Abstract
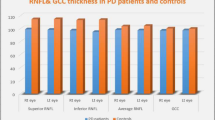
Optical coherence tomography (OCT) has been suggested as a method for detection of retinal alterations in neurodegenerative diseases. The usefulness of OCT as a diagnostic tool to differentiate Parkinson’s disease (PD) from other tremor diseases, remains unknown. We aimed to evaluate morphological changes of the retina in patients with PD, essential tremor (ET), essential tremor–Parkinson’s disease (ET–PD) using OCT. Forty-two eyes of 21 patients with PD, 24 eyes of 12 patients with ET, 24 eyes of 12 patients with ET–PD and 44 eyes of 22 age-matched healthy controls were included in the study. All participants underwent detailed neurological and ophthalmological examination. Measurements in all quadrants of macula and retinal nerve fiber layer (RNFL) thickness using OCT were recorded. There was no significant difference among the groups regarding age, sex. The average RNFL thickness was thinner in PD patients than that of ET (p = 0.032). The RNFL thickness in superior quadrant was lower in PD group compared with the ET and control group (p = 0.001, p = 0.016). Significant differences were observed in most of the macular thickness parameters excluding foveolar and foveal thickness (p = 0.865, 0.394). Correlations were found among several OCT parameters and disease duration or severity in all patient groups (p > 0.05). Retinal alterations were found in PD patients compared to ET. However, no significant retinal changes were detected by OCT in patients with ET and ET–PD compared to controls. According to our data, retinal assessments by OCT do not seem to be satisfactory for differentiation of these disorders.




Similar content being viewed by others
References
Thenganatt MA, Jankovic J (2016) The relationship between essential tremor and Parkinson’s disease. Parkinsonism Relat Disord 22:162–165
Fekete R, Jankovic J (2011) Revisiting the relationship between essential tremor and Parkinson’s disease. Mov Disord 26:391–398
Shahed J, Jankovic J (2007) Exploring the relationship between essential tremor and Parkinson’s disease. Parkinsonism Relat Disord 13(2):67–76
Minen MT, Louis ED (2008) Emergence of Parkinson’s disease in essential tremor: a study of the clinical correlates in 53 patients. Mov Disord 23(11):1602–1605
Rocca WA, Bower JH, Ahlskog JE et al (2007) Increased risk of essential tremor in first-degree relatives of patients with Parkinson’s disease. Mov Disord 22(11):1607–1614
Spanaki C, Plaitakis A (2009) Essential tremor in Parkinson’s disease kindreds from a population of similar genetic background. Mov Disord 24(11):1662–1668
Jankovic J (1989) Essential tremor and Parkinson’s disease. Ann Neurol 25(2):211–212
Iseri PK, Altinaş O, Tokay T, Yüksel N (2006) Relationship between cognitive impairment and retinal morphological and visual functional abnormalities in Alzheimer disease. J Neuroophthalmol 26(1):8–24
Moschos MM, Tagaris G, Markopoulos I et al (2011) Morphologic changes and functional retinal impairment in patients with Parkinson disease without visual loss. Eur J Ophthalmol 21(1):24–29
Archibald NK, Clarke MP, Mosimann UP, Burn DJ (2011) Retinal thickness in Parkinson’s disease. Parkinsonism Relat Disord 17(6):431–436
Witkovsky P (2004) Dopamine and retinal function. Doc Ophthalmol 108(1):7–40
Veys L, Vandenabeele M, Ortuño-Lizarán I et al (2019) Acta Neuropathol 37:379
Hajee ME, March WF, Lazzaro DR et al (2009) Inner retinal layer thinning in Parkinson disease. Arch Ophthalmol 127(6):737–741
Turkel Y, Ornek N, Dag E et al (2015) Retinal nerve fiber layer thickness in patients with essential tremor. Neurol Asia 20(4):363–366
Cubo E, Tedejo RP, Rodriguez Mendez V, Lopez Pena MJ, Trejo Gabriel Y, Galan JM (2010) Retina thickness in Parkinson’s disease and essential tremor. Mov Disord 25(14):2461–2462
Hoehn MM, Yahr MD (1967) Parkinsonism: onset, progression and mortality. Neurology 17:427–442
Fahn S, Elton R, Members of the updrs Development Committee (1987) In: Fahn S, Marsden CD, Calne DB, Goldstein M (eds) Recent developments in Parkinson’s disease. Macmillan Health Care Information, Florham park, pp 153–163, 293–304
Shrier EM, Adam CR, Spund B, Glazman S, Bodis-Wollner I (2012) Interocular asymmetry of foveal thickness in Parkinson disease. J Ophthalmol 2012:728457
Altintas O, Işeri P, Ozkan B, Caglar Y (2008) Correlation between retinal morphological and functional findings and clinical severity in Parkinson’s disease. Doc Ophthalmol 116(2):137–146
La Morgia C, Barboni P, Rizzo G et al (2013) Loss of temporal retinal nerve fibers in Parkinson disease: a mitochondrial pattern? Eur J Neurol 20(1):198–201
Aaker GD, Myung JS, Erlich JR, Mohammed M, Henchcliffe C, Kiss S (2010) Detection of retinal changes in Parkinson’s disease with spectral-domain optical coherence tomography. Clin Ophthalmol 4:1427–1432
Garcia-Martin E, Rodriguez-Mena D, Satue M et al (2014) Electrophysiology and optical coherence tomography to evaluate Parkinson disease severity. Investig Ophthalmol Vis Sci 55(2):696–705
Lee JY, Kim JM, Ahn J, Kim HJ, Jeon BS, Kim TW (2014) Retinal nerve fiber layer thickness and visual hallucinations in Parkinson’s disease. Mov Disord 29(1):61–67
Spund B, Ding Y, Liu T et al (2013) Remodeling of the fovea in Parkinson disease. J Neural Transm 120(5):745–753
La Morgia C, Di Vito L, Carelli V, Carbonelli M (2017) Patterns of retinal ganglion cell damage in neurodegenerative disorders: parvocellular vs magnocellular degeneration in optical coherence tomography studies. Front Neurol 22(8):710
Kolb H, Dekorver L (1991) Midget ganglion cells of the parafovea of the human retina: a study by electron microscopy and serial section reconstructions. J Comp Neurol 303(4):617–636
Garcia-Martin E, Larrosa JM, Polo V et al (2014) Distribution of retinal layer atrophy in patients with Parkinson disease and association with disease severity and duration. Am J Ophthalmol 157(2):470–478.e2
Kaur M, Saxena R, Singh D, Behari M, Sharma P, Menon V (2015) Correlation between structural and functional retinal changes in Parkinson disease. J Neuroophthalmol 35(3):254–258
Mailankody P, Battu R, Khanna A, Lenka A, Yadav R, Pal PK (2015) Optical coherence tomography as a tool to evaluate retinal changes in Parkinson’s disease. Parkinsonism Relat Disord 21(10):1164–1169
Acknowledgements
There are no potential conflicts of interest on financial support for each author.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tugcu, B., Melikov, A., Yildiz, G.B. et al. Evaluation of retinal alterations in Parkinson disease and tremor diseases. Acta Neurol Belg 120, 107–113 (2020). https://doi.org/10.1007/s13760-019-01228-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-019-01228-x