Abstract
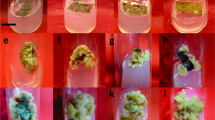
This study was performed to establish an improved and effective shoot culture system using a bioreactor for the commercial biomass production from whole plants of Vitis flexuosa. To determine the effect of different medium supplement systems on biomass and bioactive compound accumulation, in vitro shoots were cultured in several different medium supplement systems [solid culture (conventional method), liquid culture (control), continuous immersion bioreactor (CIB), temporary immersion bioreactor (TIB), CIB with net (CIB-N), and TIB with net (TIB-N)]. To investigate the optimal NH4 + and NO3 - ratio in medium, in vitro shoots were cultured with NH4 + and NO3 - ratios of 0:30, 10:20, 15:15, 20:10, and 30:0 mM in 1/2 MS medium. After 4 weeks of culture, the CIB system produced the highest biomass productivity (43.80 g·L-1 FW and 10.11% DW) but showed poor plant development. In comparison with CIB, the plantlet growth was improved in the TIB system in terms of plant height, leaf number, and root length. A greater quantity of total phenolics was also recorded in the TIB system as compared to the CIB system, whereas flavonoid content was similar in both systems. A 10:20 mM NH4 +:NO3 - ratio in the TIB system medium showed an increase of biomass (50.99 g·L-1 medium FW, 4.37 g·L-1 medium DW) and 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging activity (89.09%). Bioactive compound accumulation increased with increasing NO3 - concentration, with the highest levels being obtained from the 10:20 mM NH4 +:NO3 - ratio after 4 weeks of culture. After 7 days of elicitation with 100-μM methyl jasmonate, the increased percentage of bioactive compounds and DPPH radical scavenging activity was obtained under the 15:15 mM NH4 +:NO3 - ratio treatment. The present study indicated that the proper selection of a bioreactor system and manipulation of the NH4 + and NO3 - ratio, along with the elicitation of the shoot culture, led to the rapid production of sufficient biomass and bioactive compounds in V. flexuosa.
Similar content being viewed by others
Literature Cited
Ali, M.B., E.J. Hahn, and K.Y. Paek. 2007. Methyl jasmonate and salicylic acid induced oxidative stress and accumulation of phenolics in Panax ginseng bioreactor root suspension cultures. Molecules 12:607–621.
Ali, M.B., K.W. Wu, E.J. Hahn, and K.Y. Paek. 2006. Methyl jasmonate and salicylic acid elicitation induces ginsenosides accumulation, enzymatic and non-enzymatic antioxidant in suspension culture Panax ginseng roots in bioreactors. Plant Cell Rep. 25:613–620.
Bari, R. and D.G.J Jonathan. 2009. Role of plant hormones in plant defense responses. Plant Mol. Biol. 69:473–488.
Baque, M.A., S.H. Moh, E.J. Lee, J.J. Zhong, and K.Y. Paek. 2012. Production of biomass and useful compounds from adventitious roots of high-value added medicinal plants using bioreactor. Biotechnol. Adv. 30:1255–1267.
Chakrabarty, D., E.J. Hahn, Y.J. Yoon, and K.Y. Paek. 2003. Micropropagation of apple rootstock M.9 EMLA using bioreactor. J. Hortic. Sci. Biotechol. 78:605–609.
Chattopadhyay, S., R. Mehra, A. Srivastava, S. Bhojwani, and V. Bisaria. 2003. Effect of major nutrients on podophyllotoxin production in Podophyllum hexandrum suspension cultures. Appl. Microbiol. Biot. 60:541–546.
Choi, S.Y., H.S. Cho, and N.J. Sung. 2006. The antioxidative and nitrite scavenging ability of solvent extracts from wild grape (Vitis coignetiea) skin. J. Kor. Soc. Food Sci. Nutr. 35:961–966.
Cicco, N., M.T. Lanorte, M. Paraggio, M. Viggiano, and V. Lattanzio. 2009. A reproducible, rapid and inexpensive Folin-Ciocalteu micromethod in determining phenolics of plant methanol extracts. Microchem. J. 91:107–110.
Cui, X.H., H.N. Murthy, C.H. Wu, and K.Y. Paek. 2010. Adventitious root suspension cultures of Hypericum perforatum: effect of nitrogen source on production of biomass and secondary metabolites. In Vitro Cell. Dev. Biol. Plant. 46:437–444.
Dewir, Y.H., D. Chakrabarty, E.J. Hahn, and K.Y. Paek. 2005. Reversion of inflorescence in Euphorbia millii and its application to large scale micropropagation in an air-lift bioreactor. J. Hortic. Sci. Biotechol. 80:581–587.
Donnez, D., K.-H. Kim, S. Antoine, A. Conreux, V. De Luca, P. Jeandet, C. Clement, and E. Courot. 2011. Bioproduction of resveratrol and viniferins by an elicited grapevine cell culture in a 2 L stirred bioreactor. Proc. Biochem. 46:1056–1062.
Fazal, H., B.H. Abbasi, and N. Ahmed. 2014. Optimization of adventitious root culture for production of biomass and secondary metabolites in Prunella vulgaris L. Appl. Biochem. Biotechnol. 174:2086–2095.
Jeong, C.S., D. Chakrabarty, E.J. Hahn, H.L. Lee, and K.Y. Paek. 2005. Effects of oxygen, carbon dioxide and ethylene on growth and bioactive compound production in bioreactor culture of ginseng adventitious roots. Biochem. Engr. J. 27:252–263.
Kim, Y.S., E.J. Hahn, H.N. Murthy, and K.Y. Paek. 2004. Adventitious root growth and ginsenoside accumulation in Panax ginseng cultures as affected by methyl jasmonate. Biotechnol. Lett. 26:1619–1622.
Lee, E.J., S.Y. Park, and K.Y. Paek. 2015. Enhancement strategies of bioactive compound production in adventitious root cultures of Eleutherococcus koreanum Nakai subjected to methyl jasmonate and salicylic acid elicitation through airlift bioreactors. Plant Cell Tiss. Org. Cult. 120:1–10.
Losso, J.N., R.R. Bansode, A.I.I. Trappey, H.A. Bawadi, and A. Truax. 2004. In vitro anti-proliferative activities of ellagic acid. J. Nutr. Biochem. 15:672–678.
Murashige, T. and F. Skoog. 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497.
Murthy, H.N., S.D. Vijayalaxmi, J.J. Zhong, and K.Y. Paek. 2014a. Strategies for enhanced production of plant secondary metabolites from cell and organ cultures, p. 471–508. In: K.-Y. Paek et al. (eds.). Production of biomass and bioactive compounds using bioreactor technology. Springer, Dordrecht.
Murthy, H.N., M.I. Georgiev, Y.S. Kim, C.S. Jeong, S.J. Kim, S.Y. Park, and K.Y. Paek. 2014b. Ginsenosides: prospective for sustainable biotechnological Production. Appl. Microbiol. Biotechnol. 98:6243–6254.
Mukundan, U. and A.M. Hjortso. 1991. Growth and thiophene accumulation by hairy root cultures of Tagetes petula in media of varying initial pH. Plant Cell Rep. 9:627–630.
Paek, K.Y. and D. Chakrabarty. 2006. Application of bioreactor system for mass propagation of horticultural plants, p. 472–477. In: J.A. Teixeira da Silva (ed.). Floriculture, ornamental and plant biotechnology. Global Science Books, UK.
Paek, K.Y., D. Chakrabarty, and E.J. Hahn. 2005. Application of bioreactor system for large scale production of horticultural and medicinal plants, p. 95–116. In: A.K. Hvoslef-Eide and W. Preil (eds.). Liquid culture systems for in vitro plant propagation. Springer, New York.
Praveen, N. and H.N. Murthy. 2012. Synthesis of withanolide A depends on carbon source and medium pH in hairy root cultures of Withania somnifera. Ind. Crops Prod. 35:241–243.
Rajashekeran, T., G.A. Ravishankar, and L.V. Venkataraman. 1991. Influence of nutrient stress on pyrethrin production by cultured cells of pyrethrum (Chrysanthemum cinerariaefolium). Curr. Sci. 60:705–707.
Shim, S.W. 2002. Environmental conditions affecting in vitro growth and acclimatization of ‘5BB’ grape rootstock. PhD Diss., Chungbuk National University, Cheongju, Korea.
Shim, K.M., H.N. Murthy, S.Y. Park, I. Rusli, and K.Y. Paek. 2015. Production of biomass and bioactive compounds from cell suspension cultures of Eurycoma longifolia in balloon type bubble bioreactors. Hort. Environ. Biotechnol. 33:251–258.
Shohael, A.M., H.M. Murthy, E.J. Hahn, H.L. Lee, and K.Y. Paek. 2008. Increased eleutheroside production in Eleutherococcus sessiliflorus embryogenic suspension cultures with methyl jasmonate treatment. Biochem. Engr. J. 38:270–273.
Sivakumar, G., K.W. Yu, E.J. Hahn, and K.Y. Paek. 2005. Optimization of organic nutrients for ginseng hairy roots production in large-scale bioreactors. Curr. Sci. 89:641–649.
Talcott, S.T. and J.H. Lee. 2002. Ellagic acid and flavonoid antioxidant content of muscadine wine and juice. J. Agric. Food Chem. 50:3186–3192.
Tung, Y.T., K.C. Cheng, S.T. Ho, Y.L. Chen, T.L. Wu, K.C. Hung, and J.H. Wu. 2011. Comparison and characterization of the antioxidant potential of 3 wild grapes — Vitis thunbergii, V. flexuosa, and V. kelungeusis. J. Food Sci. 76:701–706.
Vauzour, D., K. Vafeiadou, G. Corona, S.E. Pollard, X. Tzounis, and J.P.E. Spencer. 2007. Champagne wine polyphenols protect primary cortical neurons against peroxynitrite-induced injury. J. Agric. Food Chem. 55:2854–2869.
Wu, C.H., Y.H. Dewir, E.J. Hahn, and K.Y. Paek. 2006. Optimization of culturing conditions for the production of biomass and phenolics from adventitious roots of Echinacea angustifolia. J. Plant Biol. 49:193–199.
Wu, S.Q., X.K. Yu, and M.L. Lian, S.Y. Park, and X.C. Piao. 2014. Several factors affecting hypericin production of Hypericum perforatum during adventitious root culture in airlift bioreactors. Acta Physiol. Plant. 36:975–981.
Yu, K.W., W.Y. Gao, E.J. Hahn, and K.Y. Paek. 2002. Jasmonic acid improves ginsenoside accumulation in adventitious root culture of Panax ginseng C.A. Meyer. Biochem. Engr. J. 11:211–215.
Zhao, J., C.D. Lawrence, and R. Verpoorte. 2005. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 23:283–333.
Ziv, M. 2005. Simple bioreactors for mass propagation of plants. Plant Cell Tiss. Org. Cult. 81:277–285.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, JA., Park, BJ., Kim, AH. et al. Airlift bioreactor system and nitrogen sources for biomass and antioxidant compound production from in vitro culture of Vitis flexuosa plantlets. Hortic. Environ. Biotechnol. 56, 358–365 (2015). https://doi.org/10.1007/s13580-015-0006-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-015-0006-4