Abstract
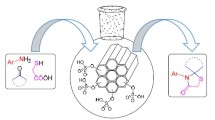
Silica-bonded ionic liquids is employed as a recyclable catalyst for the synthesis of spiro[indoline-3,4′-pyrano[2,3-c]pyrazoles] via one-pot condensation reaction of isatin, activated methylene reagents, and 3-methyl-l-phenyl-5-pyrazolone in refluxing aqueous medium in good to high yields. Also, these silica-bonded ionic liquids were used as catalysts for the synthesis of spiro[indoline-3,4′-pyrano[2,3-c]chromene derivatives in refluxing aqueous medium in high yields. Catalyst could be recycled for several times without any additional treatment.
Graphical Abstract
Silica-bonded ionic liquids is employed as a recyclable catalyst for the synthesis of spiro[indoline-3,4′-pyrano[2,3-c]pyrazoles] and spiro[indoline-3,4′-pyrano[2,3-c]chromene via one-pot condensation reaction of isatin, activated methylene reagents, and 3-methyl-l-phenyl-5-pyrazolone or 4-hydroxy coumarin respectively in refluxing aqueous medium in good to high yields.






Similar content being viewed by others
References
C.C. Galliford, K.A. Scheidt, Angew. Chem. 119, 8902–8912 (2007) [C.C. Galliford, K.A. Scheidt, Angew. Chem. Int. Ed. 46, 8748–8758 (2007)]
K. Ding, Y.P. Lu, N. Coleska, J. Med. Chem. 49, 3432–3435 (2006)
B. Yu, D.Q. Yu, H.M. Liu, Eur. J. Med. Chem. 97, 673–698 (2015)
J.P. Nandy, M. Prakesch, S. Khadem, P.T. Reddy, U. Sharma, P. Arya, Chem. Rev. 109, 1999–2060 (2009)
M. Pettersson, D. Knueppel, S.F. Martin, Org. Lett. 9, 4623–4626 (2007)
C. Marti, E.M. Carreira, Eur. J. Org. Chem. (12), 2209–2219 (2003)
C.V. Galliford, J.A. Martenson, C. Stern, K.A. Scheidt, Chem. Commun. (6), 631–633 (2007)
B. Liang, S. Kalidindi, J.A. Porco Jr, C.R.J. Stephenson, Org. Lett. 12, 572–575 (2010)
R.G. Redkin, L.A. Shemchuk, V.P. Chernykh, O.V. Shishkin, S.V. Shishkin, Tetrahedron 63, 11444–11450 (2007)
A. Dandia, R. Singh, P. Sarawgi, S. Khaturia, Chin. J. Chem. 24, 950–954 (2006)
S.L. Zhu, S.J. Ji, Y. Zhang, Tetrahedron 63, 9365–9372 (2007)
M.N. Elinson, A.S. Dorofeev, F.M. Miloserdov, G.I. Nikishin, Mol. Divers. 13, 47–52 (2009)
R. Sridhar, B. Srinivas, B. Madhav, V.P. Reddy, Y.V.D. Nageswar, K.R. Rao, Can. J. Chem. 87, 1704–1707 (2009)
A. Dandia, A.K. Jain, D.S. Bhati, Synth. Commun. 41, 2905–2919 (2011)
A. Dandia, V. Parewa, A.K. Jain, K.S. Rathore, Green Chem. 13, 2135–2145 (2011)
Y. Zou, Y. Hu, H. Liu, D. Shi, ACS Comb. Sci. 14, 38–43 (2012)
H.R. Safaei, M. Shekouhy, A. Shirinfeshan, S. Rahmanpur, Mol. Divers. 16, 669–683 (2012)
P. Saluja, K. Aggarwal, J.M. Khurana, Synth. Commun. 43, 3239–3246 (2013)
A. Thakur, M. Tripathi, U.C. Rajesh, D.S. Rawat, RSC Adv. 3, 18142–18148 (2013)
G. Mohammadi Ziarani, N. Hosseini Mohtasham, N. Lashgari, A. Badiei, M. Amanlou, R. Bazl, JNS 2, 489–500 (2013)
S.P. Satasia, P.N. Kalaria, J.R. Avalani, D.K. Raval, Tetrahedron 70, 5763–5767 (2014)
W. Liju, K. Ablajan, F. Jun, Ultrason. Sonochem. 22, 113–118 (2015)
K. Qiao, H. Hagiwara, C. Yokoyama, J. Mol. Catal. A: Chem. 246, 65–69 (2006)
R. Sugimura, K. Qiao, D. Tomida, C. Yokoyama, Catal. Commun. 8, 770–772 (2007)
A. Chrobok, S. Baj, W. Pudlo, A. Jarzebski, Appl. Catal. A Gen. 366, 22–28 (2009)
M.N. Sefat, D. Saberi, K. Niknam, Catal. Lett. 141, 1713–1720 (2011)
M. Baghernejad, K. Niknam, Int. J. Chem. 4, 52–60 (2012)
K. Niknam, A. Piran, Green Sustain. Chem. 3(2A), 1–8 (2013)
F. Shirini, M. Seddighi, M. Mazloumi, M. Makhsous, M. Abedini, J. Mol. Liq. 208, 291–297 (2015)
A.R. Moosavi-Zare, M.A. Zolfigol, M. Zarei, A. Zare, V. Khakyzadeh, J. Mol. Liq. 211, 373–380 (2015)
M. Seddighi, F. Shirini, M. Mamaghani, C. R. Chim. 18, 573–580 (2015)
K. Niknam, P. Abolpour, Monatsh. Chem. 146, 683–690 (2015)
K. Niknam, A. Jamali, M. Tajaddod, A. Deris, Chin. J. Catal. 33, 1312–1317 (2012)
K. Niknam, A. Jamali, Chin. J. Catal. 33, 1840–1849 (2012)
S.M.G. Ahmadi-Ana, M. Baghernejad, K. Niknam, Chin. J. Chem. 30, 517–521 (2012)
K. Niknam, N. Borazjani, R. Rashidian, A. Jamali, Chin. J. Catal. 34, 2245–2254 (2013)
K. Niknam, R. Rashidian, A. Jamali, Sci. Iran. C 20, 1863–1870 (2013)
K. Niknam, M. Sadeghi Habibabad, A. Deris, N. Aeinjamshid, Monatsh. Chem. 144, 987–992 (2013)
K. Niknam, S. Mojikhalifeh, Mol. Diver. 18, 111–117 (2014)
K. Niknam, N. Borazjani, Monatsh. Chem. (2015). doi:10.1007/s00706-015-1552-2
Acknowledgments
We are thankful to Persian Gulf University Research Council for partial support of this work. Also, we are thankful to the School of Chemistry, Manchester University for running NMRs.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Niknam, K., Piran, A. & Karimi, Z. Synthesis of spiro[indoline-3,4′-pyrano[2,3-c]pyrazole] and spiro[indoline-3,4′-pyrano[2,3-c]chromene] derivatives using silica-bonded ionic liquids as a recyclable catalyst in aqueous medium. J IRAN CHEM SOC 13, 859–871 (2016). https://doi.org/10.1007/s13738-015-0801-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-015-0801-y