Abstract
Background
In 2006, the Senegalese National Malaria Control Programme recommended artemisinin-based combination therapy (ACT) as the first-line treatment for uncomplicated malaria. Since the introduction of ACT, there have been very few reports on the level of resistance of P. falciparum to anti-malarial drugs. To determine whether parasite susceptibility has been affected by the new anti-malarial policies, an ex vivo susceptibility and drug resistance molecular marker study was conducted on local isolates obtained from the Centre de santé Elizabeth Diouf (Médina, Dakar, Senegal).
Methods
The prevalence of genetic polymorphisms in genes associated with anti-malarial drug resistance, i.e., pfcrt, pfdhfr, pfdhps and pfmdr1, were evaluated for a panel of 165 isolates collected from patients recruited from 17 August 2010 to 6 January 2011. The malaria isolates were assessed for susceptibility to chloroquine (CQ); quinine (QN); monodesethylamodiaquine (MDAQ), the active metabolite of amodiaquine; mefloquine (MQ); lumefantrine (LMF); dihydroartemisinin (DHA), the active metabolite of artemisinin derivatives; and doxycycline (DOX) using the Plasmodium lactate dehydrogenase (pLDH) ELISA.
Results
The prevalence of the in vitro resistant isolates, or isolates with reduced susceptibility, was 62.1% for MQ, 24.2% for CQ, 10.3% for DOX, 11.8% MDAQ, 9.7% for QN, 2.9% for LMF and 0% for DHA. The Pfcrt 76T mutation was identified in 43.6% of the samples. The pfmdr1 86Y, 184F and 1246Y mutations were found in 16.2%, 50.0% and 1.6% of the samples, respectively. The pfdhfr 108N, 51I and 59R mutations were identified in 81.9%, 77.4% and 79.4% of the samples, respectively. The double mutant (108N and 51I) was detected in 75.5% of the isolates, and the triple mutant (108N, 51I and 59R) was detected in 73.6% of the isolates. The pfdhps 437G, 436A and 613S mutations were found in 54.4%, 38.6% and 1.2% of the samples, respectively. There was only one double mutant, 437G and 540E, and one quintuple mutant, pfdhfr 108N, 51I and 59R and pfdhps 437G and 540E. The prevalence of the quadruple mutant (pfdhfr 108N, 51I and 59R and pfdhps 437G) was 36.7%.
Conclusions
The results of this study indicate that an intensive surveillance of the in vitro P. falciparum susceptibility to anti-malarial drugs must be conducted in Senegal.
Similar content being viewed by others
Background
During the past 20 years, many strains of Plasmodium falciparum have become resistant to chloroquine and other anti-malarial drugs [1]. One strategy for reducing malaria prevalence is the use of drugs in combination. Drug combinations help prevent the development of resistance to each component drug and reduce the overall transmission of malaria [2]. In response to increasing chloroquine resistance, Senegal switched in 2004, to sulphadoxine-pyrimethamine with amodiaquine as the first-line therapy. In 2006, the Senegalese National Malaria Control Programme recommended artemisinin-based combination therapy (ACT) as the first-line treatment for uncomplicated malaria. The combination sulphadoxine-pyrimethamine and amodiaquine treatment was changed to artemether-lumefantrine and artesunate-amodiaquine. Since 2006, more than 1.5 million ACT-based treatments have been administered in Senegal [3]. In 2006, the Senegalese National Malaria Control Programme also recommended testing for all suspected cases of malaria with the P. falciparum histidine-rich protein 2 (PfHRP2)-based rapid diagnostic test (RDT). Since this time, ACT use has been restricted to confirmed malaria cases to reduce drug pressure. In 2009, 184,170 doses of ACT were dispensed in Senegal [4].
Malaria is transmitted in Dakar and its surrounding suburbs with a spatial heterogeneity of the human biting rate, which ranged from 0.1 to 250 bites per person per night during the rainy season from 2007 to 2010 [5, 6]. In 2008 to 2009, the human biting rate was 0.7 bites per person per night during the rainy season in Médina, a district of the south of Dakar [5]. In 2008, the P. falciparum prevalence varied from 0.9% to 7.4% in asymptomatic women and children in Dakar [7]. Morbidity in public health facilities decreased from 17.9% in 2007 to 2.6% in 2008 in Dakar [8].
Since the introduction of ACT, there have been very few reports on the level of resistance of P. falciparum to anti-malarial drugs. To determine whether parasite susceptibility has been affected by the new anti-malarial policies, an ex vivo susceptibility study was conducted on local isolates from Dakar obtained from the Centre de santé Elizabeth Diouf (Médina, Dakar, Senegal). The malaria isolates were assessed for susceptibility to chloroquine (CQ), quinine (QN), monodesethylamodiaquine (MDAQ), the active metabolite of amodiaquine, mefloquine (MQ), lumefantrine (LMF), dihydroartemisinin (DHA), the active metabolite of artemisinin derivatives and doxycycline (DOX).
In addition, the prevalence of genetic polymorphisms in genes associated with anti-malarial drug resistance was evaluated. The genes of interest included P. falciparum CQ resistance transporter (pfcrt) for CQ [9], P. falciparum dihydrofolate reductase (pfdhfr) for pyrimethamine [10], P. falciparum dihydropteroate synthase (pfdhps) for sulphadoxine [11] and P. falciparum multidrug resistance 1 (pfmdr1) for mefloquine resistance [12] and potentially for quinoline resistance [13, 14].
The pfcrt gene was first identified in 2000 [9]. To date, at least 20 point mutations have been described [9, 15, 16], but only one is the reference mutation (K76T), which is a marker of the CQ-resistant phenotype. This mutation is often associated with other mutations in the pfcrt gene, whose role is not yet defined. The odds ratio (OR) for CQ failure associated with the 76T mutation was 2.1 (95% confidence interval: 1.5-3.0, meta-analysis of 13 studies) for a 14-day follow-up and 7.2 (95%CI: 4.5-11.5, meta-analysis of 12 studies) for a 28-day follow-up [17]. However, the existence of CQ-susceptible strains associated with the 76T mutation suggests that other genes could be involved in the resistance to chloroquine.
The S108N mutation in the pfdhfr gene is associated with resistance to anti-folate drugs [18]. The OR for sulphadoxine-pyrimethamine failure associated with S108N was 3.5 (95%CI: 1.9-6.3, meta-analysis of 10 studies) for a 28-day follow-up [17]. The additional mutations N51I, C59R or I164L increase the level of in vitro resistance to anti-folate drugs and sulphadoxine-pyrimethamine. The OR values for codon 51 and 59 single mutants were 1.7 (95%CI: 1.0-3.0) and 1.9 (95%CI: 1.4-2.6), respectively [17]. The triple mutation (51 + 59 + 108) increases the risk of in vivo resistance to sulphadoxine-pyrimethamine by 4.3 (95%CI: 3.0-6.3, meta-analysis of 22 28-day studies) [17].
Sulphones (dapsone) and sulphonamides (sulphadoxine) are inhibitors of the P. falciparum DHPS [19]. The mutations S436A, S436F, A437G and K540E are involved in resistance to sulphadoxine [11]. The single mutation A437G and the double mutation A437G + K540E increase the risk of in vivo resistance to sulphadoxine-pyrimethamine by 1.5 (95%CI: 1.0-2.4, meta-analysis of 12 studies) and 3.9 (95%CI: 2.6-5.8, meta-analysis of 10 studies), respectively [17].
The quintuple mutant of pfdhfr (codons 51 + 59 + 108) plus pfdhps (codons 437 + 540) increases the risk of in vivo resistance to sulphadoxine-pyrimethamine by 5.2 (95%CI: 3.2-8.8, meta-analysis of 3 studies) [17]. Pfmdr1, which encodes a 162 kDa protein named P. falciparum homologue of the P-glycoprotein (Pgh1), is located on chromosome 5. Field work has shown that the predictive value for CQ resistance and point mutations in the pfmdr1 sequence resulting in amino acid changes varies depending on the geographic area [20, 21]. Five point mutations have been described: N86Y, Y184F, S1034C, N1042D and D1246Y. Point mutations, most notably N86Y, have been associated with a decrease in the CQ susceptibility [22]. However, in some of these epidemiological studies, the number of CQ-susceptible samples is too limited to provide a statistically meaningful analysis [21, 23]. Using precautions, no relationship or only weak relationships are established between CQ resistance and mutations in pfmdr1 in P. falciparum[24]. However, the risk of therapeutic failure with CQ is greater for patients harbouring the 86Y mutation, with an OR of 2.2 (95%CI: 1.6-3.1) for a 14-day follow-up and 1.8 (95%CI: 1.3-2.4) for a 28-day follow-up [17]. The combination of pfmdr1 86Y and pfcrt 76T increases the risk of in vivo resistance to CQ by 3.9 (95%CI: 2.6-5.8, meta-analysis of 5 studies) [17].
In addition, the risk of therapeutic failure with amodiaquine is greater for patients harbouring the 86Y mutation with an OR of 5.4 (95%CI: 2.6-11.2, meta-analysis of six studies) [17]. This mutation increases the risk of failure with amodiaquine plus sulphadoxine-pyrimethamine by 7.9 [25].
It has been shown through heterologous expression that pfmdr1 mutations at codons 1034 and 1042 abolish or reduce the level of resistance to mefloquine [26]. Moreover, transfection with a wild-type pfmdr1 allele at codons 1034, 1042 and 1246 confers mefloquine resistance to susceptible parasites [27]. However, mutations at codons 1034, 1042 and 1246 of Pfmdr1 in P. falciparum isolates are not sufficient to explain variations in mefloquine susceptibility [28]. Analyses of P. falciparum isolates showed an association between mutation at codon 86 and an increase in susceptibility to mefloquine, halofantrine or artemisinin derivatives [12, 29, 30].
Methods
Plasmodium falciparum isolates
In total, 165 patients (63 females, 3 to 70 years old and 102 males, 2 to 67 years old) with malaria were recruited from 17 August 2010 to 6 January 2011 at the Centre de santé Elizabeth Diouf. Venous blood samples were collected in Vacutainer® ACD tubes (Becton Dickinson, Rutherford, NJ, USA) prior to patient treatment and transferred to the Hôpital Principal de Dakar within six hours. Parasitaemia ranged from 0.001% to 20% in the male group and from 0.001% to 10.6% in the female group. Informed verbal consent was obtained from patients and/or their parents before blood collection. Assessment of P. falciparum susceptibility to anti-malarial drugs was realised with the same venous blood sample used for diagnostic purposes. The study was reviewed and approved by the ethical committee of the Pasteur Institute and the Hôpital Principal de Dakar. Patients were treated by artemether-lumefantrine or artesunate-amodiaquine (depending on availability).
Thin blood smears were stained using a RAL kit (Réactifs RAL, Paris, France) and were examined to determine the P. falciparum density and to confirm monoinfection. Parasitized erythrocytes were washed three times in RPMI 1640 medium (Invitrogen, Paisley, UK) buffered with 25 mM HEPES and 25 mM NaHCO3. If parasitaemia exceeded 0.5%, infected erythrocytes were diluted to 0.5% with uninfected erythrocytes (human blood type A+) and re-suspended in RPMI 1640 medium supplemented with 10% human serum (Abcys S.A. Paris, France), for a final haematocrit of 1.5%.
Drugs
CQ, QN, DHA and DOX were purchased from Sigma (Saint Louis, MO, USA). MDAQ was obtained from the World Health Organization (Geneva, Switzerland), MQ was purchased from Roche (Paris, France), and LMF was purchased from Novartis Pharma (Basel, Switzerland). QN, MDAQ, MQ, DHA and DOX were first dissolved in methanol and then diluted in water to final concentrations ranging from 5 nM to 3200 nM for QN, 1.56 nM to 1000 nM for MDAQ, 3.2 nM to 400 nM for MQ, 0.1 nM to 100 nM for DHA and 0.1 μM to 502 μM for DOX. CQ was re-suspended and diluted in water to final concentrations ranging from 5 nM to 3200 nM. LMF was re-suspended and diluted in ethanol to obtain final concentrations ranging from 0.5 nM to 310 nM.
The batches of plates were tested and validated on the CQ-susceptible 3D7 strain (West-Africa) and the CQ-resistant W2 strain (Indochina) (MR4, Virginia, USA) in 3 to 6 independent experiments using the same conditions described in the paragraph below. The two strains were synchronised twice with sorbitol before use [31], and clonality was verified every 15 days using PCR genotyping of the polymorphic genetic markers msp1 and msp2 and microsatellite loci [32, 33] and each year by an independent laboratory from the Worldwide Anti-malarial Resistance Network (WWARN).
Ex vivo assay
For the in vitro isotopic microtests, 200 μl of synchronous parasitized red blood cells (final parasitaemia, 0.5%; final haematocrit, 1.5%) was aliquoted into 96-well plates pre-dosed with anti-malarial drugs. The plates were incubated in a sealed bag for 42 h at 37°C with the atmospheric generators for capnophilic bacteria, Genbag CO2® at 5% CO2 and 15% O2 (BioMérieux, Marcy l’Etoile, France) [34]. After thawing the plates, haemolysed cultures were homogenised by vortexing the plates. Both the success of the drug susceptibility assay and the appropriate volume of haemolysed culture to use for each assay were determined for each clinical isolate during a preliminary pLDH ELISA. This pre-test and the subsequent ELISAs were performed using a commercial kit (ELISA-Malaria antigen test, ref 750101, DiaMed AG, Cressier, Morat, Switzerland) as previously described [35]. The optical density (OD) of each sample was measured with a spectrophotometer (Multiskan EX, Thermo Scientific, Vantaa, Finland).
The concentration at which the drugs were able to inhibit 50% of parasite growth (IC50) was calculated with the inhibitory sigmoid Emax model, with estimation of the IC50 through non-linear regression using a standard function of the R software (ICEstimator version 1.2) [36]. IC50 values were validated only if the OD ratio (OD at concentration 0 / OD at concentration max) was greater than 1.8 and the confidence interval ratio (upper 95% confidence interval of the IC50 estimation/lower 95% confidence interval of the IC50 estimation) was less than 2.0 [36].
Nucleic acid extraction
The total genomic DNA of each strain was isolated using the QIAamp® DNA Mini kit according to the manufacturer’s recommendations (Qiagen, Germany).
Pfcrt single-nucleotide polymorphisms (SNPs)
A 546-nucleotide fragment of the pfcrt gene (containing codon 76) was amplified by PCR using CRTP1-sense 5′-CCG TTA ATA ATA AAT ACA CGC AG-3′ and CRTP1-antisense 5′-CGG ATG TTA CAA AAC TAT AGT TAC C-3′ primers [37]. The reaction mixture for PCR amplifications included 2.5 μl of genomic DNA, 2.5 μl of 10X reaction buffer (Eurogentec), 0.5 μM of each primer, 200 μM of a deoxynucleoside triphosphate mixture (dGTP, dATP, dTTP and dCTP) (Euromedex, Souffelweyersheim, France), 2.5 mM MgCl2 and 1 unit of RedGoldStar® DNA polymerase (Eurogentec) in a final volume of 25 μl. The thermal cycler (T3 Biometra, Archamps, France) was programmed as follows: an initial 94°C incubation for 5 min; 40 cycles of 94°C for 20 sec, 56°C for 20 sec and 60°C for 40 sec; and a final 5-min extension step at 60°C. The PCR products were loaded on a 1.5% agarose gel containing 0.5 μg/mL ethidium bromide. The PCR products were diluted 1:100 in distilled water, and 2.5 μl of the final dilution was used for the second PCR. This PCR amplified a 275 bp segment around the mutation using a common inner primer CRTP3-sense 5′-TGA CGA GCG TTA TAG AG-3′ coupled with either CRTP4m-antisense 5′-GTT CTT TTA GCA AAA ATT G-3′ (detects the 76T codon), or CRTP4w-antisense 5′-GTT CTT TTA GCA AAA ATT T-3′ (detects the 76K codon) [15]. The reaction mixture for the PCR amplifications included 2.5 μl of diluted PCR product, 2.5 μl of 10X reaction buffer (Eurogentec), 0.5 μM of each primer, 200 μM deoxynucleoside triphosphate mixture, 1.5 mM MgCl2 and 0.75 U of RedGoldStar® DNA polymerase (Eurogentec) in a final volume of 25 μl.
The PCR conditions were as follows: initiation at 94°C for 5 min; 15 cycles at 94°C for 20 sec, 48.5°C for 20 sec and 64°C for 40 sec; and a final 5 min extension step at 64°C. Purified genomic DNA from P. falciparum clones 3D7 (chloroquine sensitive) and W2 (chloroquine resistant) were used as positive controls, and water and human DNA were used as negative controls. The PCR products from the amplification reactions were evaluated by electrophoresis on 2% agarose gels.
Pfmdr1 SNPs
Two primer pairs were used to amplify pfmdr1 fragments carrying the five key codons [38]. A 590-base pair fragment was amplified with the primer pair sense 5′-AGA GAA AAA AGA TGG TAA CCT CAG-3′ and antisense 5′-ACC ACA AAC ATA AAT TAA CGG-3′ to determine the sequences of codons 86 and 184 (MDR1-1), and a second fragment (968 base pairs) was amplified with the primer pair sense 5′-CAG GAA GCA TTT TAT AAT ATG CAT-3′ and antisense 5′-CGT TTA ACA TCT TCC AAT GTT GCA-3′ to determine the sequences of codons 1034, 1042, and 1246 (MDR1-2) [38]. The reaction mixture consisted of approximately 2.5 μl of genomic DNA, 0.5 μM of forward and reverse primers, 2.5 μl of 10X reaction buffer (Eurogentec), 2.5 mM MgCl2, 200 μM deoxynucleoside triphosphate mixture (dGTP, dATP, dTTP and dCTP) (Euromedex, Souffelweyersheim, France) and 1 U of RedGoldStar® DNA polymerase (Eurogentec) in a final volume of 25 μl. The thermal cycler (T3 Biometra) was programmed for MDR1-1 as follows: an initial step at 94°C for 5 min; 40 cycles of 94°C for 30 sec, 52°C for 30 sec and 72°C for 1 min; and a final 10-min extension step at 72°C. For MDR1-2 the parameters were as follows: an initial step at 94°C for 5 min; 40 cycles of 94°C for 30 sec, 56°C for 1 min and 72°C for 1 min 30 sec; and a final 10-min extension step at 72°C. The PCR products were loaded on a 1.5% agarose gel containing 0.5 μg/mL ethidium bromide. Amplicons were purified using the QIAquick 96 PCR BioRobot Kit and an automated protocol on the BioRobot 8000 workstation (Qiagen, Courtaboeuf, France). The purified fragments were sequenced using the BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems) using the primers described above. The sequencing reaction products were purified using the BigDye XTerminator® Purification Kit (Applied Biosystems) in accordance with the manufacturer’s instructions. The purified products were sequenced using an ABI Prism 3100 analyser (Applied Biosystems). The sequences were analysed using Vector NTI advance (TM) software (version 11, Invitrogen, Cergy Pontoise, France).
Pfdhfr SNP
A 562-bp fragment corresponding to the coding region of pfdhfr was amplified using the following primers: sense 5′-ACG TTT TCG ATA TTT ATG C-3′ and antisense 5′ TCA CAT TCA TAT GTA CTA TTT ATT C-3′[39]. The reaction mixture contained 2.5 μl of genomic DNA, 2.5 μl of 10X reaction buffer (Eurogentec), 0.5 μM each primer, 2.5 mM MgCl2, 200 μM deoxynucleoside triphosphate mixture (dGTP, dATP, dTTP and dCTP) (Euromedex, Souffelweyersheim, France) and 1 U of RedGoldStar® DNA polymerase (Eurogentec) in a final volume of 25 μl. The PCR conditions were as described in [39]. The amplified fragments were purified, sequenced (with the primers used for PCR) and analysed as described above.
Pfdhps SNP
A 672-bp fragment corresponding to the coding region of pfdhps was amplified using the following primers: sense 5′-GTT GAA CCT AAA CGT GCT GT-3′ and antisense 5′-TTC ATC ATG TAA TTT TTG TTG TG-3′[39]. The fragment was amplified as described for pfdhfr, and the PCR conditions were as described in [39]. The amplified fragments were purified, sequenced (with the primers used for PCR) and analysed as described above.
Data and statistical analysis
IC50 values were analysed after logarithmic transformation and expressed as the geometric mean of the IC50 and a confidence interval of 95% (CI95%). Using the Plasmodium lactate dehydrogenase (pLDH) ELISA under Genbag conditions, the cut-off values for in vitro resistance, or reduced susceptibility, were 77 nM, 61 nM, 115 nM, 12 nM, 611 nM, 30 nM and 37 μM for CQ, MDAQ, LMF, DHA, QN, MQ and DOX, respectively [40].
Results
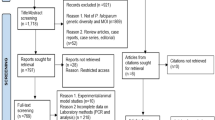
Out of the 165 patients recruited at the Centre de santé Elizabeth Diouf, 63 were tested ex vivo, and 34 isolates were successfully cultured. The average parameter estimates for the seven anti-malarial drugs utilised against the P. falciparum isolates are given in Table 1. The prevalence of P. falciparum isolates with decreased susceptibility to MQ in vitro reached 62.1%.
Pfcrt was examined in 156 of the samples. Within the pfcrt gene, the 76T mutation was identified in 43.6% of the samples. Eighteen samples (10.3%) were mixed, yielding both K76 and 76T.
The results for pfmdr1 polymorphisms are shown in Table 2. The 86Y mutation was identified in 14.9% of the tested samples. A mutation in codon 184 (184F) was identified in 49.4% of the isolates. Eighteen of the 24 isolates with the 86Y mutation were also mutated at codon 184 (184F). No new SNP was detected in the pfmdr1 gene.
The results for pfdhfr polymorphisms are presented in Table 3. There was a mutation in 81.9% of the samples for codon 108 (S108N/T), in 77.4% for codon 51 (N51I) and in 79.4% for codon 59 (C59R). The double mutant (108N and 51I) was detected in 75.5% of the isolates, and the triple mutant (108N, 51I and 59R) was detected in 73.6% of samples.
The results for the pfdhps polymorphisms are presented in Table 4. There was a mutation in 47.5% of the samples for codon 437 (A437G), in 33.5% for codon 436 (S436A), in 1.2% for codon 613 (A613S) and in 0.6% for codon 540 (K540E). There was only one (0.6%) double mutant (437G and 540E) and one (0.6%) quintuple mutant (pfdhfr 108N, 51I and 59R and pfdhps 437G and 540E), and 58 isolates (36.7%) were quadruple mutants (pfdhfr 108N, 51I and 59R and pfdhps 437G).
Discussion
This report describes the evaluation of the ex vivo susceptibility of P. falciparum isolates, taken from patients in Dakar, to seven standard anti-malarial drugs and the prevalence of several molecular markers involved in anti-malarial drug resistance. The majority of the patients, recruited at the Centre de santé Elizabeth Diouf (Médina, Dakar) from August 2010 to January 2011, said that they did not leave Dakar and its surrounding suburbs during the month preceding their malaria attack.
The prevalence of isolates with reduced susceptibility to MQ remains high (62.1%) in Dakar, but relatively stable compared with the previous year (55%) [40]. The level of in vitro resistance to MQ has increased since previous studies conducted in Senegal. In Dakar, the percent of isolates with decreased susceptibility was 17% in 2001 [41] and 13% in 2002 [33]. In Dielmo and Ndiop (280 km south-east of Dakar), the prevalence of in vitro resistance to MQ was 22% in 1995 [42] and 15% in 1999 [43, 44]. Prophylaxis failure with MQ has been previously described in Senegal [45], and MQ is one of the three anti-malarial drugs recommended for travellers as an anti-malarial prophylaxis in Senegal. Clinical trials are in progress to evaluate the efficacy of MQ for intermittent preventive treatment of infants and pregnant women, while MQ is still used for the treatment of uncomplicated malaria in infants in Dakar. Nevertheless, MQ has been employed relatively infrequently in Africa compared to Asia. The combination of artesunate-mefloquine, which is administered to patients in Asia, is not yet used in Senegal. However, scientific data are not available for MQ monotherapy, and very few data are available on the in vitro decreased susceptibility to MQ and its clinical implications in Africa. It is important to monitor the evolution of P. falciparum susceptibility to MQ, to archive suspicious isolates and to correlate clinical outcomes with pharmacokinetic and phenotypic responses and with molecular markers.
As far back as 1988, in vitro P. falciparum resistance to CQ was reported in Dakar, and reports of resistance in other regions of the country followed shortly [46]. From 1991 to 1995, parasitological failures were observed in 21% of patients in Pikine and in 23% of patients in another region of Senegal [47]. The in vitro resistance to CQ increased from 1995 to 1999 in Dielmo with 32% resistance in 1995 [48] compared to 49% resistance in 1996 [49], 44% resistance in 1997 [50] and 55% resistance in 1999 [43]. In 2010, the prevalence of in vitro resistance to CQ in Dakar was low and stable in comparison with the previous year (24.2% versus 22%) [40]. These data are consistent with previous work on in vitro resistance in Thies in 2007 (23% of isolates exhibiting CQ resistance) [51]. The evolution of susceptibility to CQ is confirmed by evaluation of molecular markers of CQ resistance, and mutations in pfcrt have been shown to be correlated with CQ resistance in different parts of the world [52]. The prevalence of the pfcrt 76T mutation has decreased since 2004 in Dakar. From 2000 to 2001 in Guediawaye, a suburb of Dakar, a prevalence of 92% of was observed for the 76T mutation in pregnant women with malaria [53]. In Pikine, another suburb of Dakar, the prevalence of the 76T mutant was 79% in 2000 [54], 63.9% in 2001 [55] and 59.5% in 2004 [56]. In 2002, the prevalence of the pfcrt 76T mutation was 65% in patients hospitalised for malaria at the Hôpital Principal de Dakar [33]. From 2001 to 2002, the prevalence of the pfcrt 76T mutation was 75.8% in pregnant women taking chloroquine prophylaxis in Thiadiaye (84 km southeast of Dakar) [57]. In this study, the pfcrt 76T mutation was identified in 43.6% of the patients recruited from August 2010 to January 2011. These data are consistent with previous works on CQ molecular resistance in Dakar in 2009 (37.8%) [58], in central Senegal (Mbour, Fatick and Bambey) in 2009 (29.3%) and in 2010 (25.1%); and in south Senegal (Tambacounda, Velingara and Saraya) with 2010 (34.5%) and in 2011 (28.8%) [59]. However, in Pikine, the prevalence of the pfcrt 76T mutation ranged from 64% to 79% before CQ withdrawal (2000 to 2003) [54, 55, 60], the prevalence then decreased to 47-60% [56, 60] while amodiaquine plus pyrimethamine-sulphadoxine was the first-line treatment (2004–2005); this prevalence has increased slightly to 59% since ACT has been implemented (2006 to 2009) [60].
This decrease in CQ resistance parallels the withdrawal of CQ treatment and the introduction of ACT in 2002 in Senegal. However, in 2003, CQ was still being administered to patients. The prevalence of CQ in the urine ranged from 14.5% to 47.5% in two- to nine-year-old children from northern Senegal and from 9.0% to 21.4% in children from southern Senegal [61]. In 2006, the Senegalese National Malaria Control Programme recommended ACT as the first-line treatment for uncomplicated malaria, in this same year, Senegal reported 10.6% chloroquine use and 9.7% ACT use [62]. In Dakar in 2006, CQ represented 5.1% of the anti-malarial drugs used in children [63] and 3.5% in 2009 [64]. Since 2006, more than 1.5 million ACT treatments have been administered in Senegal [3], and 184,170 doses of ACT were dispensed in 2009 [4]. A reduction in CQ resistance was also reported in Malawi after the withdrawal of CQ treatment [65]. This observation prompted an in vivo CQ study in Malawi five years later, in which CQ was found to be 99% effective [66]. The rapid dissemination of CQ resistance in Dielmo, despite strictly controlled anti-malarial drug use, argues against the re-introduction of CQ, at least in mono-therapy, in places where the resistant allele has dropped to very low levels following the discontinuation of CQ treatment [67]. Despite the regain of CQ susceptibility, any reintroduction would likely result in a rapid re-emergence of resistant strains.
The prevalence of isolates with in vitro reduced susceptibility to MDAQ slightly increased in 2010, 11.8% versus 6% in 2009 [40]. The prevalence of reduced susceptibility to MDAQ was 0% in 1996 and 1999 in Dielmo [44, 48], while it was 5% in Mlomp (Casamance, south-western Senegal) in 2004 [68].
The prevalence of the pfmdr1 mutations 86Y, 184F and 1246Y were 16.2%, 50.0% and 1.6%, respectively. In 2000 and 2001, prevalence rates of 31% and 30.6%, respectively, were observed for pfmdr1 86Y in Pikine [54, 55], and a prevalence rate of 17.2% was observed in Dakar in 2009 [58]. However, the role of polymorphism in pfmdr1 is still debated. The pfmdr1 86Y mutation has been shown to be associated with in vivo resistance to amodiaquine in recrudescence after monotherapy with amodiaquine [69] or after combination therapy with artesunate-amodiaquine [70]. The pfmdr1 1246Y mutation has also been found to be associated with in vitro resistance to amodiaquine [71] and with recrudescent infection after treatment with amodiaquine or amodiaquine-artesunate [70, 72]. In a meta-analysis, the pfmdr1 86Y mutation was found to be associated with amodiaquine failure, with an odds ratio of 5.4 [17]. Based on this hypothesis, the 16.2% prevalence of pfmdr1 of 86Y predicts that 16.2% of isolates would be resistant to amodiaquine in 2010 in Senegal. The resistance to amodiaquine has remained low even after the introduction of artesunate-amodiaquine in 2006 in Senegal. A study in Dakar and Mlomp from 1996 to 1998 showed that monotherapy with amodiaquine remained effective for treating uncomplicated malaria in areas where CQ resistance was prevalent [73]. The artesunate-amodiaquine–associated cure rates were > 99.3% in Mlomp and Keur-Socé when administered either as a single daily dose or as two daily doses [74]. The fixed-dose combination of artesunate-amodiaquine (ASAQ) exhibits a cure rate > 98.5% [75]. However, ACT efficacy and resistance must be monitored because clinical failures, or at least extended parasite clearance times, have been described in Cambodia [76, 77]. In this context, it is important to implement in vitro and in vivo surveillance programmes, such as those championed by the Worldwide Antimalarial Resistance Network [78, 79].
No isolate exhibited reduced in vitro susceptibility to DHA. This result is consistent with previous studies that did not show any parasites resistant to artesunate [33, 43, 44]. However, Agnamey et al. reported that 3% - 23% of isolates had IC50 values greater than 15 nM in Mlomp between 2000 and 2004 [73]. High IC50 values can also be found for artemisinin, with an IC50 > 30 nM in Dakar [51] and artesunate with an IC50 > 45 nM [41].
The other ACT first-line treatment for uncomplicated P. falciparum malaria in Senegal is the combination of artemether-lumefantrine. Only 2.9% of the isolates presented reduced susceptibility to LMF, and this prevalence did not rise in Senegal after the introduction of ACT. In 1996, 6% of isolates from Dielmo were resistant to LMF in vitro[80]; and 1% of the isolates were resistant to LMF in 2009 in Dakar [40].
In 2009, 7% of isolates showed low reduced susceptibility to QN, which is in accordance with previous studies in Dakar [33, 40, 41] and Dielmo [42–44]. Isolates with a high IC50 to QN were already identified in 1984 [81]. Even in areas where QN efficacy remains good, such as sub-Saharan Africa, the susceptibility of individual P. falciparum isolates to QN has varied widely. The IC50s for isolates collected in Senegal were 31 to 765 nM in 1984 (Thies and Kaolack) [81], 5 to 932 nM in 1996 (Dielmo) [49] and 6 to 1291 nM in 2009 (Dakar) [40]. The wide range in QN susceptibility and recent evidence for QN treatment failure seen across Africa [82, 83] or in Senegal in a patient who spent two months in Dielmo in 2007 [84] suggest that the evolution of parasites with reduced susceptibility may contribute to QN decreased efficacy. QN used in combination with DOX in the treatment of severe malaria in Dakar.
A prevalence of 10.3% of isolates with in vitro reduced susceptibility to DOX was observed in Dakar in 2010, which is similar to the prevalence observed in 2009 (12%) [40]. The mean IC50 of DOX was similar to those estimated in Dielmo in 1998 [85, 86]. The slow activity of DOX in vitro has a delayed effect upon growth and requires the prolonged incubation of parasites [85, 87]. However, the standard 42 h test is still used to monitor DOX in vitro susceptibility.
The pfdhfr 108N mutation has been shown to be correlated with in vitro and in vivo resistance to pyrimethamine [10, 17]. The OR for sulphadoxine-pyrimethamine failure associated with Ser108Asn has been shown to be 3.5 (95%CI: 1.9-6.3, meta-analysis of 10 studies) for a 28-day follow-up [17]. The additional mutations N51I, C59R or I164L increase the level of in vitro resistance to anti-folate drugs and sulphadoxine-pyrimethamine. The OR values for single mutants of codons 51 and 59 are 1.7 (95%CI: 1.0-3.0) and 1.9 (95%CI: 1.4-2.6), respectively [17]. In 2010, the prevalence of pfdhfr 108N was 81.9% in patients with malaria who were treated at the Centre de santé Elizabeth Diouf, which is similar to the results of those treated in 2009 at the Hôpital Principal de Dakar (82.4%) [58]. The triple mutation (51 + 59 + 108) increases the risk of in vivo resistance to sulphadoxine-pyrimethamine by 4.3 (95%CI: 3.0-6.3, meta-analysis of 22 28-day studies). Isolates carrying a combination of three mutations (108N, 51I and 59R) were associated with high-level pyrimethamine resistance and represented 73.6% of isolates. In 2002, in Dakar, the prevalence of pfdhfr 108N was 65%, and triple mutants were identified in 50% of the isolates [33]. In 2003, the prevalence of mutations in pfdhfr codon 108 was 78% in Pikine, and the prevalence of the triple mutant was 61% [88]. Additionally, in 2007, in the rural area Keur Soce, triple mutants were identified in 67% of patients treated with sulphadoxine-pyrimethamine combined with amodiaquine [89].
The pfdhps 437G mutation has been shown to be correlated with in vitro and in vivo resistance to sulphadoxine [11, 17]. The single mutation Ala437Gly and the double mutation Ala437Gly + Lys540Glu increase the risk of in vivo resistance to sulphadoxine-pyrimethamine by 1.5 (95%CI: 1.0-2.4, meta-analysis of 12 studies) and 3.9 (95%CI: 2.6-5.8, meta-analysis of 10 studies), respectively [17]. In 2010, the prevalence of the pfdhps 437G mutation was 54.4% in patients with malaria in Dakar. Only one isolate (0.6%) carried the double mutation (437G and 540E) that is associated with high-level sulphadoxine resistance. The mutation of codon 613 (A613S) (1.2%) is very rare in Africa. In 2002, in Dakar, only 20% of isolates harboured the pfdhps 437G mutation [33]. In 2003, the mutation rate in pfdhps codon 437G was 40% in Pikine [88]. Several studies from 2006 to 2008 in Senegal showed that the prevalence of pfdhps 437G significantly increased after intermittent preventive treatment of infants with sulphadoxine-pyrimethamine [89, 90]. Given the prevalence of the triple and quadruple mutants in the population of Dakar (73.6% for the pfdhfr 108N, 51I and 59R triple mutant and 36.7% for the quadruple mutant pfdhfr 108N, 51I and 59R and pfdhps 437G), the use of sulphadoxine-pyrimethamine as an intermittent preventive treatment (IPT) must be monitored. Encouragingly, only one quintuple mutant, Pfdhfr 108N, 51I and 59R and Pfdhps 437G and 540E, which is associated with high-level sulphadoxine-pyrimethamine resistance has been identified to date.
IPT with anti-malarial drugs given to all children and pregnant women once per month during the transmission season can provide a high degree of protection against malaria. Seasonal IPT with sulphadoxine-pyrimethamine and one dose of artesunate resulted in a 90% reduction in the incidence of clinical malaria in Senegal [6]. The combination of sulphadoxine-pyrimethamine and amodiaquine was more effective than the combination of sulphadoxine-pyrimethamine and artesunate or the combination of amodiaquine and artesunate in preventing malaria [91]. During IPT with sulphadoxine-pyrimethamine and piperaquine, only 3.4% of the treated children developed malaria [89]. However, the single use of sulphadoxine-pyrimethamine as seasonal IPT is inadvisable; for instance, sulphadoxine-pyrimethamine must be used in combination with amodiaquine, artesunate or piperaquine [89, 92].
The introduction of ACT in 2002 in Senegal did not induce a decrease in P. falciparum susceptibility to individual drug components, such as DHA, MDAQ and LMF. However, the prevalence of P. falciparum isolates with reduced drug susceptibility to MQ increased, and clinical failures with QN have been reported in Senegal. Additionally, in Senegal, isolates with high IC50 values for artemisinin derivatives. Since 2004, the prevalence of chloroquine resistance has decreased, but the data argue against the re-introduction of chloroquine for mono-therapy in places where the resistant allele has dropped to very low levels following discontinuation of chloroquine treatment. The prevalence of isolates resistant to pyrimethamine is high (81.9%), with 73.6% of parasites exhibiting high-level pyrimethamine resistance. The prevalence of isolates resistant to sulphadoxine was 54.4%. Susceptibility to anti-malarial drugs remains stable between 2009 and 2010 in Dakar. Nevertheless, an intensive surveillance of the susceptibility of P. falciparum to anti-malarial drugs in vitro must be conducted in Senegal. In addition, maximising the efficacy and longevity of ACT as a tool to control malaria will critically depend on pursuing intensive research into identifying in vitro markers as well as implementing ex vivo and in vivo surveillance programmes. In this context, there is a need to identify molecular markers that predict ACT resistance, which can provide an active surveillance method to monitor temporal trends in parasite susceptibility.
References
Le Bras J, Musset L, Clain J: Antimalarial drug resistance. Med Mal Infect. 2006, 36: 401-405. 10.1016/j.medmal.2006.05.005.
White NJ: Preventing antimalarial drug resistance through combinations. Drug. Resist Updat. 2001, 1: 3-9.
Ndiaye JLA, Faye B, Gueye A, Tine R, Ndiaye D, Tchania C, Ndiaye I, Barry A, Cisse B, Lameyre V, Gaye O: Repeated treatment of recurrent uncomplicated Plasmodium falciparum malaria in Senegal with fixed-dose artesunate plus amodiaquine versus fixed-dose artemether plus lumefantrine: a randomized, open-label trial. Malar J. 2011, 10: 237-10.1186/1475-2875-10-237.
Thiam S, Thior M, Faye B, Diouf ML, Diouf MB, Diallo I, Fall FB, Ndiaye JL, Albertini A, Lee E, Jorgensen P, Gaye O, Bell D: Major reduction in anti-malarial drug consumption in Senegal after nation-wide introduction of malaria rapid diagnostic tests. Plos One. 2011, 6: 18419-10.1371/journal.pone.0018419.
Gadiaga L, Machault V, Pagès F, Gaye A, Jarjaval F, Godefroy L, Cissé B, Lacaux JP, Sokhna C, Trape JF, Rogier C: Conditions of malaria transmission in Dakar from 2007 to 2010. Malar J. 2011, 10: 312-10.1186/1475-2875-10-312.
Drame PM, Machault V, Diallo A, Cornélie S, Poinsignon A, Lalou R, Sembène M, Dos Santos S, Rogier C, Pagès F, Le Hesran JY, Remoué F: IgG responses to the gSG6-P1 salivary peptide for evaluating human exposure to Anopheles bites in urban areas of Dakar. Senegal. Malar J. 2012, 11: 72-10.1186/1475-2875-11-72.
Diallo A, Ndam NT, Moussiliou A, Dos Santos S, Ndonky A, Borderon M, Oliveau S, Lalou R, Le Hesran JY: Asymptomatic carriage of Plasmodium in urban Dakar: the risk of malaria should not be underestimated. Plos One. 2012, 7: 31300-10.1371/journal.pone.0031300.
Ministère de la Santé et de la Prévention: Rapport sur la morbidité et la mortalité palustre au Sénégal en 2008. 2010, Dakar: Programme National de Lutte contre le Paludisme,http://www.pnlp.sn/UserFiles/File/donnee.pdf,
Fidock DA, Nomura T, Talley AK, Cooper RA, Dzekunov SM, Ferdig MT, Ursos LMB, Sidhu ABS, Naudé B, Deitsch KW, Su XZ, Wootton JC, Roepe PD, Wellems TE: Mutations in the P. falciparum digestive vacuole transmembrane protein PfCRT and evidence for their role in chloroquine resistance. Mol Cell. 2000, 6: 861-871. 10.1016/S1097-2765(05)00077-8.
Zolg JW, Plitt JR, Chen GX, Palmer S: Point mutations in the dihydrofolate reductase-thymidylate synthase gene as the molecular basis for pyrimethamine resistance in Plasmodium falciparum. Mol Biochem Parasitol. 1989, 36: 253-262. 10.1016/0166-6851(89)90173-4.
Wang P, Read M, Sims PF, Hyde JE: Sulfadoxine resistance in the human malaria parasite Plasmodium falciparum is determined by mutations in dihydropteroate synthetase and an additional factor associated with folate utilisation. Mol Microbiol. 1997, 23: 979-986. 10.1046/j.1365-2958.1997.2821646.x.
Price RN, Cassar C, Brockman A, Duraisingh M, van Vugt M, White NJ, Nosten F, Krishna S: The pfmdr1 gene is associated with a multidrug-resistant phenotype in Plasmodium falciparum from the Western border of Thailand. Antimicrob Agents Chemother. 1999, 43: 2943-2949.
Henry M, Alibert S, Orlandi-Pradines E, Bogreau H, Fusai T, Rogier C, Barbe J, Pradines B: Chloroquine resistance reversal agents as promising antimalarial drugs. Curr Drug Targets. 2006, 7: 935-948. 10.2174/138945006778019372.
Henry M, Alibert S, Rogier C, Barbe J, Pradines B: Inhibition of efflux of quinolines as new therapeutic strategy in malaria. Curr Top Med Chem. 2008, 8: 563-578. 10.2174/156802608783955593.
Nagesha HS, Casey GJ, Rieckmann H, Fryauff DJ, Laksana BS, Reeder JC, Maguire JD, Baird J: New haplotypes of the Plasmodium falciparum chloroquine resistance transporter (pfcrt) gene among chloroquine-resistant parasite isolates. Am J Trop Med Hyg. 2003, 68: 398-402.
Johnson DJ, Fidock DA, Mungthin M, Lakshmanan V, Sidhu AB, Bray PG, Ward SA: Evidence for a central role for PfCRT in conferring Plasmodium falciparum resistance to diverse antimalarial agents. Mol Cell. 2004, 15: 867-877. 10.1016/j.molcel.2004.09.012.
Picot S, Olliaro P, de Monbrison F, Bienvenu AL, Price RN, Ringwald P: A systematic review and meta-analysis of evidence for correlation between molecular markers of parasite resistance and treatment outcome in falciparum malaria. Malar J. 2009, 8: 89-10.1186/1475-2875-8-89.
Sibley CH, Hyde JE, Sims PF, Plowe CV, Kublin JG, Mbenu EK, Cowman AF, Winstanley PA, Watkins WM, Nzila AM: Pyrimethamine-sulfadoxine resistance in Plasmodium falciparum: what next?. Trends Parasitol. 2001, 17: 582-588. 10.1016/S1471-4922(01)02085-2.
Zhang Y, Meshnick SR: Inhibition of Plasmodium falciparum dihydropteroate synthetase and growth in vitro by sulfa drugs. Antimicrob Agents Chemother. 1991, 35: 267-271. 10.1128/AAC.35.2.267.
Basco LK, Ringwald P: Molecular epidemiology of malaria in Yaoundé, Cameroon. III. Analysis of chloroquine resistance and point mutations in the multidrug resistance 1 (pfmdr 1) gene of Plasmodium falciparum. Am J Trop Med Hyg. 1998, 59: 577-581.
Duraisingh MT, Drakeley CJ, Muller O, Bailey R, Snounou G, Targett GA, Greenwood BM, Warhurst DC: Evidence for selection for the tyrosine-86 allele of the pfmdr 1 gene of Plasmodium falciparum by chloroquine and amodiaquine. Parasitology. 1997, 114: 205-211. 10.1017/S0031182096008487.
Foote SJ, Kyle DE, Martin RK, Oduola AM, Forsyth K, Kemp DJ, Cowman AF: Several alleles of the multidrug-resistance gene are closely linked to chloroquine resistance. Nature. 1990, 345: 255-258. 10.1038/345255a0.
Grobusch MP, Adagu IS, Kremsner PG, Warhurst DC: Plasmodium falciparum: in vitro chloroquine susceptibility and allele-specific PCR detection of Pfmdr1 Asn86Tyr polymorphism in Lambarene, Gabon. Parasitology. 1998, 116: 211-217. 10.1017/S0031182097002266.
Basco LK, de Pecoulas PE, Le Bras J, Wilson EN: Plasmodim falciparum: Molecular characterization of multi drug-resistant Cambodian isolates. Exp Parasitol. 1996, 82: 97-103.
Marfurt J, Muller I, Sie A, Oa O, Reeder JC, Smith TA, Beck HP, Genton B: The usefulness of twenty-four molecular markers in predicting treatment outcome with combination therapy of amodiaquine plus sulphadoxine-pyrimethamine against falciparum malaria in Papua New Guinea. Malar J. 2008, 7: 61-10.1186/1475-2875-7-61.
Ruetz S, Delling U, Brault M, Schurr E, Gros P: The pfmdr1 gene of Plasmodium falciparum confers cellular resistance to antimalarial drugs in yeast cells. Proc Natl Acad Sci USA. 1996, 93: 9942-9947. 10.1073/pnas.93.18.9942.
Reed MB, Saliba KJ, Caruana SR, Kirk K, Cowman AF: Pgh1 modulates sensitivity and resistance to multiple antimalarials in Plasmodium falciparum. Nature. 2000, 403: 906-909. 10.1038/35002615.
Pillai DR, Hijar G, Montoya I, Marquino W, Ruebush TK, Wrongsrichanalai C, Kain KC: Lack of prediction of mefloquine and mefloquine-artesunate treatment outcome by mutations in the Plasmodium falciparum multidrug resistance 1 (pfmdr1) gene for P. falciparum malaria in Peru. Am J Trop Med Hyg. 2003, 68: 107-110.
Duraisingh MT, Jones P, Sambou I, von Seidlein L, Pinder M, Warhurst DC: The tyrosine-86 allele of the pfmdr1 gene of Plasmodium falciparum is associated with increased sensitivity to the anti-malarials mefloquine and artemisinin. Mol Biochem Parasitol. 2000, 108: 13-23. 10.1016/S0166-6851(00)00201-2.
Duraisingh MT, Roper C, Walliker D, Warhurst DC: Increased sensitivity to the antimalarials mefloquine and artemisinin is conferred by mutations in the pfmdr1 gene of Plasmodium falciparum. Mol Microbiol. 2000, 36: 955-961. 10.1046/j.1365-2958.2000.01914.x.
Lambros C, Vanderberg JP: Synchronization of Plasmodium falciparum erythrocytic stages in culture. J Parasitol. 1979, 65: 418-420. 10.2307/3280287.
Bogreau H, Renaud F, Bouchiba H, Durand P, Assi SB, Henry MC, Garnotel E, Pradines B, Fusai T, Wade B, Adehossi E, Parola P, Kamil MO, Puijalon O, Rogier C: Genetic diversity and structure of African Plasmodium falciparum populations in urban and rural areas. Am J Trop Med Hyg. 2006, 74: 953-959.
Henry M, Diallo I, Bordes J, Ka S, Pradines B, Diatta B, M’Baye PS, Sane M, Thiam M, Gueye PM, Wade B, Touze JE, Debonne JM, Rogier C, Fusai T: Urban malaria in Dakar, Senegal: chemosusceptibility and genetic diversity of Plasmodium falciparum isolates. Am J Trop Med Hyg. 2006, 75: 146-151.
Pascual A, Basco LK, Baret E, Amalvict R, Travers D, Rogier C, Pradines B: Use of the atmospheric generators for capnophilic bacteria Genbag CO2® for the evaluation of in vitro Plasmodium falciparum susceptibility to standard anti-malarial drugs. Malar J. 2011, 10: 8-10.1186/1475-2875-10-8.
Kaddouri H, Nakache S, Houzé S, Mentré F, Le Bras J: Assessment of the drug susceptibility of Plasmodium falciparum clinical isolates from Africa using a Plasmodium lactate dehydrogenase immunodetection assay and an inhibitory maximum effect model for precise measurement of the 50-percent inhibitory concentration. Antimicrob Agents Chemother. 2006, 50: 3343-3349. 10.1128/AAC.00367-06.
Le Nagard H, Vincent C, Mentré F, Le Bras J: Online analysis of in vitro resistance to antimalarial drugs through nonlinear regression. Comput Methods Programs Biomed. 2011, 104: 10-18. 10.1016/j.cmpb.2010.08.003.
Tinto H, Ouédraogo JB, Erhart A, van Overmeir C, Dujardin JC, van Marck E, Guiguemdé TR, D’Alessandro U: Relationship between the Pfcrt T76 and the Pfmdr-1 Y86 mutations in Plasmodium falciparum and in vitro/in vivo chloroquine resistance in Burkina Faso, West Africa. Infect Genet Evol. 2003, 3: 287-292. 10.1016/j.meegid.2003.08.002.
Basco LK, Ringwald P: Molecular epidemiology of malaria in Cameroon.X. Evaluation of Pfmdr1 mutations as genetic markers for resistance to amino alcohols and artemisinin derivatives. Am J. Trop Med Hyg. 2002, 66: 667-671.
Parola P, Pradines B, Simon F, Carlotti MP, Minodier P, Ranjeva MP, Badiaga S, Bertaux L, Delmont J, Morillon M, Silai R, Brouqui P, Parzy D: Antimalarial drug susceptibility and point mutations associated with resistance in 248 Plasmodium falciparum isolates imported from Comoros to Marseille. Am J Trop Med Hyg. 2007, 77: 431-437.
Fall B, Diawara S, Sow K, Baret E, Diatta B, Fall KB, Mbate PS, Fall F, Diémé Y, Rogier C, Wade B, Bercion R, Pradines B: Ex vivo susceptibility of Plasmodium isolates from Dakar, Senegal, to seven standard anti-malarial drugs. Malar J. 2011, 10: 310-10.1186/1475-2875-10-310.
Jambou R, Legrand E, Niang M, Khim N, Lim P, Volney B, Ekala MT, Bouchier C, Esterre P, Fandeur T, Mercereau-Puijalon O: Resistance of Plasmodium falciparum field isolates to in vitro artemether and point mutations of the SERCA-type PfATPase6. Lancet. 2005, 366: 1960-1963. 10.1016/S0140-6736(05)67787-2.
Pradines B, Rogier C, Fusai T, Tall A, Doury JC: Sensibilité in vitro de 85 isolats de Plasmodium falciparum au Sénégal. Med Trop. 1996, 56: 141-145.
Pradines B, Tall A, Rogier C, Spiegel A, Mosnier J, Marrama L, Fusai T, Millet P, Panconi E, Trape JF, Parzy D: In vitro activities of ferrochloroquine against 55 Senegalese isolates of Plasmodium falciparum in comparison with those of standard antimalarial drugs. Trop Med Int Health. 2002, 7: 265-270. 10.1046/j.1365-3156.2002.00848.x.
Pradines B, Mabika Mamfoumbi M, Tall A, Sokhna C, Koeck JL, Fusai T, Mosnier J, Czarnecki E, Spiegel A, Trape JF, Kombila M, Rogier C: In vitro activity of tafenoquine against the asexual blood stages of Plasmodium falciparum isolates from Gabon, Senegal, and Djibouti. Antimicrob Agents Chemother. 2006, 50: 3225-3226. 10.1128/AAC.00777-06.
Gari-Toussaint M, Pradines B, Mondain V, Keundjian A, Dellamonica P, Le Fichoux Y: Sénégal et paludisme. Echec prophylactique vrai de la méfloquine. Presse Med. 2002, 31: 1136-
Hatin I, Trape JF, Legros F, Bauchet J, Le Bras J: Susceptibility of Plasmodium falciparum strains to mefloquine in an urban area in Senegal. Bull World Health Organ. 1992, 70: 363-367.
Sokhna C, Molez JF, Ndiaye P, Sane B, Trape JF: Tests in vivo de chimiosensibilite de Plasmodium falciparum à la chloroquine au Sénégal: évolution de la résistance et estimation de l’efficacité thérapeutique. Bull Soc Pathol Exot. 1997, 90: 83-89.
Pradines B, Rogier C, Fusai T, Tall A, Trape JF, Doury JC: In vitro activity of artemether against African isolates (Senegal) of Plasmodium falciparum in comparison with standard antimalarial drugs. Am J Trop Med Hyg. 1998, 58: 354-357.
Pradines B, Tall A, Parzy D, Spiegel A, Fusai T, Hienne R, Trape JF, Doury JC: In vitro activity of pyronaridine and amodiaquine against African isolates (Senegal) of Plasmodium falciparum in comparison with standard antimalarial agents. J Antimicrob Chemother. 1998, 42: 333-339. 10.1093/jac/42.3.333.
Pradines B, Tall A, Ramiandrasoa F, Spiegel A, Sokhna C, Fusai T, Mosnier J, Daries W, Trape JF, Kunesch G, Parzy D, Rogier C: In vitro activity of iron-binding compounds against Senegalese isolates of Plasmodium falciparum. J Antimicrob Chemother. 2006, 57: 1093-1099. 10.1093/jac/dkl117.
Ndiaye D, Patel V, Demas A, LeRoux M, Ndir O, Mboup S, Clardy J, Lakshmanan V, Daily JP, Wirth DF: Short report: an non-radioactive DAPI-based high-throughput in vitro assay to assess Plasmodium falciparum responsiveness to antimalarials – Increased sensitivity of P. falciparum to chloroquine in Senegal. Am J Trop Med Hyg. 2010, 82: 228-230. 10.4269/ajtmh.2010.09-0470.
Djimdé A, Doumbo OK, Cortese JF, Kayentao K, Doumbo S, Diourté Y, Dicko A, Su XZ, Nomura T, Fidock DA, Wellems TE, Plowe CV: A molecular marker for chloroquine-resistant falciparum malaria. N Engl J Med. 2001, 344: 257-263. 10.1056/NEJM200101253440403.
Niang M, Marrama L, Ekala MT, Gaye A, Tall A, Ndiaye JL, Sarr D, Dangou JM, Lehesran JY, Bouchier C, Mercereau-Puijalon O, Jambou R: Accumulation of CIEVT Pfcrt allele of Plasmodium falciparum in placenta of pregnant women living in an urban area of Dakar, Senegal. J Antimicrob Chemother. 2008, 62: 921-928. 10.1093/jac/dkn299.
Thomas SM, Ndir O, Dieng T, Mboup S, Wypij D, Maguire JH, Wirth DF: In vitro chloroquine susceptibility and PCR analysis of pfcrt and pfmdr1 polymorphisms in Plasmodium falciparum isolates from Senegal. Am J Trop Med Hyg. 2002, 66: 474-480.
Sarr O, Myrick A, Daily J, Diop BM, Dieng T, Ndir O, Salif Sow P, Mboup S, Wirth DF: In vivo and in vitro analysis of chloroquine resistance in Plasmodium falciparum isolates from Senegal. Parasitol Res. 2005, 97: 136-140. 10.1007/s00436-005-1406-7.
Sarr O, Ahouidi AD, Ly O, Daily JP, Ndiaye D, Ndir O, Mboup S, Wirth DF: Mutations in PfCRT K76T do no correlate with sulfadoxine-pyrimethamine-amodiaquine failure in Pikine. Parasitol Res. 2008, 103: 765-769. 10.1007/s00436-008-1038-9.
Bertin G, Ndam NT, Jafari-Guemouri S, Fievet N, Renart E, Sow S, Le Hesran JY, Deloron P: High prevalence of Plasmodium falciparum pfcrt K76T mutation in pregnant women taking chloroquine prophylaxis in Senegal. J Antimicrob Chemother. 2005, 55: 788-791. 10.1093/jac/dki097.
Wurtz N, Fall B, Pascual A, Diawara S, Sow K, Baret E, Diatta B, Fall KB, Mbaye PS, Fall F, Diémé Y, Rogier C, Bercion R, Briolant S, Wade B, Pradines B: Prevalence of molecular markers of Plasmodium falciparum drug resistance in Dakar. Senegal. Malar J. 2012, 11: 197-10.1186/1475-2875-11-197.
Ndiaye M, Faye B, Tine R, Ndiaye JL, Lo A, Abiola A, Dieng Y, Ndiaye D, Hallett R, Alifrangis M, Gaye O: Assessment of the molecular marker of Plasmodium falciparum chloroquine resistance (Pfcrt) in Senegal after several years of chloroquine withdrawal. Am J Trop Med Hyg. 2012, 87: 640-645. 10.4269/ajtmh.2012.11-0709.
Ly O, Gueye PE, Deme AB, Dieng T, Badiane AS, Ahouidi AD, Diallo M, Bei AK, Wirth DF, Mboup S, Sarr O: Evolution of the pfcrt T76 and pfmdr1 Y86 markers and chloroquine susceptibility 8 years after cessation of chloroquine use in Pikine, Senegal. Parasitol Res. 2012, 111: 1541-1546. 10.1007/s00436-012-2994-7.
Gardella F, Assi S, Simon F, Bogreau H, Eggelte T, Ba F, Foumane V, Henry MC, Traore Kientega P, Basco L, Trape JF, Lalou R, Martelloni M, Desbordes M, Baragatti M, Briolant S, Almeras L, Pradines B, Fusai T, Rogier C: Antimalarial drug use in general populations of tropical Africa. Malar J. 2008, 7: 124-10.1186/1475-2875-7-124.
Frosch AEP, Venkatesan M, Laufer MK: Patterns of chloroquine use and resistance in sub-Saharan Africa: a systematic review of household survey and molecular data. Malar J. 2011, 10: 116-10.1186/1475-2875-10-116.
Ministère de la Santé et de la Prévention Médicale: Enquête nationale sur le paludisme au Sénégal 2006. 2007,http://www.measuredhs.com/pubs/pdf/MIS1/MIS1.pdf,
Ministère de la Santé et de la Prévention Médicale: Enquête nationale sur le paludisme au Sénégal 2008–2009. 2009,http://www.measuredhs.com/publications/publication-MIS5-MIS-Final-Reports.cfm,
Kublin JG, Cortese JF, Njunju EM, Mukadam RA, Wirima JJ, Kazembe PN, Djimdé AA, Kouriba B, Taylor TE, Plowe CV: Remergence of chloroquine-sensitive Plasmodium falciparum malaria after cessation of chloroquine in Malawi. J Infect Dis. 2003, 187: 1870-1875. 10.1086/375419.
Laufer MK, Thesing PC, Eddington ND, Masonga R, Dzinjalamala FK, Takala SL, Taylor TE, Plowe CV: Return of chloroquine antimalarial efficacy in Malawi. N Engl J Med. 2006, 355: 1959-1966. 10.1056/NEJMoa062032.
Noranate N, Durand R, Tall A, Marrama L, Spiegel A, Sokhna C, Pradines B, Cojean S, Guillotte M, Bischoff E, Ekala MT, Bouchier C, Fandeur T, Ariey F, Patarapotikul J, Le Bras J, Trape JF, Rogier C, Mercereau-Puijalon O: Rapid dissemination of Plasmodium falciparum drug resistance despite strictly controlled antimalarial use. Plos One. 2007, 1: 139-
Agnamey P, Brasseur P, de Pecoulas E, Vaillant M, Olliaro P: Plasmodium falciparum in vitro susceptibility to antimalarial drugs in Casamance (Southwestern Senegal) during the first 5 years of routine use of artesunate-amodiaquine. Antimicrob Agents Chemother. 2006, 50: 1531-1534. 10.1128/AAC.50.4.1531-1534.2006.
Holmgren G, Gil JP, Ferreira PM, Veiga MI, Obonyo CO, Bjorkman A: Amodiaquine resistant Plasmodium falciparum malaria in vivo is associated with selection of pfcrt 76T and pfmdr1 86Y. Infect Gen Evol. 2006, 6: 309-314. 10.1016/j.meegid.2005.09.001.
Nsobya SL, Dokomajilar C, Joloba M, Dorsey G, Rosenthal PJ: Resistance-mediating Plasmodium falciparum pfcrt and pfmdr1 alleles after treatment with artesunate-amodiaquine in Uganda. Antimicrob Agents Chemother. 2007, 51: 3023-3025. 10.1128/AAC.00012-07.
Echeverry DF, Holmgren G, Murillo C, Higuita JC, Gil JP, Osorio L: Polymorphisms in the pfcrt and pfmdr1 genes of Plasmodium falciparum and in vitro susceptibility to amodiaquine and desethylamodiaquine. Am J Trop Med Hyg. 2007, 77: 1034-1038.
Danquah I, Coulibaly B, Meissner P, Petruschke I, Muller O, Mockenhaupt FP: Selection of pfmdr1 and pfcrt alleles in amodiaquine treatment failure in north-western Burkina Faso. Acta Trop. 2010, 114: 63-33. 10.1016/j.actatropica.2009.12.008.
Brasseur P, Guiguemde R, Diallo S, Guiyedi V, Kombila M, Ringwald P, Olliaro P: Amodiaquine remains effective for treating ucomplicated malaria in West and Central Africa. Trans R Soc Trop Med Hyg. 1999, 93: 645-650. 10.1016/S0035-9203(99)90083-4.
Ndiaye JL, Faye B, Diouf AM, Kuété T, Cisse M, Seck PA, Brasseur P, Same-Ekobo A, Lameyre V, Gaye O: Randomized, comparative study of the efficacy and safety of artesunate plus amodiaquine, administered as a single daily intake versus two daily intakes in the treatment of uncomplicated falciparum malaria. Malar J. 2008, 7: 16-10.1186/1475-2875-7-16.
Ndiaye JL, Randrianarivelojosia M, Sagara I, Brasseur P, Ndiaye I, Faye B, Randrianasolo L, Ratsimbasoa A, Forlemu D, Ama Moor V, Traore A, Dicko Y, Dara N, Lameyre V, Diallo M, Djimdé A, Same-Ekobo A, Gaye O: Randomized, multicentre assessment of the efficacy and safety of ASAQ – a fixed dose artesunate-amodiaquine combination therapy in the treatment of uncomplicated Plasmodium falciparum malaria. Malar J. 2009, 8: 125-10.1186/1475-2875-8-125.
Dondorp AM, Nosten F, Yi P, Das D, Phyo AP, Tarning J, Lwin KM, Ariey F, Hanpithakpong W, Lee SJ, Ringwald P, Silamut K, Imwrong M, Chotivanish K, Lim P, Herdman T, An SS, Yeung S, Singhasivanon P, Day NPJ, Lindegardh N, Socheat D, White NJ: Artemisinin resistance in Plasmodium falciparum malaria. N Engl J Med. 2009, 361: 455-467. 10.1056/NEJMoa0808859.
Noedl H, Se Y, Schaecher K, Smith BL, Socheat D, Fukuda MM: Evidence of artemisinin-resistant malaria in western Cambodia. N Engl J Med. 2008, 359: 2619-2620. 10.1056/NEJMc0805011.
Sibley CH, Barnes KI, Plowe CV: The rationale and plan for creating a World Antimalarial Resistance Network (WARN). Malar J. 2007, 6: 118-10.1186/1475-2875-6-118.
Sibley CH, Barnes KI, Watkins WM, Plowe CV: A network to monitor antimalarial drug resistance: a plan for moving forward. Trends Parasitol. 2008, 24: 43-48. 10.1016/j.pt.2007.09.008.
Pradines B, Tall A, Fusai T, Spiegel A, Hienne R, Rogier C, Trape JF, Le Bras J, Parzy D: In vitro activities of benflumetol against 158 Senegalese isolates of Plasmodium falciparum in comparison with those of standard antimalarial drugs. Antimicrob Agents Chemother. 1999, 43: 418-420.
Brandicourt O, Druilhe P, Diouf F, Brasseur P, Turk P, Danis M: Decreased sensitivity to chloroquine and quinine of some Plasmodium falciparum strains from Senegal in September 1984. Am J Trop Med Hyg. 1986, 35: 717-721.
Jelinek T, Schelbert P, Loscher T, Eichenlaub D: Quinine resistant falciparum malaria acquired in east Africa. Trop Med Parasitol. 1995, 46: 38-40.
Palmieri F, Petrosillo N, Paglia MG, Conte A, Goletti D, Pucillo LP, Menegon M, Sannella A, Severini C, Majori G: Genetic confirmation of quinine-resistant Plasmodium falciparum malaria followed by postmalaria neurological syndrome in a traveler from Mozambique. J Clin Microbiol. 2004, 42: 5424-5426. 10.1128/JCM.42.11.5424-5426.2004.
Pradines B, Pistone T, Ezzedine K, Briolant S, Bertaux L, Receveur MC, Parzy D, Millet P, Rogier C, Malvy D: Quinine-resistant malaria in traveler returning from Senegal, 2007. Emerg Infect Dis. 2010, 16: 546-548. 10.3201/eid1603.091669.
Pradines B, Spiegel A, Rogier C, Tall A, Mosnier J, Fusai T, Trape JF, Parzy D: Antibiotics for prophylaxis of Plasmodium falciparum infections: in vitro activity of doxycycline against Senegalese isolates. Am J Trop Med Hyg. 2000, 62: 82-85.
Briolant S, Baragatti M, Parola P, Simon F, Tall A, Sokhna C, Hovette P, Mabika Mamfoumbi M, Koeck JL, Delmont J, Spiegel A, Castello J, Gardair JP, Trape JF, Kombila M, Minodier P, Fusai T, Rogier C, Pradines B: Multinormal in vitro distribution model suitable for the distribution of Plasmodium falciparum chemosusceptibility to doxycycline. Antimicrob Agents Chemother. 2009, 53: 688-695. 10.1128/AAC.00546-08.
Pradines B, Rogier C, Fusai T, Mosnier J, Daries W, Baret E, Parzy D: In vitro activities of antibiotics against Plasmodium falciparum are inhibited by iron. Antimicrob Agents Chemother. 2001, 45: 1746-1750. 10.1128/AAC.45.6.1746-1750.2001.
Ndiaye D, Dailly JP, Sarr O, Ndir O, Gaye O, Mboup S, Wirth DF: Mutations in Plasmodium falciparum dihydrofolate reductase and dihydropteroate synthase genes in Senegal. Trop Med Int Health. 2005, 10: 1176-1179. 10.1111/j.1365-3156.2005.01506.x.
Cissé B, Cairns M, Faye E, NDiaye O, Faye B, Cames C, Cheng Y, NDiaye M, Colle Lo A, Simondon K, Trape JF, Faye O, NDiaye JL, Gaye O, Greenwood B, Milligan P: Randomized trial of piperaquine with sulfadoxine-pyrimethamine or dihydroartemisinin for malaria intermittent preventive treatment in children. Plos One. 2009, 4: 7164-10.1371/journal.pone.0007164.
Faye B, Ndiaye M, Ndiaye JL, Annie A, Tine RC, Collé Lo A, Ndiaye M, Sow D, De Sousa A, Gaye O: Prevalence of molecular markers of Plasmodium falciparum resistance to sulfadoxine-pyrimethamine during the intermittent preventive treatment in infants coupled with expanded program immunization in Senegal. Parasitol Res. 2011, 109: 133-138. 10.1007/s00436-010-2236-9.
Sokhna C, Cissé B, Ba EH, Milligan P, Hallett R, Sutherland C, Gaye O, Boulanger D, Simondon K, Simondon F, Targett G, Lines J, Greenwood B, Trape JF: A trial of the efficacy, safety and impact on drug resistance of four drug regimens for seasonal intermittent preventive treatment for malaria in Senegalese children. Plos One. 2008, 3: 1471-10.1371/journal.pone.0001471.
Cissé B, Sokhna C, Boulanger D, Milet J, Ba EH, Richardson K, Hallett R, Sutherland C, Simondon K, Simondon F, Alexander N, Gaye O, Targett G, Lines J, Greenwood B, Trappe JF: Seasonal intermittent preventive treatment with artesunate and sulfadoxine-pyrimethamine for prevention of malaria in Senegalese children: a randomized, placebo-controlled, double-blind trial. Lancet. 2006, 367: 659-667. 10.1016/S0140-6736(06)68264-0.
Acknowledgements
The authors thank the patients of the Centre de santé Elizabeth Diouf, Ndeye Fatou Diop and Maurice Gomis from the Hôpital Principal de Dakar, Marie-Louise Senghor, Fatou Bintou Badji, Joseph Faye and Baba Diakhaby from Institut Pasteur de Dakar and Thiaba Sene Ndour and Fatou Seck from the Centre de Santé Elizabeth Diouf for technical support.
This work was supported by the Etat Major des Armées Françaises (grant schéma directeur paludisme LR 607).
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
BF, EB, YD, RB and BP carried out the in vitro testing of drug susceptibility. AP and NW carried out the molecular genetic studies. FDS, VR and AT supervised, carried out and coordinated the field collection of isolates from patients. RB, BW and BP conceived and coordinated the study. BP and SB analysed the data. BF, AP, NW, SB and BP drafted the manuscript. All authors read and approved the final manuscript.
Bécaye Fall, Aurélie Pascual contributed equally to this work.
Rights and permissions
Open Access This article is published under license to BioMed Central Ltd. This is an Open Access article is distributed under the terms of the Creative Commons Attribution License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Fall, B., Pascual, A., Sarr, F.D. et al. Plasmodium falciparum susceptibility to anti-malarial drugs in Dakar, Senegal, in 2010: an ex vivo and drug resistance molecular markers study. Malar J 12, 107 (2013). https://doi.org/10.1186/1475-2875-12-107
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1475-2875-12-107