Abstract
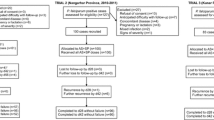
In 2003, the high level of chloroquine (CQ) treatment failure for uncomplicated Plasmodium falciparum malaria cases has led Senegal to adopt a new combination therapy with sulfadoxine–pyrimethamine and amodiaquine (SP-AQ). From September through November 2004, we used the 14-day World Health Organization follow-up protocol to assess the therapeutic response in patients with uncomplicated P. falciparum malaria in an area of high prevalence of pfcrt T76 mutant allele and SP resistance mutations. Of the 82 patients who were recruited, 68 (82.9%) completed follow-up. The response of the patients to treatment was adequate clinical response for 63 out of 68 patients (92.6%), while five (7.4%) clinical failures were recorded, four early treatment failures, and one late treatment failure. The prevalence of the pfcrt T76 allele at day 0 was 59.5%. The two-sided Fisher’s exact test did not show an association between pfcrt T76 allele and treatment failure (p = 0.167). The transitory treatment is effective and safe. However, the presence of high levels of mutant alleles points out the need to closely monitor the new therapeutic regimen.
Similar content being viewed by others
References
Basco LK, Same-ekobo A, Ngane VF, Ndounga M, Metoh T, Ringwald P, Soula G (2002) Therapeutic efficacy of sulfadoxine–pyrimethamine, amodiaquine and the sulfadoxine–pyrimethamine–amodiaquine combination against uncomplicated Plasmodium falciparum malaria in young children in Cameroon. Bull World Health Organ 80:538–545
Bertin G, Nicaise TN, Sayeh JG, Fievet N, Renart E, Sow S, Le Hesrant JY, Deloron P (2005) High prevalence of pfcrt K76T mutation in pregnant women taking chloroquine as prophylaxis in Senegal. J Antimicrob Chemother 55:788–791
Botella DMJ, Valls FJM, Martinez PML, Espacio CA (1991) Plasmodium falciparum resistant to sulfadoxine/pyrimethamine in Senegal. An Med Interna 8:79–81
Brasseur P, Diallo S, Guiguemde R, Guiyedi V, Kombila M, Ringwald P, Olliaro P (1999) Amodiaquine remains effective for treating uncomplicated malaria in West and Central Africa. Trans R Soc Trop Med Hyg 93:645–650
Djimde A, Doumbo OK, Cortese JF, Kayentao K, Doumbo S, Diourte Y, Dicko A, Su XZ, Nomura T, Fidock DA, Wellems TE, Plowe CV, Coulibaly D (2001a) A molecular marker of chloroquine-resistant falciparum malaria. N Engl J Med 344:257–263
Faye B, Ndiaye JL, Ndiaye D, Dieng Y, Faye O, Gaye O (2007) Efficacy and tolerability of four antimalarial combinations in the treatment of uncomplicated falciparum malaria in Senegal. Malar J 6:80
Gaye O, Soumare M, Sambou B, Faye O, Dieng Y, Diouf M, Bah IB, Dieng T, Ndir O, Diallo S (1999) Heterogeneity of chloroquine resistant malaria in Senegal. Bull Soc Pathol Exot 92:149–152
Graupner J, Goebels K, Grobusch MP, Lund A, Richter J, Haussinger D (2005) Efficacy of amodiaquine in uncomplicated falciparum malaria in Nigeria in an area with high-level resistance to chloroquine and sulfadoxine/pyrimethamine. Parasitol Res 96:162–165
Kublin JG, Cortese JF, Njunju EM, Mukadam RAG, Wirima JJ, Kaezembe PN, Djimde AA, Kouriba B, Taylor TE, Plowe CV (2003) Reemergence of chloroquine-sensitive Plasmodium falciparum malaria after cessation of chloroquine use in Malawi. J Infect Dis 187:1870–1875
MacIntosh HM (2000) Chloroquine or amodiaquine combined with sulfadoxine–pyrimethamine for treating uncomplicated malaria. Cochrane Database System Review 2, CD000386
Menard D, Madji N, Manirakiza A, Djalle D, Koula MR, Talarmin A (2005) Efficacy of chloroquine, amodiaquine, and sulfadoxine–pyrimethamine, chloroquine–sulfadoxine–pyrimethamine combination, amodiaquine–sulfadoxine–pyrimethamine combination in Central African children with noncomplicated malaria. Am J Trop Med Hyg 72:581–585
Mita T, Kaneko A, Lum JK, Bwijo B, Takechi N, Zungu IL, Tsukahara T, Tanabe K, Kobayaka T, Bjorkman A (2003) Recovery of chloroquine sensitivity and low prevalence of the Plasmodium falciparum chloroquine resistance transporter gene mutation K76T following the discontinuance use of chloroquine in Malawi. Am J Trop Med Hyg 68:413–415
Ndiaye D, Daily JP, Sarr O, Ndir O, Gaye O, Mboup S, Wirth DF (2005) Mutations in Plasmodium falciparum dihydrofolate reductase and dihydropteroate synthase genes in Senegal. Trop Med Int Health 10:1176–1179
Ochong EO, Van Den Broek IVF, Keus K, Nzila A (2003) Short report: association between chloroquine and amodiaquine resistance and allelic variation in the Plasmodium falciparum multiple drug resistance 1 gene and the chloroquine resistance transporter gene in isolates from the upper Nile in Southern Sudan. Am J Trop Med Hyg 69:184–187
Olliaro P, Mussano P (2002) Amodiaquine for treating malaria (Cochrane Review). The Cochrane Library Issue 3. Oxford: Update Software
Robert V, Awono-Ambene HP, Le Hesran JY, Trape JF (2000) Gametocytemia and infectivity to mosquitoes of patients with uncomplicated Plasmodium falciparum malaria attacks treated with chloroquine or sulfadoxine plus pyrimethamine. Am J Trop Med Hyg 62:210–216
Sarr O, Myrick A, Daily JP, Diop BM, Dieng T, Ndir O, Sow PS, Mboup S, Wirth DF (2005) In vivo and in vitro analysis of chloroquine resistance in Plasmodium falciparum isolates from Senegal. Parasitol Res 97:136–140
Sokhna CS, Trape JF, Robert V (2001) Gametocytaemia in Senegalese children with uncomplicated falciparum malaria treated with chloroquine, amodiaquine or sulfadoxine plus pyrimethamine. Parasite 8:243–250
Thomas SM, Dieng T, Ndir O, Mboup S, Wirth DF (2002) In vitro chloroquine susceptibility and PCR analysis of pfcrt and pfmdr1 polymorphisms in Plasmodium falciparum isolates from Senegal. Am J Trop Med Hyg 66:474–480
Trape JF, Pison G, Preziosi MP, Enel C, Desgrees DLA, Delauney V, Samb B, Lagarde E, Molez JF, Simondon F (1998) Impact of chloroquine resistance on malaria mortality. C R Acad Sci III 321:689–697
Viriyakosol S, Siripoon N, Petcharapirat C, Petcharapiart P, Jarra W, Thaithong S, Brown KN, Snounou G (1995) Genotyping of Plasmodium falciparum isolates by the polymerase chain reaction and potential use in epidemiologic studies. Bull World Health Organ 73:85–95
Wang X, Jianbing M, Guoqiao L, Peiqan C, Xingbo G, Linchun F, Chen L, Xinzhuan X, Wellems TE (2005) Decrease resistance of the Plasmodium falciparum chloroquine resistance transporter 76T marker associated with cessation of chloroquine use against P. falciparum malaria in Hainan, People’s Republic of China. Am J Trop Med Hyg 72:410–414
Warhust DC (2003) Polymorphisms in the plasmodium falciparum resistance transported protein links verapamil enhancement of chloroquine sensitivity with the clinical efficacy of amodiaquine. Malar J 2:31
WHO (1996) Vol. WHO/MAL/96.1077 World Health Organization, Geneva
WHO (2006) Vol. WHO/MAL/HTM/ 2006.1108 World Health Organization, Geneva
Zongo I, Dorsey G, Noel R, Dokomajilar C, Lankoande M, Ouedraogo JB, Rosenthal P (2005) Amodiaquine, sulfadoxine–pyrimethamine, and combination therapy for uncomplicated falciparum malaria: a randomized controlled trial from Burkina Faso. Am J Trop Med Hyg 73:826–832
Acknowledgments
We thank the patients from whom blood samples were drawn and the health workers at Pikine for their assistance. Dr. Danny Milner is thanked for his help in reviewing the manuscript. Ousmane Diouf has helped in data analysis. The work was supported by a National Institutes of Health grant 5D43TW001503-09 to Dyann F. Wirth.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarr, O., Ahouidi, A.D., Ly, O. et al. Mutations in PFCRT K76T do not correlate with sulfadoxine–pyrimethamine–amodiaquine failure in Pikine, Senegal. Parasitol Res 103, 765–769 (2008). https://doi.org/10.1007/s00436-008-1038-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-008-1038-9