood circulation. Although the mechanisms underlying cargo sorting into EVs are not yet fully understood, it is evident that this process is highly selective [27]. Upon their release into the extracellular milieu, extracellular vesicles engage in intricate interactions with recipient cells, exerting profound influences on their functional capacities and physiological states. The levels of many cargo molecules in EVs do not directly correlate with their intracellular levels, indicating a regulated and specific sorting mechanism. Their multifaceted involvement spans both normal physiological dynamics and disease pathogenesis, endowing them with significant potential as biomarkers and promising tools for drug delivery and therapeutic interventions [28, 29].
Nowadays, cancer poses a formidable global threat to human life, driving researchers to seek novel effective anti-cancer therapeutics. In recent years, numerous drugs have showcased remarkable anti-cancer efficacy. However, the utilization of certain drugs is often hindered by intrinsic attributes such as limited target selectivity, short half-life in circulation, and unfavorable treatment-related side effects. EVs have emerged as highly advantageous vehicles for cancer treatment and drug delivery. In this review, we provide an in-depth exploration of the advancements in utilizing EVs for the translational study of cancer treatment, including their potential as innate delivery vehicles for anti-cancer drugs. Additionally, we discuss novel challenges within the realm of EV-based drug-loading strategies.
Biogenesis of Extracellular Vesicle
EVs represent a heterogeneous population of membrane vesicles generated via diverse mechanisms. A growing body of evidence suggests that EVs play a key role in normal physiology as well as disease pathology. EVs are involved in the removal of unnecessary cellular components, mediating specific intercellular information exchange and communication, and activating intracellular signaling pathways [30]. By participating in cell-to-cell communication, EVs serve as homeostatic regulators in maintaining physiological and dynamic balance, as well as in the development and progression of diseases [31]. Moreover, EVs are also involved in various systemic pathological conditions, including blood coagulation, immune responses, infectious diseases, metabolic diseases, central nervous system-related diseases, musculoskeletal diseases, and cancer [32,33,34]. Discriminating EVs subtypes, defining their physiological relevance, regulating their production under pathological conditions, and harnessing them as therapeutic tools hold significant importance in the field.
Nowadays, the mechanisms underlying the generation, release, and uptake of EVs are relatively well understood. The generation of EVs typically relies on fundamental steps shared by various intracellular trafficking processes occurring in cellular compartments. These steps involve the formation of membrane microdomain enriched in specific cargoes and then budding and fission of the microdomain to generate vesicle. Several studies have demonstrated that various types of machinery regulate these steps [14, 15, 35,36,37,38,39], such as Rab family proteins, the ESCRT machinery, the syntenin-Alix pathway, tetraspanins, the cytoskeleton, and lipids. These sorting mechanisms selectively enrich specific cargoes into EVs, and their depletion can hinder the generation of specific EV subpopulations. The intracellular trafficking of recruiting cargoes between the plasma membrane and endosomes is a crucial regulatory factor governing the biogenesis of ectosomes and exosomes.
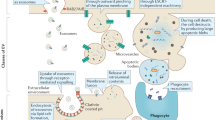
Extracellular factors including Microenvironmental pH, hypoxia, radiation, adhesion, and increased external pressure promote the release of EVs [34, 40,41,42]. EVs can influence adjacent and distant cells through autocrine or paracrine mechanisms [43]. Moreover, EVs can enter recipient cells through three main pathways: direct fusion with the cell membrane, receptor-ligand interaction, and fusion with the inner membrane after endocytosis [39]. The complex relationships between the generation, classification, and cargo of EVs still require continued investigation, as they can potentially give rise to distinct subpopulations of EVs, ultimately leading to potential physiological functions.
Intercellular Communication of Extracellular Vesicle
EVs employ various mechanisms to mediate intercellular communication. The similarity of lipid membrane characteristics between EVs and the plasma membrane unveils a mechanism akin to phagocytic engulfment [44, 45], allowing for direct fusion with the recipient cell’s plasma membrane and facilitating the exchange of transmembrane proteins and lipids [46]. Moreover, EV uptake has been observed to transpire through a diverse array of well-established endocytic pathways and membrane fusion events, encompassing both grid protein-dependent and grid protein-independent mechanisms [47, 48]. The intrinsic heterogeneity exhibited by distinct subpopulations of EVs circulating in the organism poses a formidable challenge in unraveling the intricate physiological functional network governed by EVs-mediated intercellular communication. The precise capacity of EVs to selectively target specific cells and tissues remains an area of ongoing investigation. More and more studies are utilizing EV uptake mechanisms and cargo-loading strategies to develop novel therapeutic approaches.
Sources of Drug Delivery
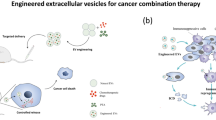
Drugs are limited in cancer treatment due to their poor targeting ability, poor bioavailability, unspecific cytotoxicity, and consequent systemic side effects [49]. There are many studies have demonstrated that EVs can serve as drug delivery vehicles, increasing drug accumulation in tumor tissues, extending blood circulation time, reducing systemic toxicity, and improving treatment efficacy (Fig. 1) [50,51,52,53,54,55,56].
Nanotechnology Engineering
Recently, many studies have been utilizing nanotechnology engineering to enhance the efficacy of drug delivery. Lipid nanoparticles (LNPs) have garnered attention as nanocarriers for pharmaceuticals due to their size and ability to transport small molecules. LNP was approved by the FDA for encapsulating small molecules like doxorubicin and daunorubicin in order to treat cancer [57,58,59]. Cholesterol-conjugate loaded liposomes exhibited higher in vitro cytotoxicity compared to the use of 5-fluorouracil alone in the treatment of hepatocellular carcinoma [60]. Besides that, Niu et al. demonstrate a distinct design by patching doxorubicin-loaded heparin-based nanoparticles (DNs) onto the surface of natural grapefruit EVs, to fabricate biomimetic EV-DNs, achieving efficient drug delivery and thus significantly enhancing anti-glioma efficacy [61]. Although nanotechnology has various advantages in drug delivery vehicles, they are also accompanied by several drawbacks, such as high production costs and low drug loading capacity [62, 63]. These limitations have restricted further applications of nanotechnology.
Extracellular Vesicles
Extracellular Vesicles Derived from Cells
Nowadays, EVs have advantages such as low toxicity, high biocompatibility, low immunogenicity, and inherent targeting ability, making them suitable as drug delivery carriers for cancer treatment. EVs that loaded with drugs derived from various cell sources, such as macrophages, dendritic cells, and red blood cells, have shown better anti-cancer effects [53, 64]. Therefore, selecting the most appropriate cell type for isolating EVs is crucial for drug delivery research.
Extracellular Vesicles Derived from Mesenchymal Stem Cells
Mesenchymal stem cells are widely used to produce EVs (MSC-EVs) due to their regenerative and immunomodulatory effects [65,66,67,68]. Pascucci et al. found that MSC-EVs containing paclitaxel can inhibit the proliferation of pancreatic cancer cells [54]. However, the application of mesenchymal stem cells in therapy has been limited due to their potential tumorigenicity.
Extracellular Vesicles Derived from Immune Cells
Another cell type that is widely used for EV isolation is immune cells, including macrophages, and dendritic cells [52, 64, 69]. Macrophage-derived exosomes loaded with paclitaxel have shown inhibitory effects in lung cancer metastasis mouse models. Furthermore, modifying these EVs with aminoethyl anhydride-polyethylene glycol carrier moieties is a common approach to reduce the immunogenicity of nanoscale particles. This modification improves the circulation time of EVs and enhances their lung-targeting capabilities for lung metastasis [51, 52]. EVs that derived from immature dendritic cells were engineered to express Lamp2b and fused with av integrin-specific iRGD targeting peptide. These modified exosomes were then loaded with doxorubicin using electroporation [53]. The presence of iRGD peptide enhanced the in vivo targeting of doxorubicin-loaded exosomes to MDA-MB-231 tumor cells, thereby enhancing their anti-tumor effects in established tumors. These findings suggest that immune cell-derived exosomes can be modified through various methods to enhance their targeting capabilities, making them more effective delivery vehicles.
Extracellular Vesicles Derived from Tumor Cells
Interestingly, EVs that are extracted from tumor cells are also utilized as a drug delivery vehicles. Due to the influence of the tumor microenvironment, cancer cell-derived EVs are generated abundantly and possess specific homing abilities [68, 70]. These EVs derived from cancer cells express tumor-specific antigens on their membranes, which may aid in generating anti-tumor immune responses in mouse models [71]. Notable outcomes were achieved in an investigation that focused on a mouse model of lung cancer. Compared with traditional chemotherapy, the administration of chemotherapy-loaded EVs resulted in a notable reduction in tumor burden and a prolongation of the survival period [70]. Moreover, a study showed that the utilization of platinum-loaded EVs derived from A549 cell line to treat three advanced lung cancer patients who exhibited resistance to platinum-based therapy. The results revealed a significant decrease in the number of tumor cells, while free platinum treatment failed to demonstrate beneficial effects for the patients. These collective findings underscore the considerable potential of cancer cell-derived EVs as a valuable approach in the treatment of lung cancer [70, 72]. Besides that, after loading with paclitaxel or gemcitabine, EVs derived from pancreatic cancer cells are employed for the treatment of pancreatic cancer. In vivo, pancreatic cell derived-EVs containing gemcitabine concentrate at the tumor site, leading to the inhibition of tumor growth while minimizing damage to normal tissues, significantly prolonging the survival rate of mice. This observation can be attributed to the potential tropism of EVs towards the tumor microenvironment, making EVs a competitive drug delivery vehicles for targeted chemotherapy [73]. However, cancer cell-derived EVs may promote tumor growth and metastasis by activating pathological pathways and exerting immunosuppressive effects. Subsequent research aimed to counteract this immunosuppressive response and found that when cancer cell-derived EVs were mixed with sufficient immune-stimulating adjuvants, the immunosuppressive effects were inhibited, thereby promoting anti-tumor responses [71].
Extracellular Vesicles Derived from Commonly Used Cellular Lines
In addition to the mentioned cells, there are other common cell lines that frequently used as sources of EVs for drug delivery, including Human Embryonic Kidney 293 cells (HEK293T), Chinese Hamster Ovary cells (CHO), and HeLa cells line. Among these, the HEK293T cell line is one of the most extensively utilized cell lines for EV-mediated drug delivery and has demonstrated potential value in industrial applications. While EVs derived from HEK293T cells can enrich certain cancer-related signaling molecules, they possess a higher transfection efficiency and are easily loaded with small therapeutic RNA molecules [74].
Extracellular Vesicles Derived from Body Fluids
Most body fluids contain EVs, including blood, urine, saliva, cerebrospinal fluid, ascites, and amniotic fluid, making these molecules useful for clinical diagnosis [75]. Among them, EVs derived from blood sources have been extensively studied as drug delivery vehicles [76, 77]. Usman et al. found that utilizing EVs derived from red blood cells can achieve highly efficient delivery of RNA, including small interfering RNA, antisense oligonucleotides, and CRISPR-associated protein 9 genome-editing guide RNA [77]. Zhang et al. showed that miR-155-loaded EVs from red blood cells exhibited excellent protective effects in acute liver failure, while EVs loaded with DOX or sorafenib showed significant therapeutic effects on in situ hepatocellular carcinoma without systemic toxicity [78]. In a clinical trial, Dai et al. showed EVs from ascites of colon cancer patients and combined them with granulocyte–macrophage colony-stimulating factor for immunotherapy. Combining these therapy strategy resulted in enhanced cytotoxic T lymphocyte responses specific to tumor antigens [79].
Extracellular Vesicles Derived from Other Sources
Milk
Recent research indicates that bovine milk-derived EVs have the potential value to serve as a drug delivery vehicles [80]. Bovine milk contains a higher abundance of separable EVs, and when injected into mouse models, bovine milk-derived EVs did not exhibit cytotoxicity or allergic reactions [81]. Importantly, bovine milk-derived EVs demonstrate high stability and low immunogenicity in the intestinal environment, making them potential carriers for chemotherapy drugs [82,83,84]. Furthermore, some studies have shown that bovine milk-derived EVs loaded with a range of drugs such as paclitaxel, docetaxel, and doxorubicin significantly improved the bioavailability and efficacy of these drugs in both in vitro and in vivo cancer models [85,86,87]. Therefore, researchers propose that drug-loaded milk EVs hold promise as a biocompatible, safe, effective, and cost-efficient targeted drug delivery mode for cancer treatment.
Plant
It has been discovered that plant-derived EVs for cancer treatment can be extracted from edible plants such as ginger, lemon, and grapefruit, which are non-toxic and are capable of being produced in large quantities [55, 88,89,90,91]. EVs derived from plants have established oral tolerance due to interactions with the intestinal immune system and food that we eat on a daily basis [92]. It has been reported that plant-derived EVs exhibit high resistance to gastric proteolytic enzymes and intestinal pancreas and bile extracts, making oral administration the most reasonable route, especially when targeting tumors located in the gastrointestinal tract [93]. Zhang et al. found that folate-modified and doxorubicin-loaded ginger-derived exosome-mimetic nanovesicles demonstrated excellent tissue compatibility and anti-tumor effects in colorectal cancer [55]. A study also demonstrated that grapefruit-derived nano-carriers effectively delivered various therapeutic drugs and enhanced their homing ability to inflammatory tumor tissues [90].
Bacteria
It has been demonstrated that bacterial outer membrane vesicles can carry immune stimulants and inducing the corresponding immune response can be used to treat tumors. Bacterial outer membrane vesicles derived from attenuated pneumococcal strains have been shown to effectively transport doxorubicin into A549 cells and interact with macrophages to activate the immune system, thereby enhancing the anti-tumor effect of doxorubicin [56]. It is worth noting that bacterial protoplasts, which lack toxic outer wall components, have become the preferred source of EVs. Kim et al. used Bacterial outer membrane vesicles derived from bacteria overexpressing epidermal growth factor to demonstrate the efficient delivery of doxorubicin and idarubicin to tumor cells in vitro and in vivo, resulting in the inhibition of tumor growth in mice [94].
EVs as Drug Delivery Systems in Cancer Treatment
EVs have shown successful application in cancer therapy, facilitating the delivery of various therapeutic cargoes, including chemotherapy drugs, nucleic acids, and proteins [95, 96]. EVs are non-replicative and non-transformative, leading to fewer adverse reactions [97]. Moreover, the biodistribution of EVs varies based on the cell source, route of administration, and targeting methods [98, 99]. Various strategies of EVs have been investigated for tumor therapy (Table 2), and several ongoing clinical trials are investigating their potential as therapeutic delivery vehicles (Table 3) [72, 100,101,102,103,104].
Chemotherapy Drugs
Doxorubicin and paclitaxel are commonly used carrier drugs for EVs drug delivery. However, their clinical application is limited due to dose-limiting toxicity and poor water solubility [105]. Additionally, their faces challenges in treating brain metastases due to its limited ability to cross the blood–brain barrier [106, 107]. And some studies have indicated that EVs can reduce the cardiac toxicity of doxorubicin by limiting its penetration into cardiac endothelial cells and accumulation in the heart [108, 109]. Furthermore, paclitaxel-loaded EVs have shown promising results in targeting and treating lung, breast, and pancreatic cancers [54, 64]. Besides that, Salarpour et al. demonstrated that EVs derived from U-87 MG cells can deliver paclitaxel across the blood–brain barrier, thereby improving the therapeutic effect of glioblastoma [110]. Barani et al. employed the film hydration method to develop novel niosomes containing cholesterol, Span, Tween, and gemcitabine. The efficacy of the niosomes was evaluated in vitro and in vivo. The developed niosomes show great potential as carriers for specific chemotherapeutic agents [111].
Protein
Protein-based therapy is emerging as a promising approach for cancer treatment, addressing concerns related to maintaining activity and extending protein half-life. Various studies have explored the modification of EVs with specific proteins to enhance targeting abilities towards breast cancer cells in both in vitro and in vivo settings [112]. Koh et al. found that EVs with overexpressing SIRPα enhanced the phagocytosis of macrophages and effectively inhibited tumor growth in tumor-bearing mice [113]. Similarly, Hong et al. showed that EVs expressing PH20 hyaluronidase on the their surface can effectively degrade HA and induce DC activation through the TLR4 pathway, thereby inhibiting tumor growth [114]. Additionally, MSC-derived EVs with TRAIL overexpression such as cancer cell apoptosis in a dose-dependent manner [115].
Recently, a study has reported an innovative immunotherapy that uses EVs as targeted delivery vehicles for antibodies to breast cancer cells. By genetic modifications, exosomes are engineered to express two monoclonal antibodies, leading to a potent anti-tumor immune response by influencing T lymphocytes and breast cancer cells [116, 117]. Furthermore, researchers discovered that T-DM2, a promising drug for HER2-positive cancer, can be loaded into exosomes derived from HER2-positive cancer cells and delivered to other cancer cells via exosomes, resulting in apoptosis [118].
Nucleic Acids
Currently, nucleic acids, such as miRNAs and siRNAs, hold promise as potential therapeutic strategies in cancer treatment [119]. Its clinical application is limited by its short half-life, immunogenicity, inability to penetrate physical barriers, and off-target effects. To address these challenges, EVs have gained significant attention as nucleic acid delivery vehicles, leveraging their unique properties to overcome these obstacles [120].
MicroRNAs
miRNAs, short non-coding RNAs involved in various cellular processes, play a significant part in cancer development and progression [121]. Some studies have showed that EVs derived from MSCs with deliver miRNAs to treat liver cancer by promoting apoptosis, and enhancing chemotherapy sensitivity in many cancers including breast, ovarian, pancreatic, and osteosarcoma [122,123,124,125,126]. Conversely, the delivery of miRNA inhibitors can also achieve anti-tumor effects, particularly when targeting miRNAs with tumor growth-promoting properties [127]. Enhancing the loading efficiency of miRNAs into EVs is an active area of research. For instance, Li et al. successfully enriched miR-155 in EVs by fusing the exosome surface marker protein CD9 with HuR, as demonstrated in their study [128]. Lang et al. found that EVs loaded with miR-124a significantly increased the median survival rate of glioblastoma mice [129]. These findings hold promise in advancing miRNA-based therapies using EVs as drug delivery vehicles.
Small Interfering RNAs
Delivering siRNA to target cells for gene silencing is a crucial gene therapy approach. Various studies have demonstrated the potential of EVs in efficiently loading and delivering siRNAs to tumor cells [130, 131]. To enhance delivery accuracy and efficiency, Pi and Zheng et al. modified EVs with RNA aptamers or folic acid in order to improve the efficiency and accuracy of siRNA delivery and uptake by prostate and breast cancer cells [132, 133]. Moreover, Kamerkar et al. targeted the delivery of siRNA-loaded engineered exosomes to pancreatic cancer cells, which showed significant tumor suppression both in vitro and in vivo, and this technology has been approved and entered clinical trials [101, 134].
Encapsulation of Therapeutics
There are two main strategies for loading drugs into EVs: pre-loading and post-loading. Pre-loading involves loading the drug into the parent cells before EVs are isolated, resulting in EVs that carry the loaded drug. Post-loading, on the other hand, is the process of directly loading drugs into EVs after they have been separated, using passive or active methods.
Pre-loading
Pre-loading facilitates the consistent and straightforward production of drug loaded EVs while preserving membrane integrity. Two prevalent methods of pre-loading include co-incubation and transfection.
Under specific conditions, the drugs are co-cultured with parent cells, facilitating their spontaneous absorption by cells through interaction with the lipid bilayer. The co-incubation method is employed to load various chemotherapeutic drugs, especially lipophilic ones like DOX and PTX [108, 135, 136]. Co-incubation is a relatively straightforward technique, but it is associated with lower loading efficiency. Additionally, its effectiveness is notably influenced by many factors inckuding drug properties, drug concentration gradients, and the specific type of parent cells [137]. Recent studies have indicated that Ultraviolet Induction cells can more efficiently load co-incubated drugs into EVs [72]. Furthermore, parent cells can overexpress therapeutic cargo and encapsulate cargo in EVs through cell transfection. ExoIL-12 loaded with PTGFRN is the world’s first engineered exosome candidate drug to enter clinical trials [138]. Cell transfection offers the advantages of high repeatability and simplicity. However, its drawbacks include low transfection efficiency and high dependence on cell viability [139, 140].
Post-loading
Post-loading involves directly loading drugs into isolated EVs. Compared to pre-loading, this strategy is more customizable and minimizes interference from other substances. Currently, post-loading is mainly divided into passive loading and active loading methods.
When high concentrations of drugs are co-incubated with EVs, they passively diffuse into the lumens of EVs through interactions with the lipid bilayer. Passive loading method has been widely applied in cancer treatment [80]. However, the primary limitations of passive loading application are its low loading efficiency and limited selectivity.
Some cargo cannot passively diffuse through the EV membranes, physical induction or chemical induction are required to temporarily affect the permeability of EV membranes to enable the cargo entry. Physical induction typically involves instantaneous disruption of EV membranes by external forces. And chemical induction utilizes transfection agents to facilitate cargo loading without damaging EV membranes [141, 142]. Fuhrmann et al. found that saponin significantly increased the loading efficiency of porphyrins (derived from MDA-MB-231) into EVs [141]. Additionally, Zhang et al. developed an improved method for transfecting miRNAs into EVs using calcium chloride [143]. In comparison to electroporation, this approach exhibits comparable transfection efficiency with the added benefits of being simpler and more stable.
Isolation and Purification of EVs
EVs are isolated from large volumes of conditioned media in industrial manufacturing. The primary contaminants include various vesicles, EV aggregates, cellular debris and organelles, DNA, cell necrosis products, free proteins, and protein aggregates. It is crucial to avoid microbial contaminants such as bacteria, fungi, and mycoplasma. Besides that, EVs are isolated from small quantities of highly complex biological samples in scientific research. These samples demonstrate notable diversity in their composition. The main contaminants are non-EV nanoparticles, primarily lipoproteins, ribonucleoproteins, and protein aggregates. Common indicators used to assess EVs purity include the particle-to-protein ratio and the protein-to-lipid ratio [144]. In summary, there are significant differences in the requirements and challenges for isolating EVs between industrial manufacturing and scientific research, primarily due to variations in sample volume, sample complexity, and the nature of contaminants. Customized isolation techniques are employed to ensure the purity and integrity of isolated EVs for their intended applications in these contexts. Currently, various methods have been developed to specifically isolate different subsets of EVs, such as centrifugation-based methods, precipitation-based methods, and others.
Ultracentrifugation (UC) is considered the gold standard of EV isolation in entrifugation-based techniques. UC employs centrifugal force to pellet EV particles, effectively separating them from major contaminants such as proteins and small molecular compounds, which remain in the supernatant. However, UC also has several drawbacks, including low EV yield, partial vesicle damage and aggregation, co-pelleting of non-exosomal components, and the formation of aggregated contaminants, primarily protein aggregates [145].
In precipitation-based methods, EVs can be reversibly aggregated after treatment with various chemical reagents, such as polyethylene glycol, precipitation with cationic polymers, and PROSPR approach [146, 147]. EVs can be separated through medium-speed centrifugation. These techniques are straightforward, relatively cost-effective, and do not require complex equipment. However, as precipitation methods lack selectivity towards EVs, the purity of the isolated EVs tends to be relatively low.
Other methods for isolating EVs include Size-based methods (ultrafiltration, tangential flow filtration, size-exclusion chromatography, asymmetrical flow-field-flow fractionation), chromatography methods (anion-exchange chromatography, hydrophobic chromatography), and affinity-based isolation methods. Table 4 summarizes the comparison of EV isolation methods.
Engineering of EVs
Surface engineering is a significant milestone in the field of EVs formulations. Assessing the effectiveness of surface engineering is crucial for evaluating the therapeutic efficacy of EV formulations, especially in the context of precision medicine. Genetic methods involves modifying the cells responsible for EV production to express a fused genetic construct, which includes a fundamental EV protein linked to a targeting moiety associated with the targeting molecule, such as Lamp2b, CD9, CD63, and PTGFRN [148,149,150]. For example, HER2 is significantly overexpression in various tumors such as breast cancer, ovarian cancer, and gastric cancer. DARPins are a class of recombinant binding proteins that can bind to HER2 with high specificity. A study using lentiviral transduction of donor cells, successfully prepared hybrid EVs that expressing Lamp2b-DARPin G3 [130]. This achievement enables precise targeted delivery of TPD52 siRNA to HER2-positive breast cancer cells. Furthermore, PDGFR is a single-chain transmembrane glycoprotein that commonly employed to anchor fusion proteins. the PDGFR-GE11 peptide can specifically bind to EGFR, thereby targeting tumors with EGFR expression [151].
The fusion of the target molecule with the membrane protein to create a chimeric protein has the potential to modify the structure, integrity, and functionality of the native anchoring proteins on the surface of EVs. Many studies have indicated that the surface of EVs is enriched with negatively charged phosphatidylserines [152, 153], and lactadherin’s C1C2 domains specifically bind to phosphatidylserines. Kooijmans et al. utilized genetic engineering to equip EVs with a lactadherin-streptavidin fusion protein. They created C1C2-anti-EGFR recombinant fusion proteins, enabling precise targeting of tumor cells [154]. This engineering strategy holds promising potential as an effective system for cancer therapy. Furthermore, glycosylphosphatidylinositol-anchored proteins are also abundant on the surface of EVs. Various functional ligands, such as nanobodies, reporter proteins, and immune-stimulatory molecules, can bind to glycosylphosphatidylinositol and be expressed on the surface of EVs [155]. CD47 is a transmembrane protein that enriched on the surface of EVs, with its N-terminus positioned on the external surface of the EV. A study fused two different peptides (CDX or CREKA peptides) to the N-terminus of CD47, enabling specific targeting of U87 and GL261 glioblastoma cells, thus achieving a targeted effect on glioblastoma [156]. Besides that, a study engineered T cells to release EVs that carrying chimeric antigen receptors (CARs) with single-chain variable fragments of the antibodies cetuximab or trastuzumab [157]. These CAR-EVs demonstrated significant anti-tumor effects and exhibited low toxicity.
The Challenges of Extracellular Vesicles as Drug Delivery
The main challenges of drug delivery include off-target toxicity within target tissues, rapid clearance, low bioavailability. There are many synthetic delivery vehicles have been developed. Liposomes are the most widely clinically approved carriers on the clinical. The advantages of EVs are clearly superior to liposomes in the field of drug delivery vehicles. EVs, originating from the organism itself, with low immunogenicity, thereby ensuring excellent tolerance and safety. Additionally, EVs have the capability to traverse the blood–brain barrier and enter the bloodstream within the brain [158]. This allows for the rapid delivery of drugs to lesions within the brain, offering a potential treatment option for intracranial diseases. EVs can undergo artificial surface modification to express specific molecules, thereby enhancing their targeting capabilities. Therefore, many studies have demonstrated that EVs as drug delivery vehicles are an ideal strategy for treating various types of cancer. However, there are several challenges that require clarification [159].
First, Optimal methods for loading drugs into EVs and quantification need a thorough evaluation. Some methods, like ultrasound, may be more efficient than others but could compromise the structural integrity of EVs [110]. Loading external chemical or biomolecules into EVs is a significant challenge. The composition of EVs introduces another issue, as it may lead to the transfer of undesirable content derived from the parent cells, potentially triggering immunogenic or oncogenic responses [160]. Rapid clearance of EVs and excessive immune system activation after administration are potential drawbacks of using EVs as drug delivery tools, choosing of administration route critical [159].
Secondly, EVs are inherently highly heterogeneous, and their functions and effects may vary depending on the carried cargo [161]. Therefore, careful selection of the appropriate EV subtype for specific drug delivery is essential [162]. EVs reflect their source cells and may carry substances that could unexpectedly promote cancer development during cancer treatment [163]. Additionally, EVs may exhibit significant differences in their biological distribution and half-life based on their cell of origin [164]. In the context of utilizing EVs as drug delivery systems, it’s crucial to assess potential interactions between the loaded exogenous cargo and endogenous cargo. This evaluation is pivotal in determining whether off-target effects [138]. Thus, comprehensive preclinical evaluations on cells, tissues, and animal models are crucial before considering their application.
Third, Other disadvantages of using EVs as drug delivery vehicles include challenges in their production, purification processes, and a lack of reproducibility in drug loading techniques [165]. The commercialization of EVs also encounters challenges including technical, economic, and regulatory issues [166].
Fourth, interdisciplinary collaboration plays a crucial role in the development of therapeutic strategies of EVs. Collaboration between researchers from fields such as cell biology, engineering, and immunology is instrumental in advancing more effective EV-based therapeutic approaches. It is critical to understanding of the generation and release mechanisms of EVs. The classification of EVs remains unclear. Recently discovered like exomeres and suprameres demand further investigation. Moreover, it is crucial to refine production processes or innovate new isolation techniques to augment both the yield and purity of EVs. This advancement will significantly broaden the spectrum of drug-loading options for EVs. Additionally, it is essential to investigation the interaction between EVs and the human immune system. This involves exploring strategies for surface engineering of EVs to further improve their targeted drug delivery capabilities while reducing their immunogenicity.
In summary, EVs as drug delivery requires utmost caution. One of the main advantages of employing EV-based drug delivery is their ability to reduce cytotoxicity. Therefore, drug-loaded EVs should demonstrate superior efficacy, tolerability, and safety in cancer treatment.
Discussion
Nearly all cells release EVs as heterogeneous lipid nanoparticles. They participate in both proximal and distal cell communication [167]. EVs play a crucial role in regulating various physiological and pathophysiological processes. The majority of cell- and animal-based experimental evidence supports the significant role of EVs in almost all aspects of cancer, spanning from cancer initiation and progression to the development of paraneoplastic syndromes [168]. While several drugs have been developed to inhibit the biogenesis or secretion of tumor-derived EVs, they have not yet received clinical approval. Further research involving relevant preclinical human cancer models and clinical trials targeting the depletion of tumor EVs may contribute to the development of novel anticancer therapies.
EVs are a group of small lipid-based nanoparticles decorated with complex surface proteins and lipids, facilitating homing to specific tissues. The composition and biogenesis of EVs directly depend on their sources. These characteristics and their natural advantage makes EVs a useful vehicle for delivering therapeutic payloads due to their advantages as nanocarriers. Unlike artificially engineered nanoparticles, EVs occur naturally and, therefore, do not elicit inflammatory reactions. The tissue-homing ability of EVs allows them to reach distant target sites. This novel therapeutic strategy is currently under preclinical investigation for various cancer types, showing promising results with minimal side effects. In recent years, new approaches have been continuously developed to improve these nanovesicles, such as the development of superparamagnetic nanoparticles based on EVs [169,170,171]. However, a number of challenges must be overcome before drug-loaded EVs for cancer treatment can be commercialized. Therefore, further research and exploration of new strategies are required to enhance the production and drug-loading efficiency of EVs.
Availability of Data and Materials
Not applicable.
References
Lötvall J, et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. J Extracell Vesicles. 2014;3:26913.
O’Brien K, et al. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat Rev Mol Cell Biol. 2020;21(10):585–606.
Nicolás-Ávila JA, et al. A network of macrophages supports mitochondrial homeostasis in the heart. Cell. 2020;183(1):94–109.e23.
Ashley J, et al. Retrovirus-like Gag protein Arc1 binds RNA and traffics across synaptic boutons. Cell. 2018;172(1–2):262–274.e11.
Zhou M, et al. Apoptotic bodies for advanced drug delivery and therapy. J Control Release. 2022;351:394–406.
Cocucci E, Meldolesi J. Ectosomes and exosomes: shedding the confusion between extracellular vesicles. Trends Cell Biol. 2015;25(6):364–72.
Colombo M, Raposo G, Théry C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol. 2014;30:255–89.
Hoshino A, et al. Extracellular vesicle and particle biomarkers define multiple human cancers. Cell. 2020;182(4):1044–1061.e18.
Zhang H, et al. Identification of distinct nanoparticles and subsets of extracellular vesicles by asymmetric flow field-flow fractionation. Nat Cell Biol. 2018;20(3):332–43.
Zhang Q, et al. Supermeres are functional extracellular nanoparticles replete with disease biomarkers and therapeutic targets. Nat Cell Biol. 2021;23(12):1240–54.
Théry C, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750.
Mulcahy LA, Pink RC, Carter DR. Routes and mechanisms of extracellular vesicle uptake. J Extracell Vesicles. 2014;3:24641.
Russell AE, et al. Biological membranes in EV biogenesis, stability, uptake, and cargo transfer: an ISEV position paper arising from the ISEV membranes and EVs workshop. J Extracell Vesicles. 2019;8(1):1684862.
Baietti MF, et al. Syndecan-syntenin-ALIX regulates the biogenesis of exosomes. Nat Cell Biol. 2012;14(7):677–85.
Wehman AM, et al. The P4-ATPase TAT-5 inhibits the budding of extracellular vesicles in C. elegans embryos. Curr Biol. 2011;21(23):1951–9.
Mathieu M, et al. Specificities of exosome versus small ectosome secretion revealed by live intracellular tracking of CD63 and CD9. Nat Commun. 2021;12(1):4389.
van Niel G, D’Angelo G, Raposo G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol. 2018;19(4):213–28.
Liu XM, Ma L, Schekman R. Selective sorting of microRNAs into exosomes by phase-separated YBX1 condensates. Elife. 2021;10:e71982.
Leidal AM, et al. The LC3-conjugation machinery specifies the loading of RNA-binding proteins into extracellular vesicles. Nat Cell Biol. 2020;22(2):187–99.
El Andaloussi S, et al. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013;12(5):347–57.
Valadi H, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654–9.
Skog J, et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol. 2008;10(12):1470–6.
Villarroya-Beltri C, et al. Sorting it out: regulation of exosome loading. Semin Cancer Biol. 2014;28:3–13.
Pegtel DM, Gould SJ. Exosomes. Annu Rev Biochem. 2019;88:487–514.
Abhange K, et al. Small extracellular vesicles in cancer. Bioact Mater. 2021;6(11):3705–43.
Théry C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis and function. Nat Rev Immunol. 2002;2(8):569–79.
Garcia-Martin R, et al. MicroRNA sequence codes for small extracellular vesicle release and cellular retention. Nature. 2022;601(7893):446–51.
Maacha S, et al. Extracellular vesicles-mediated intercellular communication: roles in the tumor microenvironment and anti-cancer drug resistance. Mol Cancer. 2019;18(1):55.
Becker A, et al. Extracellular vesicles in cancer: cell-to-cell mediators of metastasis. Cancer Cell. 2016;30(6):836–48.
Kalluri R. The biology and function of exosomes in cancer. J Clin Invest. 2016;126(4):1208–15.
Stahl PD, Raposo G. Extracellular vesicles: exosomes and microvesicles, integrators of homeostasis. Physiology (Bethesda). 2019;34(3):169–77.
Pardo F, et al. Extracellular vesicles in obesity and diabetes mellitus. Mol Aspects Med. 2018;60:81–91.
Wang H, et al. Overexpression of MEG3 sensitizes colorectal cancer cells to oxaliplatin through regulation of miR-141/PDCD4 axis. Biomed Pharmacother. 2018;106:1607–15.
Wortzel I, et al. Exosome-mediated metastasis: communication from a distance. Dev Cell. 2019;49(3):347–60.
Colombo M, et al. Analysis of ESCRT functions in exosome biogenesis, composition and secretion highlights the heterogeneity of extracellular vesicles. J Cell Sci. 2013;126(Pt 24):5553–65.
van Niel G, et al. The tetraspanin CD63 regulates ESCRT-independent and -dependent endosomal sorting during melanogenesis. Dev Cell. 2011;21(4):708–21.
Thom SR, et al. Neutrophil microparticle production and inflammasome activation by hyperglycemia due to cytoskeletal instability. J Biol Chem. 2017;292(44):18312–24.
Trajkovic K, et al. Ceramide triggers budding of exosome vesicles into multivesicular endosomes. Science. 2008;319(5867):1244–7.
Zhang X, et al. Exosomes in cancer: small particle, big player. J Hematol Oncol. 2015;8:83.
Parolini I, et al. Microenvironmental pH is a key factor for exosome traffic in tumor cells. J Biol Chem. 2009;284(49):34211–22.
Zhang X, et al. Hypoxic BMSC-derived exosomal miRNAs promote metastasis of lung cancer cells via STAT3-induced EMT. Mol Cancer. 2019;18(1):40.
Martínez MC, Andriantsitohaina R. Extracellular vesicles in metabolic syndrome. Circ Res. 2017;120(10):1674–86.
Milman N, Ginini L, Gil Z. Exosomes and their role in tumorigenesis and anticancer drug resistance. Drug Resist Updat. 2019;45:1–12.
Feng D, et al. Cellular internalization of exosomes occurs through phagocytosis. Traffic. 2010;11(5):675–87.
Shiratsuchi A, et al. Phosphatidylserine-mediated phagocytosis of influenza A virus-infected cells by mouse peritoneal macrophages. J Virol. 2000;74(19):9240–4.
Prada I, et al. A new approach to follow a single extracellular vesicle-cell interaction using optical tweezers. Biotechniques. 2016;60(1):35–41.
Tian T, et al. Exosome uptake through clathrin-mediated endocytosis and macropinocytosis and mediating miR-21 delivery. J Biol Chem. 2014;289(32):22258–67.
Svensson KJ, et al. Exosome uptake depends on ERK1/2-heat shock protein 27 signaling and lipid Raft-mediated endocytosis negatively regulated by caveolin-1. J Biol Chem. 2013;288(24):17713–24.
Senapati S, et al. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct Target Ther. 2018;3:7.
Saari H, et al. Microvesicle- and exosome-mediated drug delivery enhances the cytotoxicity of Paclitaxel in autologous prostate cancer cells. J Control Release. 2015;220(Pt B):727–37.
Kim MS, et al. Engineering macrophage-derived exosomes for targeted paclitaxel delivery to pulmonary metastases: in vitro and in vivo evaluations. Nanomedicine. 2018;14(1):195–204.
Kim MS, et al. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomedicine. 2016;12(3):655–64.
Tian Y, et al. A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials. 2014;35(7):2383–90.
Pascucci L, et al. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: a new approach for drug delivery. J Control Release. 2014;192:262–70.
Zhang M, et al. Edible ginger-derived nano-lipids loaded with doxorubicin as a novel drug-delivery approach for colon cancer therapy. Mol Ther. 2016;24(10):1783–96.
Kuerban K, et al. Doxorubicin-loaded bacterial outer-membrane vesicles exert enhanced anti-tumor efficacy in non-small-cell lung cancer. Acta Pharm Sin B. 2020;10(8):1534–48.
Bangham AD, Standish MM, Watkins JC. Diffusion of univalent ions across the lamellae of swollen phospholipids. J Mol Biol. 1965;13(1):238–52.
Prescott LM. Doxil offers hope to KS sufferers. J Int Assoc Physicians AIDS Care. 1995;1(11):43–4.
Forssen EA, et al. Fluorescence imaging studies for the disposition of daunorubicin liposomes (DaunoXome) within tumor tissue. Cancer Res. 1996;56(9):2066–75.
Alanazi SA, et al. Cholesterol-conjugate as a new strategy to improve the cytotoxic effect of 5-fluorouracil on liver cancer: impact of liposomal composition. Curr Drug Deliv. 2020;17(10):898–910.
Niu W, et al. A biomimetic drug delivery system by integrating grapefruit extracellular vesicles and doxorubicin-loaded heparin-based nanoparticles for glioma therapy. Nano Lett. 2021;21(3):1484–92.
Akbarzadeh A, et al. Liposome: classification, preparation, and applications. Nanoscale Res Lett. 2013;8(1):102.
Ghasemiyeh P, Mohammadi-Samani S. Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: applications, advantages and disadvantages. Res Pharm Sci. 2018;13(4):288–303.
Haney MJ, et al. Macrophage-derived extracellular vesicles as drug delivery systems for triple negative breast cancer (TNBC) therapy. J Neuroimmune Pharmacol. 2020;15(3):487–500.
De Jong OG, et al. Extracellular vesicles: potential roles in regenerative medicine. Front Immunol. 2014;5:608.
Kou M, et al. Mesenchymal stem cell-derived extracellular vesicles for immunomodulation and regeneration: a next generation therapeutic tool? Cell Death Dis. 2022;13(7):580.
Dabrowska S, et al. Immunomodulatory and regenerative effects of mesenchymal stem cells and extracellular vesicles: therapeutic outlook for inflammatory and degenerative diseases. Front Immunol. 2020;11:591065.
Chulpanova DS, et al. Therapeutic prospects of extracellular vesicles in cancer treatment. Front Immunol. 2018;9:1534.
Kanchanapally R, et al. Drug-loaded exosomal preparations from different cell types exhibit distinctive loading capability, yield, and antitumor efficacies: a comparative analysis. Int J Nanomedicine. 2019;14:531–41.
Walker S, et al. Extracellular vesicle-based drug delivery systems for cancer treatment. Theranostics. 2019;9(26):8001–17.
Lener T, et al. Applying extracellular vesicles based therapeutics in clinical trials - an ISEV position paper. J Extracell Vesicles. 2015;4:30087.
Ma J, et al. Reversing drug resistance of soft tumor-repopulating cells by tumor cell-derived chemotherapeutic microparticles. Cell Res. 2016;26(6):713–27.
Li YJ, et al. Gemcitabine loaded autologous exosomes for effective and safe chemotherapy of pancreatic cancer. Acta Biomater. 2020;101:519–30.
Li J, et al. Identification and characterization of 293T cell-derived exosomes by profiling the protein, mRNA and MicroRNA components. PLoS ONE. 2016;11(9):e0163043.
Zhou B, et al. Application of exosomes as liquid biopsy in clinical diagnosis. Signal Transduct Target Ther. 2020;5(1):144.
Shi J, et al. Engineered red blood cells as carriers for systemic delivery of a wide array of functional probes. Proc Natl Acad Sci U S A. 2014;111(28):10131–6.
Usman WM, et al. Efficient RNA drug delivery using red blood cell extracellular vesicles. Nat Commun. 2018;9(1):2359.
Zhang G, et al. Extracellular vesicles: natural liver-accumulating drug delivery vehicles for the treatment of liver diseases. J Extracell Vesicles. 2020;10(2):e12030.
Dai S, et al. Phase I clinical trial of autologous ascites-derived exosomes combined with GM-CSF for colorectal cancer. Mol Ther. 2008;16(4):782–90.
Munagala R, et al. Bovine milk-derived exosomes for drug delivery. Cancer Lett. 2016;371(1):48–61.
Somiya M, Yoshioka Y, Ochiya T. Biocompatibility of highly purified bovine milk-derived extracellular vesicles. J Extracell Vesicles. 2018;7(1):1440132.
Sanwlani R, et al. Milk-derived extracellular vesicles in inter-organism, cross-species communication and drug delivery. Proteomes. 2020;8(2):11.
Samuel M, et al. Oral administration of bovine milk-derived extracellular vesicles induces senescence in the primary tumor but accelerates cancer metastasis. Nat Commun. 2021;12(1):3950.
Kandimalla R, et al. Milk exosomes: a biogenic nanocarrier for small molecules and macromolecules to combat cancer. Am J Reprod Immunol. 2021;85(2):e13349.
Agrawal AK, et al. Milk-derived exosomes for oral delivery of paclitaxel. Nanomedicine. 2017;13(5):1627–36.
Zhang Q, et al. Milk-exosome based pH/light sensitive drug system to enhance anticancer activity against oral squamous cell carcinoma. RSC Adv. 2020;10(47):28314–23.
Chen C, et al. Active cargo loading into extracellular vesicles: Highlights the heterogeneous encapsulation behaviour. J Extracell Vesicles. 2021;10(13):e12163.
Raimondo S, et al. Citrus limon-derived nanovesicles inhibit cancer cell proliferation and suppress CML xenograft growth by inducing TRAIL-mediated cell death. Oncotarget. 2015;6(23):19514–27.
Rahimi Ghiasi M, et al. Leucine-rich repeat-containing g-protein coupled receptor 5 gene overexpression of the rat small intestinal progenitor cells in response to orally administered grape exosome-like nanovesicles. Adv Biomed Res. 2018;7:125.
Wang Q, et al. Grapefruit-derived nanovectors use an activated leukocyte trafficking pathway to deliver therapeutic agents to inflammatory tumor sites. Cancer Res. 2015;75(12):2520–9.
Wang Q, et al. Delivery of therapeutic agents by nanoparticles made of grapefruit-derived lipids. Nat Commun. 2013;4:1867.
Pabst O, Mowat AM. Oral tolerance to food protein. Mucosal Immunol. 2012;5(3):232–9.
Wang B, et al. Targeted drug delivery to intestinal macrophages by bioactive nanovesicles released from grapefruit. Mol Ther. 2014;22(3):522–34.
Kim OY, et al. Bacterial protoplast-derived nanovesicles for tumor targeted delivery of chemotherapeutics. Biomaterials. 2017;113:68–79.
Elsharkasy OM, et al. Extracellular vesicles as drug delivery systems: why and how? Adv Drug Deliv Rev. 2020;159:332–43.
Nedeva C, Mathivanan S. Engineering extracellular vesicles for cancer therapy. Subcell Biochem. 2021;97:375–92.
Han Y, et al. Overview and update on methods for cargo loading into extracellular vesicles. Processes (Basel). 2021;9(2):356.
Wiklander OP, et al. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J Extracell Vesicles. 2015;4:26316.
Kang M, et al. Biodistribution of extracellular vesicles following administration into animals: a systematic review. J Extracell Vesicles. 2021;10(8):e12085.
Jang SC, et al. ExoSTING, an extracellular vesicle loaded with STING agonists, promotes tumor immune surveillance. Commun Biol. 2021;4(1):497.
Mendt M, et al. Generation and testing of clinical-grade exosomes for pancreatic cancer. JCI Insight. 2018;3(8):e99263.
Tang K, et al. Delivery of chemotherapeutic drugs in tumour cell-derived microparticles. Nat Commun. 2012;3:1282.
Guo M, et al. Autologous tumor cell-derived microparticle-based targeted chemotherapy in lung cancer patients with malignant pleural effusion. Sci Transl Med. 2019;11(474):eaat5690.
Escudier B, et al. Vaccination of metastatic melanoma patients with autologous dendritic cell (DC) derived-exosomes: results of thefirst phase I clinical trial. J Transl Med. 2005;3(1):10.
Gelderblom H, et al. Cremophor EL: the drawbacks and advantages of vehicle selection for drug formulation. Eur J Cancer. 2001;37(13):1590–8.
Wang P, et al. Exosomes from M1-polarized macrophages enhance paclitaxel antitumor activity by activating macrophages-mediated inflammation. Theranostics. 2019;9(6):1714–27.
Fellner S, et al. Transport of paclitaxel (Taxol) across the blood-brain barrier in vitro and in vivo. J Clin Invest. 2002;110(9):1309–18.
Toffoli G, et al. Exosomal doxorubicin reduces the cardiac toxicity of doxorubicin. Nanomedicine (Lond). 2015;10(19):2963–71.
Hadla M, et al. Exosomes increase the therapeutic index of doxorubicin in breast and ovarian cancer mouse models. Nanomedicine (Lond). 2016;11(18):2431–41.
Salarpour S, et al. Paclitaxel incorporated exosomes derived from glioblastoma cells: comparative study of two loading techniques. Daru. 2019;27(2):533–9.
Barani M, et al. Preparation, characterization, cytotoxicity and pharmacokinetics of niosomes containing gemcitabine: in vitro, in vivo, and simulation studies. J Drug Deliv Sci Technol. 2023;84:104505.
Zhao C, et al. Multifunctional transmembrane protein ligands for cell-specific targeting of plasma membrane-derived vesicles. Small. 2016;12(28):3837–48.
Koh E, et al. Exosome-SIRPα, a CD47 blockade increases cancer cell phagocytosis. Biomaterials. 2017;121:121–9.
Hong Y, et al. Degradation of tumour stromal hyaluronan by small extracellular vesicle-PH20 stimulates CD103(+) dendritic cells and in combination with PD-L1 blockade boosts anti-tumour immunity. J Extracell Vesicles. 2019;8(1):1670893.
Yuan Z, et al. TRAIL delivery by MSC-derived extracellular vesicles is an effective anticancer therapy. J Extracell Vesicles. 2017;6(1):1265291.
Shi X, et al. Genetically engineered cell-derived nanoparticles for targeted breast cancer immunotherapy. Mol Ther. 2020;28(2):536–47.
Cheng Q, et al. Reprogramming exosomes as nanoscale controllers of cellular immunity. J Am Chem Soc. 2018;140(48):16413–7.
Barok M, et al. Cancer-derived exosomes from HER2-positive cancer cells carry trastuzumab-emtansine into cancer cells leading to growth inhibition and caspase activation. BMC Cancer. 2018;18(1):504.
Castanotto D, Rossi JJ. The promises and pitfalls of RNA-interference-based therapeutics. Nature. 2009;457(7228):426–33.
Shahabipour F, Banach M, Sahebkar A. Exosomes as nanocarriers for siRNA delivery: paradigms and challenges. Arch Med Sci. 2016;12(6):1324–6.
Anastasiadou E, Jacob LS, Slack FJ. Non-coding RNA networks in cancer. Nat Rev Cancer. 2018;18(1):5–18.
Naseri Z, et al. Exosome-mediated delivery of functionally active miRNA-142-3p inhibitor reduces tumorigenicity of breast cancer in vitro and in vivo. Int J Nanomedicine. 2018;13:7727–47.
O’Brien KP, et al. Employing mesenchymal stem cells to support tumor-targeted delivery of extracellular vesicle (EV)-encapsulated microRNA-379. Oncogene. 2018;37(16):2137–49.
Lou G, et al. Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. J Hematol Oncol. 2015;8:122.
Zhang K, et al. Extracellular vesicle-mediated delivery of miR-101 inhibits lung metastasis in osteosarcoma. Theranostics. 2020;10(1):411–25.
Li P, Xin H, Lu L. Extracellular vesicle-encapsulated microRNA-424 exerts inhibitory function in ovarian cancer by targeting MYB. J Transl Med. 2021;19(1):4.
Wang X, et al. Exosomes Serve as nanoparticles to deliver anti-miR-214 to reverse chemoresistance to cisplatin in gastric cancer. Mol Ther. 2018;26(3):774–83.
Li Z, et al. In vitro and in vivo RNA inhibition by CD9-HuR functionalized exosomes encapsulated with miRNA or CRISPR/dCas9. Nano Lett. 2019;19(1):19–28.
Lang FM, et al. Mesenchymal stem cells as natural biofactories for exosomes carrying miR-124a in the treatment of gliomas. Neuro Oncol. 2018;20(3):380–90.
Limoni SK, et al. Engineered exosomes for targeted transfer of siRNA to HER2 positive breast cancer cells. Appl Biochem Biotechnol. 2019;187(1):352–64.
Aqil F, et al. Milk exosomes - natural nanoparticles for siRNA delivery. Cancer Lett. 2019;449:186–95.
Pi F, et al. Nanoparticle orientation to control RNA loading and ligand display on extracellular vesicles for cancer regression. Nat Nanotechnol. 2018;13(1):82–9.
Zheng Z, et al. Folate-displaying exosome mediated cytosolic delivery of siRNA avoiding endosome trapping. J Control Release. 2019;311–312:43–9.
Kamerkar S, et al. Exosomes facilitate therapeutic targeting of oncogenic KRAS in pancreatic cancer. Nature. 2017;546(7659):498–503.
Bonomi A, et al. Gemcitabine-releasing mesenchymal stromal cells inhibit in vitro proliferation of human pancreatic carcinoma cells. Cytotherapy. 2015;17(12):1687–95.
Yang Y, et al. Increased anti-tumour activity by exosomes derived from doxorubicin-treated tumour cells via heat stress. Int J Hyperthermia. 2015;31(5):498–506.
Smyth T, et al. Biodistribution and delivery efficiency of unmodified tumor-derived exosomes. J Control Release. 2015;199:145–55.
Herrmann IK, Wood MJA, Fuhrmann G. Extracellular vesicles as a next-generation drug delivery platform. Nat Nanotechnol. 2021;16(7):748–59.
Gresch O, et al. New non-viral method for gene transfer into primary cells. Methods. 2004;33(2):151–63.
Felgner PL, et al. Lipofection: a highly efficient, lipid-mediated DNA-transfection procedure. Proc Natl Acad Sci U S A. 1987;84(21):7413–7.
Fuhrmann G, et al. Active loading into extracellular vesicles significantly improves the cellular uptake and photodynamic effect of porphyrins. J Control Release. 2015;205:35–44.
Sancho-Albero M, et al. Efficient encapsulation of theranostic nanoparticles in cell-derived exosomes: leveraging the exosomal biogenesis pathway to obtain hollow gold nanoparticle-hybrids. Nanoscale. 2019;11(40):18825–36.
Zhang D, et al. Enrichment of selective miRNAs in exosomes and delivery of exosomal miRNAs in vitro and in vivo. Am J Physiol Lung Cell Mol Physiol. 2017;312(1):L110–l121.
Webber J, Clayton A. How pure are your vesicles? J Extracell Vesicles. 2013;2:19861.
Brezgin S, et al. Technological aspects of manufacturing and analytical control of biological nanoparticles. Biotechnol Adv. 2023;64:108122.
Deregibus MC, et al. Charge-based precipitation of extracellular vesicles. Int J Mol Med. 2016;38(5):1359–66.
Gámez-Valero A, et al. Size-exclusion chromatography-based isolation minimally alters extracellular vesicles’ characteristics compared to precipitating agents. Sci Rep. 2016;6:33641.
Alvarez-Erviti L, et al. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol. 2011;29(4):341–5.
Stickney Z, et al. Development of exosome surface display technology in living human cells. Biochem Biophys Res Commun. 2016;472(1):53–9.
Dooley K, et al. A versatile platform for generating engineered extracellular vesicles with defined therapeutic properties. Mol Ther. 2021;29(5):1729–43.
Ohno S, et al. Systemically injected exosomes targeted to EGFR deliver antitumor microRNA to breast cancer cells. Mol Ther. 2013;21(1):185–91.
Llorente A, et al. Molecular lipidomics of exosomes released by PC-3 prostate cancer cells. Biochim Biophys Acta. 2013;1831(7):1302–9.
Yáñez-Mó M, et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015;4:27066.
Kooijmans SAA, et al. Recombinant phosphatidylserine-binding nanobodies for targeting of extracellular vesicles to tumor cells: a plug-and-play approach. Nanoscale. 2018;10(5):2413–26.
Kooijmans SA, et al. Display of GPI-anchored anti-EGFR nanobodies on extracellular vesicles promotes tumour cell targeting. J Extracell Vesicles. 2016;5:31053.
Yang Z, et al. Large-scale generation of functional mRNA-encapsulating exosomes via cellular nanoporation. Nat Biomed Eng. 2020;4(1):69–83.
Fu W, et al. CAR exosomes derived from effector CAR-T cells have potent antitumour effects and low toxicity. Nat Commun. 2019;10(1):4355.
Salarpour S, et al. The application of exosomes and exosome-nanoparticle in treating brain disorders. J Mol Liq. 2022;350:118549.
Burnouf T, Agrahari V, Agrahari V. Extracellular vesicles as nanomedicine: hopes and hurdles in clinical translation. Int J Nanomedicine. 2019;14:8847–59.
Rufino-Ramos D, et al. Extracellular vesicles: novel promising delivery systems for therapy of brain diseases. J Control Release. 2017;262:247–58.
Fonseka P, Marzan AL, Mathivanan S. Introduction to the community of extracellular vesicles. Subcell Biochem. 2021;97:3–18.
Jeppesen DK, et al. Reassessment of exosome composition. Cell. 2019;177(2):428–445.e18.
Pathan M, et al. Vesiclepedia 2019: a compendium of RNA, proteins, lipids and metabolites in extracellular vesicles. Nucleic Acids Res. 2019;47(D1):D516–d519.
Kalra H, et al. Extracellular vesicles containing oncogenic mutant β-catenin activate Wnt signalling pathway in the recipient cells. J Extracell Vesicles. 2019;8(1):1690217.
Gaurav I, et al. Factors affecting extracellular vesicles based drug delivery systems. Molecules. 2021;26(6):1544.
Mehryab F, et al. Exosomes as a next-generation drug delivery system: an update on drug loading approaches, characterization, and clinical application challenges. Acta Biomater. 2020;113:42–62.
Möller A, Lobb RJ. The evolving translational potential of small extracellular vesicles in cancer. Nat Rev Cancer. 2020;20(12):697–709.
Shahi S, et al. Extracellular vesicles regulate cancer metastasis. Subcell Biochem. 2021;97:275–96.
Silva AK, et al. Combining magnetic nanoparticles with cell derived microvesicles for drug loading and targeting. Nanomedicine. 2015;11(3):645–55.
Zhang F, et al. Application of engineered extracellular vesicles for targeted tumor therapy. J Biomed Sci. 2022;29(1):14.
Qi H, et al. Blood exosomes endowed with magnetic and targeting properties for cancer therapy. ACS Nano. 2016;10(3):3323–33.
Acknowledgements
Not applicable.
Funding
This study was supported by the Excellent Academic Leader of Shanghai “Science and Technology Innovation Action Plan” (grant number: 22XD1402500), the Shanghai Shenkang Hospital Development Center (grant number: SHDC12020110, the Shanghai anti-Cancer Association “Eagle” program (grant number: SACA-CY21A05) and the Shanghai Shenkang development research physician project (grant number: SHDC2022CRD048).
Author information
Authors and Affiliations
Contributions
Manuscript writing: WL, YX, and ZJ. Manuscript revision: CS. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Wang, L., Yu, X., Zhou, J. et al. Extracellular Vesicles for Drug Delivery in Cancer Treatment. Biol Proced Online 25, 28 (2023). https://doi.org/10.1186/s12575-023-00220-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12575-023-00220-3