Abstract
Background:
This study investigates the effects of a neuropeptide, secretoneurin (SN), on bone regeneration in an experimental mouse model.
Methods:
The effects of SN on cell proliferation, osteoblast marker genes expression, and mineralization were evaluated using the CCK-8 assay, quantitative reverse transcriptase polymerase chain reaction (RT-PCR), and alizarin red S staining, respectively. To examine the effects of SN on bone regeneration in vivo, bone defects were created in the calvaria of ICR mice, and 0.5 or 1 µg/ml SN was applied. New bone formation was analyzed by micro-computed tomography (micro-CT) and histology. New blood vessel formation was assessed by CD34 immunohistochemistry.
Results:
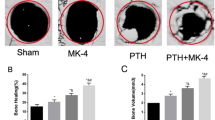
SN had no significant effect on proliferation and mineralization of MC3T3-E1 cells. However, SN partially induced the gene expression of osteoblast differentiation markers such as runt-related transcription factor 2, alkaline phosphatase, collagen type I alpha 1, and osteopontin. A significant increase of bone regeneration was observed in SN treated calvarial defects. The bone volume (BV), BV/tissue volume, trabecular thickness and trabecular number values were significantly increased in the collagen sponge plus 0.5 or 1 µg/ml SN group (p < 0.01) compared with the control group. Histologic analysis also revealed increased new bone formation in the SN-treated groups. Immunohistochemical staining of CD34 showed that the SN-treated groups contained more blood vessels compared with control in the calvarial defect area.
Conclusion:
SN increases new bone and blood vessel formation in a calvarial defect site. This study suggests that SN may enhance new bone formation through its potent angiogenic activity.






Similar content being viewed by others
References
Wirth T, Ylä-Herttuala S. Gene transfer vectors DNA vehicles and their incorporation into biomaterials for bone repair. In: Dubruel P, Van Vlierberghe S, editors. Biomaterials for Bone Regeneration. Novel Techniques and Applications. Elsvier; 2014. p.374–405.
Ghiasi MS, Chen J, Vaziri A, Rodriguez EK, Nazarian A. Bone fracture healing in mechanobiological modeling: a review of principles and methods. Bone Rep. 2017;6:87–100.
Carmeliet P. Angiogenesis in life, disease and medicine. Nature. 2005;438:932–6.
Vartanian KB, Chen HY, Kennedy J, Beck SK, Ryaby JT, Wang H, et al. The non-proteolytically active thrombin peptide TP508 stimulates angiogenic sprouting. J Cell Physiol. 2006;206:175–80.
Carano RA, Filvaroff EH. Angiogenesis and bone repair. Drug Discov Today. 2003;8:980–9.
Stegen S, van Gastel N, Carmeliet G. Bringing new life to damaged bone: the importance of angiogenesis in bone repair and regeneration. Bone. 2015;70:19–27.
Hankenson KD, Dishowitz M, Gray C, Schenker M. Angiogenesis in bone regeneration. Injury. 2011;42:556–61.
Glowacki J. Angiogenesis in fracture repair. Clin Orthop Relat Res. 1998;355:S82–9.
Hausman MR, Schaffler MB, Majeska RJ. Prevention of fracture healing in rats by an inhibitor of angiogenesis. Bone. 2001;29:560–4.
Shibuya M. Vascular endothelial growth factor (VEGF) and its receptor (VEGFR) signaling in angiogenesis: a crucial target for anti- and pro-angiogenic therapies. Genes Cancer. 2011;2:1097–105.
Ji C, Bi L, Li J, Fan J. Salvianolic acid B-loaded chitosan/hydroxyapatite scaffolds promotes the repair of segmental bone defect by angiogenesis and osteogenesis. Int J Nanomedicine. 2019;14:8271–84.
Dashnyam K, Buitrago JO, Bold T, Mandakhbayar N, Perez RA, Knowles JC, et al. Angiogenesis-promoted bone repair with silicate-shelled hydrogel fiber scaffolds. Biomater Sci. 2019;7:5221–31.
Kirchmair R, Hogue-Angeletti R, Gutierrez J, Fischer-Colbrie R, Winkler H. Secretoneurin-a neuropeptide generated in brain, adrenal medulla and other endocrine tissues by proteolytic processing of secretogranin II (chromogranin C). Neuroscience. 1993;53:359–65.
Kirchmair R, Marksteiner J, Troger J, Mahata SK, Mahata M, Donnerer J, et al. Human and rat primary C-fibre afferents store and release secretoneurin, a novel neuropeptide. Eur J Neurosci. 1994;6:861–8.
Li JY, Leitner B, Lovisetti-Scamihorn P, Winkler H, Dahlström A. Proteolytic processing, axonal transport and differential distribution of chromogranins A and B, and secretogranin II (secretoneurin) in rat sciatic nerve and spinal cord. Eur J Neurosci. 1999;11:528–44.
Fischer-Colbrie R, Laslop A, Kirchmair R. Secretogranin II: molecular properties, regulation of biosynthesis and processing to the neuropeptide secretoneurin. Prog Neurobiol. 1995;46:49–70.
Albrecht-Schgoer K, Schgoer W, Theurl M, Stanzl U, Lener D, Dejaco D, et al. Topical secretoneurin gene therapy accelerates diabetic wound healing by interaction between heparan-sulfate proteoglycans and basic FGF. Angiogenesis. 2014;17:27–36.
Kirchmair R, Gander R, Egger M, Hanley A, Silver M, Ritsch A, et al. The neuropeptide secretoneurin acts as a direct angiogenic cytokine in vitro and in vivo. Circulation. 2004;109:777–83.
Kirchmair R, Egger M, Walter DH, Eisterer W, Niederwanger A, Woell E, et al. Secretoneurin, an angiogenic neuropeptide, induces postnatal vasculogenesis. Circulation. 2004;110:1121–7.
Lee J, Song M, Kim J, Park Y. Comparison of angiogenic activities of three neuropeptides, substance P, Secretoneurin, and neuropeptide Y using myocardial infarction. Tissue Eng Regen Med. 2018;15:493–502.
Shyu WC, Lin SZ, Chiang MF, Chen DC, Su CY, Wang HJ, et al. Secretoneurin promotes neuroprotection and neuronal plasticity via the Jak2/Stat3 pathway in murine models of stroke. J Clin Invest. 2008;118:133–48.
El Bialy I, Jiskoot W, Reza Nejadnik M. Formulation, delivery and stability of bone morphogenetic proteins for effective bone regeneration. Pharm Res. 2017;34:1152–70.
Lim S, Kim JA, Lee T, Lee D, Nam SH, Lim J, et al. Stimulatory effects of KPR-A148 on osteoblast differentiation and bone regeneration. Tissue Eng Regen Med. 2019;16:405–13.
Bae EB, Kim HJ, Ahn JJ, Bae HY, Kim HJ, Huh JB. Comparison of bone regeneration between porcine-derived and bovine-derived xenografts in rat calvarial defects: a non-inferiority study. Materials (Basel). 2019;12:3412.
Liao H, Zhong Z, Liu Z, Li L, Ling Z, Zou X. Bone mesenchymal stem cells co-expressing VEGF and BMP-6 genes to combat avascular necrosis of the femoral head. Exp Ther Med. 2018;15:954–62.
Zou D, Zhang Z, Ye D, Tang A, Deng L, Han W, et al. Repair of critical-sized rat calvarial defects using genetically engineered bone marrow-derived mesenchymal stem cells overexpressing hypoxia-inducible factor-1alpha. Stem Cells. 2011;29:1380–90.
Liu W, Wang F, Zhao M, Fan Y, Cai W, Luo M. The neuropeptide secretoneurin exerts a direct effect on arteriogenesis in vivo and in vitro. Anat Rec (Hoboken). 2018;301:1917–27.
Grosso A, Burger MG, Lunger A, Schaefer DJ, Banfi A, Di Maggio N. It takes two to tango: coupling of angiogenesis and osteogenesis for bone regeneration. Front Bioeng Biotechnol. 2017;5:68.
Kaigler D, Wang Z, Horger K, Mooney DJ, Krebsbach PH. VEGF scaffolds enhance angiogenesis and bone regeneration in irradiated osseous defects. J Bone Miner Res. 2006;21:735–44.
Ogilvie CM, Lu C, Marcucio R, Lee M, Thompson Z, Hu D, et al. Vascular endothelial growth factor improves bone repair in a murine nonunion model. Iowa Orthop J. 2012;32:90–4.
Saran U, Gemini Piperni S, Chatterjee S. Role of angiogenesis in bone repair. Arch Biochem Biophys. 2014;561:109–17.
Lian JB, Stein GS. Runx2/Cbfa1: a multifunctional regulator of bone formation. Curr Pharm Des. 2003;9:2677–85.
Park SS, Kim KA, Lee SY, Lim SS, Jeon YM, Lee JC. X-ray radiation at low doses stimulates differentiation and mineralization of mouse calvarial osteoblasts. BMB Rep. 2012;45:571–6.
Choi JY, Lee BH, Song KB, Park RW, Kim IS, Sohn KY, et al. Expression patterns of bone-related proteins during osteoblastic differentiation in MC3T3-E1 cells. J Cell Biochem. 1996;61:609–18.
Huang S, Xu L, Zhang Y, Sun Y, Li G. Systemic and local administration of allogeneic bone marrow-derived mesenchymal stem cells promotes fracture healing in rats. Cell Transplant. 2015;24:2643–55.
Otsuru S, Tamai K, Yamazaki T, Yoshikawa H, Kaneda Y. Bone marrow-derived osteoblast progenitor cells in circulating blood contribute to ectopic bone formation in mice. Biochem Biophys Res Commun. 2007;354:453–8.
Zhang YB, Wang L, Jia S, Du ZJ, Zhao YH, Liu YP, et al. Local injection of substance P increases bony formation during mandibular distraction osteogenesis in rats. Br J Oral Maxillofac Surg. 2014;52:697–702.
Shi L, Feng L, Zhu ML, Yang ZM, Wu TY, Xu J, et al. Vasoactive intestinal peptide stimulates bone marrow-mesenchymal stem cells osteogenesis differentiation by activating WNT/beta-catenin signaling pathway and promotes rat skull defect repair. Stem Cells Dev. 2020;29:655–66.
Wang L, Zhao R, Shi X, Wei T, Halloran BP, Clark DJ, et al. Substance P stimulates bone marrow stromal cell osteogenic activity, osteoclast differentiation, and resorption activity in vitro. Bone. 2009;45:309–20.
Mei G, Zou Z, Fu S, Xia L, Zhou J, Zhang Y, et al. Substance P activates the Wnt signal transduction pathway and enhances the differentiation of mouse preosteoblastic MC3T3-E1 cells. Int J Mol Sci. 2014;15:6224–40.
Kittaka M, Shiba H, Kajiya M, Fujita T, Iwata T, Rathvisal K, et al. The antimicrobial peptide LL37 promotes bone regeneration in a rat calvarial bone defect. Peptides. 2013;46:136–42.
Zhang W, Zhu C, Wu Y, Ye D, Wang S, Zou D, et al. VEGF and BMP-2 promote bone regeneration by facilitating bone marrow stem cell homing and differentiation. Eur Cell Mater. 2014;27:1–11.
Zhang C, Wang KZ, Qiang H, Tang YL, Li Q, Li M, et al. Angiopoiesis and bone regeneration via co-expression of the hVEGF and hBMP genes from an adeno-associated viral vector in vitro and in vivo. Acta Pharmacol Sin. 2010;31:821–30.
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean Government (MSIT) (NRF-2017R1A5A2015391), and the Bio & Medical Technology Development Program of the NRF, which was funded by the Korean Government (MSIT) (2017M3A9E4047244).
Author information
Authors and Affiliations
Contributions
F. A. and E. K. P. designed the research and wrote the manuscript; F. A., H. J. I., J. A. K., J. L., S. L., and S.‐H. N. performed the experiments and analyzed the results; F. A., H. J. I., Y. C. B. and E. K. P. analyzed the data and revised the manuscript; and E. K. P. supervised the project.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial conflicts of interest.
Ethical statement
Animal experiments were performed in accordance with the guidelines approved by Kyungpook National University (No. 2018–0153).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Assefa, F., Lim, J., Kim, JA. et al. Secretoneurin, a Neuropeptide, Enhances Bone Regeneration in a Mouse Calvarial Bone Defect Model. Tissue Eng Regen Med 18, 315–324 (2021). https://doi.org/10.1007/s13770-020-00304-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-020-00304-1