Abstract
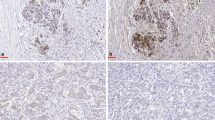
Metabolic diseases like diabetes and obesity are major risk factors for breast cancer. Aberrant expression of metabolic effectors such as fibroblast growth factor 19 (FGF19) could be therefore associated with the disease. The expression of FGF19 was examined in 193 archival breast tumor samples by immunohistochemistry and evaluated semi-quantitatively by determining the staining index and correlating it with clinicopathological parameters using Fisher's exact test. The correlation between FGF19 expression and 5-year disease-specific survival rate was determined using the univariate Kaplan–Meier analysis. The prognostic value of FGF19 expression was evaluated using the multivariate Cox regression analysis. Of the 193 tumors analyzed, 40 % were classified with low FGF19 expression, whereas 60 % were categorized as tumors with high FGF19 expression. There was a highly significant correlation between high FGF19 expression and patients' age (p = 0.008) as well as 5-year disease-specific survival (p = 0.001). However, FGF19 expression did not show any significant correlations with other clinicopathological parameters, including hormonal status, tumor grade, tumor size, or lymph node status. Univariate Kaplan–Meier log rank analysis showed that patients with high FGF19 expression exhibited a significantly shorter disease-specific 5-year survival (p = 0.007). This effect was exacerbated by lymph node metastasis (p = 0.001), negative estrogen receptor (ER) status (p = 0.002), or old age (p = 0.013). Multivariate analysis showed that high FGF19 expression could be an independent prognostic marker of disease-specific survival in breast cancer patients (p = 0.030). Quantification of FGF19 expression appears to provide valuable prognostic information in breast cancer, particularly in older patients with lymph node metastasis and negative ER status.


Similar content being viewed by others
References
Forouzanfar MH, Foreman KJ, Delossantos AM, et al. Breast and cervical cancer in 187 countries between 1980 and 2010: a systematic analysis. Lancet. 2011;378:1461–84.
Ermiah E, Abdalla F, Buhmeida A, Larbesh E, Pyrhonen S, Collan Y. Diagnosis delay in Libyan female breast cancer. BMC Research Notes. 2012;5:452.
Ikpatt F. Nigerian breast cancer: prognostic value of histomorphometry. Turku: Annales Universitatis Turkuensis 2002; 487, 128 pages
Missaoui N, Landolsi H, Jaidaine L, Ben Abdelkader A, Yaacoubi MT, Hmissa S. Breast cancer in central Tunisia: an earlier age at diagnosis and incidence increase over a 15-year period. Breast J. 2012;18:289–91.
Najjar H, Easson A. Age at diagnosis of breast cancer in Arab nations. Int J Surg (London, England). 2010;8:448–52.
Taghipour S, Banjevic D, Fernandes J, et al. Predictors of competing mortality to invasive breast cancer incidence in the Canadian National Breast Screening study. BMC Cancer. 2012;12:299.
Jung SP, Kim S, Nam SJ, Kim I, Bae JW. The role of the CDH1 promoter hypermethylation in the axillary lymph node metastasis and prognosis. J Breast Cancer. 2013;16:16–22.
Reyal F, Hajage D, Savignoni A, et al. Long-term prognostic performance of ki67 rate in early stage, p T1–pT2, pN0, invasive breast carcinoma. PLoS One. 2013;8:e55901.
Huang Y, Zhang H, Cai J, et al. Overexpression of MACC1 and its significance in human breast cancer progression. Cell Biosci. 2013;3:16.
Galea MH, Blamey RW, Elston CE, Ellis IO. The Nottingham Prognostic Index in primary breast cancer. Breast Cancer Res Treat. 1992;22:207–19.
Jiang L, Zhou Y, Wang Z, Lu X, Chen M, Zhou C. Is there different correlation with prognostic factors between “non-mass" and “mass” type invasive ductal breast cancers? Eur J Radiol. 2013;82(9):1404–9
Thomas F, Arriagada R, Mouriesse H, et al. Radical radiotherapy alone in non-operable breast cancer: the major impact of tumor size and histological grade on prognosis. Radiother Oncol: J Eur Soc Ther Radiol Oncol. 1988;13:267–76.
Visscher DW, Zarbo RJ, Greenawald KA, Crissman JD. Prognostic significance of morphological parameters and flow cytometric DNA analysis in carcinoma of the breast. Pathology Annual. 1990; 25 Pt 1: 171–210.
Metzger Filho O, Ignatiadis M, Sotiriou C. Genomic Grade Index: an important tool for assessing breast cancer tumor grade and prognosis. Crit Rev Oncol Hematol. 2011;77:20–9.
Pereira CB, Leal MF, de Souza CR, et al. Prognostic and predictive significance of MYC and KRAS alterations in breast cancer from women treated with neoadjuvant chemotherapy. PLoS One. 2013;8:e60576.
Zhou L, Luo Y, Li K, et al. Molecular markers of therapeutic resistance in breast cancer. Hum Pathol. 2013;44(7):1421–8.
Cicione C, Degirolamo C, Moschetta A. Emerging role of fibroblast growth factors 15/19 and 21 as metabolic integrators in the liver. Hepatology (Baltimore, Md). 2012;56:2404–11.
Lin BC, Desnoyers LR. FGF19 and cancer. Adv Exp Med Biol. 2012;728:183–94.
Wu X, Yan Q, Huang Y, et al. Isolation of a novel basic FGF-binding peptide with potent antiangiogenetic activity. J Cell Mol Med. 2010;14:351–6.
Nakayama Y, Miyake A, Nakagawa Y, et al. Fgf19 is required for zebrafish lens and retina development. Dev Biol. 2008;313:752–66.
Feng S, Dakhova O, Creighton CJ, Ittmann M. Endocrine fibroblast growth factor FGF19 promotes prostate cancer progression. Cancer Res. 2013;73:2551–62.
Sawey ET, Chanrion M, Cai C, et al. Identification of a therapeutic strategy targeting amplified FGF19 in liver cancer by oncogenomic screening. Cancer Cell. 2011;19:347–58.
Miura S, Mitsuhashi N, Shimizu H, et al. Fibroblast growth factor 19 expression correlates with tumor progression and poorer prognosis of hepatocellular carcinoma. BMC Cancer. 2012;12:56.
Ly M, Valent A, Diallo G, et al. Gene copy number variations in breast cancer of Sub-Saharan African women. Breast (Edinburgh, Scotland). 2013;22:295–300.
Nicholes K, Guillet S, Tomlinson E, et al. A mouse model of hepatocellular carcinoma: ectopic expression of fibroblast growth factor 19 in skeletal muscle of transgenic mice. Am J Pathol. 2002;160:2295–307.
Desnoyers LR, Pai R, Ferrando RE, et al. Targeting FGF19 inhibits tumor growth in colon cancer xenograft and FGF19 transgenic hepatocellular carcinoma models. Oncogene. 2008;27:85–97.
Wu X, Ge H, Gupte J, et al. Co-receptor requirements for fibroblast growth factor-19 signaling. J Biol Chem. 2007;282:29069–72.
Pai R, Dunlap D, Qing J, Mohtashemi I, Hotzel K, French DM. Inhibition of fibroblast growth factor 19 reduces tumor growth by modulating beta-catenin signaling. Cancer Res. 2008;68:5086–95.
Lin BC, Wang M, Blackmore C, Desnoyers LR. Liver-specific activities of FGF19 require Klotho beta. J Biol Chem. 2007;282:27277–84.
Kim J, Eskiocak U, Stadler G, et al. Short hairpin RNA screen indicates that Klotho beta/FGF19 protein overcomes stasis in human colonic epithelial cells. J Biol Chem. 2011;286:43294–300.
Rubinek T, Shulman M, Israeli S, et al. Epigenetic silencing of the tumor suppressor klotho in human breast cancer. Breast Cancer Res Treat. 2012;133:649–57.
Martin A, David V, Quarles LD. Regulation and function of the FGF23/klotho endocrine pathways. Physiol Rev. 2012;92:131–55.
Wu X, Lemon B, Li X, et al. C-terminal tail of FGF19 determines its specificity toward Klotho co-receptors. J Biol Chem. 2008;283:33304–9.
Lipponen P, Collan Y. Simple quantitation of immuno-histochemical staining positivity in microscopy. Acta Stereol. 1992;11:125–32.
Elzagheid A, Buhmeida A, Korkeila E, Collan Y, Syrjanen K, Pyrhonen S. Up-regulation of alpha-catenin is associated with increased lymph node involvement in colorectal cancer. World J Gastroenterol. 2008;14:4903–8.
Abdalla F, Boder J, Markus R, Hashmi H, Buhmeida A, Collan Y. Correlation of nuclear morphometry of breast cancer in histological sections with clinicopathological features and prognosis. Anticancer Res. 2009;29:1771–6.
Buhmeida A, Merdad A, Al-Maghrabi J, et al. RASSF1A methylation is predictive of poor prognosis in female breast cancer in a background of overall low methylation frequency. Anticancer Res. 2011;31:2975–81.
Ermiah E, Abdalla F, Buhmeida A, et al. Prognostic significance of DNA image cytometry in Libyan breast cancer. Oncology. 2012;83:165–76.
Ermiah E, Buhmeida A, Khaled BR, et al. Prognostic value of bcl-2 expression among women with breast cancer in Libya. Tumour biology: the Journal of the International Society for Oncodevelopmental Biology and Medicine 2013.
Pfannenberg C, Werner MK, Ripkens S, et al. Impact of age on the relationships of brown adipose tissue with sex and adiposity in humans. Diabetes. 2010;59:1789–93.
Feng B, Zhang T, Xu H. Human adipose dynamics and metabolic health. Ann N Y Acad Sci. 2013;1281:160–77.
French DM, Lin BC, Wang M, et al. Targeting FGFR4 inhibits hepatocellular carcinoma in preclinical mouse models. PLoS One. 2012;7:e36713.
Acknowledgments
The authors would like thank the Ministry of Higher Education (MOHE) and King Abdulaziz City for Science and Technology (KACST) for their financial support to this research (ARP-29-292). The authors also would like to thank Samira Dini, Basmat Abdullah, Noha Abozaid, and Saher Al-Hakami from CEGMR laboratory and Muneeb Ashoor and Kifah Abunar from Al-Jeel Company, the exclusive representative of Ventana in KSA, for their highly significant technical assistance.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Buhmeida, A., Dallol, A., Merdad, A. et al. High fibroblast growth factor 19 (FGF19) expression predicts worse prognosis in invasive ductal carcinoma of breast. Tumor Biol. 35, 2817–2824 (2014). https://doi.org/10.1007/s13277-013-1374-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-013-1374-y