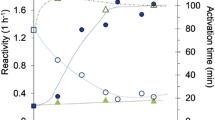
Abstract
Adsorption using porous carbon is an effective method to remove dyes from wastewater. The performance of the adsorbent depends on the pore structures; hence, selecting an optimal structural parameter for removal of a specific dye is required. This work studies the effect of pore structures on the methylene blue adsorption. Porous carbons featuring a similar surface area, but different pore characters, were synthesized, and the influences of their pore structure on the removal of the dye were assessed. The porous carbon was produced by pyrolysis of phenolic resin at 850 °C. To produce nanoporous carbon with different pore structures, the composition of resin precursors (resorcinol, phenol, ethylene glycol and formaldehyde) was altered. The pore- and microstructures of material were characterized by N2-sorption analysis, scanning electron microscopy and thermogravimetric analysis. An equal specific surface area of 1300 m2 g−1 was demonstrated, but it results in different pore properties (micropores or mesopores). Application performance of porous carbon was investigated for adsorption of methylene blue under ambient conditions. The experimental data (equilibrium and adsorption kinetic) were successfully described by Langmuir model and the pseudo-first order of adsorption rate. The adsorption parameters showed that carbon with mesoporous network is more beneficial.





Similar content being viewed by others
References
Aban Tanyildizi M (2011) Modeling of adsorption isotherms and kinetics of reactive dye from aqueous solution by peanut hull. Chem Eng J 168:1234–1240. https://doi.org/10.1016/j.cej.2011.02.021
Alver E, Metin AU (2012) Anionic dye removal from aqueous solutions using modified zeolite: adsorption kinetics and isotherm studies. Chem Eng J 200–202:59–67. https://doi.org/10.1016/j.cej.2012.06.038
Ariyanto T, Dyatkin B, Zhang G, Kern A, Gogotsi Y, Etzold BJM (2015) Synthesis of carbon core-shell pore structures and their performance as supercapacitors. Microporous Mesoporous Mater 218:130–136. https://doi.org/10.1016/j.micromeso.2015.07.007
Ariyanto T, Laziz AM, Gläsel J, Zhang G-R, Garbes J, Etzold BJM (2016) Producing high quality carbide-derived carbon from low quality byproducts stemming from SiC production. Chem Eng J 283:676–681. https://doi.org/10.1016/j.cej.2015.08.007
Ariyanto T, Kern A, Etzold BJM, Zhang G-R (2017a) Carbide-derived carbon with hollow core structure and its performance as catalyst support for methanol electro-oxidation. Electrochem Commun 82:12–15. https://doi.org/10.1016/j.elecom.2017.07.010
Ariyanto T, Zhang G-R, Riyahi F, Gläsel J, Etzold BJM (2017b) Controlled synthesis of core-shell carbide-derived carbons through in situ generated chlorine. Carbon 115:422–429. https://doi.org/10.1016/j.carbon.2017.01.032
Barbieri O, Hahn M, Herzog A, Kotz R (2005) Capacitance limits of high surface area activated carbons for double layer capacitors. Carbon 43:1303–1310. https://doi.org/10.1016/j.carbon.2005.01.001
Chiou MS, Chuang GS (2006) Competitive adsorption of dye metanil yellow and RB15 in acid solutions on chemically cross-linked chitosan beads. Chemosphere 62:731–740. https://doi.org/10.1016/j.chemosphere.2005.04.068
Crini G (2006) Non-conventional low-cost adsorbents for dye removal: a review. Bioresour Technol 97:1061–1085. https://doi.org/10.1016/j.biortech.2005.05.001
Glasel J, Diao JY, Feng ZB et al (2015) Mesoporous and graphitic carbide-derived carbons as selective and stable catalysts for the dehydrogenation reaction. Chem Mater 27:5719–5725. https://doi.org/10.1021/acs.chemmater.5b02262
Gor GY, Thommes M, Cychosz KA, Neimark AV (2012) Quenched solid density functional theory method for characterization of mesoporous carbons by nitrogen adsorption. Carbon 50:1583–1590. https://doi.org/10.1016/j.carbon.2011.11.037
Hameed BH, Ahmad AL, Latiff KNA (2007a) Adsorption of basic dye (methylene blue) onto activated carbon prepared from rattan sawdust. Dye Pigment 75:143–149. https://doi.org/10.1016/j.dyepig.2006.05.039
Hameed BH, Din ATM, Ahmad AL (2007b) Adsorption of methylene blue onto bamboo-based activated carbon: kinetics and equilibrium studies. J Hazard Mater 141:819–825. https://doi.org/10.1016/j.jhazmat.2006.07.049
Kannan N, Sundaram MM (2001) Kinetics and mechanism of removal of methylene blue by adsorption on various carbons—a comparative study. Dye Pigment 51:25–40. https://doi.org/10.1016/S0143-7208(01)00056-0
Li Y, Du Q, Liu T et al (2013) Comparative study of methylene blue dye adsorption onto activated carbon, graphene oxide, and carbon nanotubes. Chem Eng Res Des 91:361–368. https://doi.org/10.1016/j.cherd.2012.07.007
Lin JX, Zhan SL, Fang MH, Qian XQ, Yang H (2008) Adsorption of basic dye from aqueous solution onto fly ash. J Environ Manag 87:193–200. https://doi.org/10.1016/j.jenvman.2007.01.001
Pereira MFR, Soares SF, Olfao JJM, Figueiredo JL (2003) Adsorption of dyes on activated carbons: influence of surface chemical groups. Carbon 41:811–821. https://doi.org/10.1016/S0008-6223(02)00406-2
Perez CR, Yeon SH, Segalini J et al (2013) Structure and electrochemical performance of carbide-derived carbon nanopowders. Adv Funct Mater 23:1081–1089. https://doi.org/10.1002/adfm.201200695
Prasetyo I, Rochmadi, Ariyanto T, Yunanto R (2013) Simple method to produce nanoporous carbon for various applications by pyrolysis of specially synthesized phenolic resin. Indones J Chem 13:95–100. https://doi.org/10.22146/ijc.21290
Prasetyo I, Rochmadi, Wahyono E, Ariyanto T (2017) Controlling synthesis of polymer-derived carbon molecular sieve and its performance for CO2/CH4 separation. Eng J. https://doi.org/10.4186/ej.2017.21.4.83
Tahir SS, Rauf N (2006) Removal of a cationic dye from aqueous solutions by adsorption onto bentonite clay. Chemosphere 63:1842–1848. https://doi.org/10.1016/j.chemosphere.2005.10.033
Thommes M, Kaneko K, Neimark AV et al (2015) Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl Chem. https://doi.org/10.1515/pac-2014-1117
Wang S, Zhu ZH (2007) Effects of acidic treatment of activated carbons on dye adsorption. Dye Pigment 75:306–314. https://doi.org/10.1016/j.dyepig.2006.06.005
Wong YC, Szeto YS, Cheung WH, McKay G (2004) Adsorption of acid dyes on chitosan—equilibrium isotherm analyses. Process Biochem 39:693–702. https://doi.org/10.1016/S0032-9592(03)00152-3
Xia Y, Yao Q, Zhang W, Zhang Y, Zhao M (2015) Comparative adsorption of methylene blue by magnetic baker’s yeast and EDTAD-modified magnetic baker’s yeast: equilibrium and kinetic study. Arab J Chem. https://doi.org/10.1016/j.arabjc.2015.03.010
Zhang W, Zhou C, Zhou W et al (2011) Fast and considerable adsorption of methylene blue dye onto graphene oxide. Bull Environ Contam Toxicol 87:86–90. https://doi.org/10.1007/s00128-011-0304-1
Acknowledgements
This work was partially funded by the Ministry of Research, Technology and Higher Education of the Republic of Indonesia (MENRISTEKDIKTI) through Penelitian Unggulan Perguruan Tinggi (PUPT) Program (Grant No. 2483/UN1.P.III/DIT-LIT/LT/2017). The authors gratefully acknowledge the funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Editorial responsibility: M. Abbaspour.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ariyanto, T., Kurniasari, M., Laksmana, W.T. et al. Pore size control of polymer-derived carbon adsorbent and its application for dye removal. Int. J. Environ. Sci. Technol. 16, 4631–4636 (2019). https://doi.org/10.1007/s13762-018-2166-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-018-2166-0