Abstract
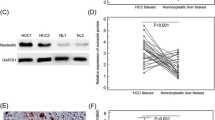
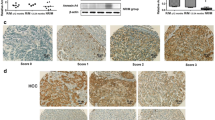
The aim of this study was to evaluate the association between activating enhancer binding protein 4 (AP-4) tissue expression and patient prognosis in hepatocellular carcinoma (HCC). The levels of AP-4 mRNA and protein in tumor and para-tumor tissue were evaluated in 30 HCC cases by quantitative real-time polymerase chain reaction (qRT-PCR) and Western blot. Additionally, AP-4 protein expression in 112 HCC was analyzed by immunohistochemistry. The correlation of AP-4 expression and patients’ clinicopathological parameters was evaluated. Survival analysis was performed using the Kaplan–Meier method and Cox’s proportional hazards model. By RT-PCR and Western blot, the levels of AP-4 mRNA and protein were significantly higher in HCC, compared to that in para-tumor tissue (p < 0.001). Immunohistochemical staining revealed that AP-4 was highly expressed in 53.6 % of the HCC patients. The AP-4 expression level was closely associated with serum alpha fetoprotein elevation, tumor size, histological differentiation, tumor recurrence, tumor metastasis, and tumor stage. Kaplan–Meier survival analysis showed that a high expression level of AP-4 resulted in a significantly poor prognosis of HCC patients. Multivariate analysis revealed that AP-4 expression level was an independent prognostic parameter for the overall survival rate of HCC patients. These findings provide evidence that a high expression level of AP-4 serves as a biomarker for poor prognosis for HCC. Thus, we speculate that AP-4 may be a potential target of antiangiogenic therapy for HCC.




Similar content being viewed by others
References
Shariff MI, Cox IJ, Gomaa AI, Khan SA, Gedroyc W, TaylorRobinson SD. Hepatocellular carcinoma: current trends in worldwide epidemiology, risk factors, diagnosis and therapeutics. Expert Rev Gastroenterol Hepatol. 2009;3:353–7.
Lau WY, Lai EC. Hepatocellular carcinoma: current management and recent advances. Hepatobiliary Pancreat Dis Int. 2008;7:237–57.
Koorey D. Hepatocellular carcinoma: prevention, detection and treatment in the real world. Intern Med J. 2007;37:513–5.
Pleguezuelo M, Marelli L, Misseri M, Germani G, Calvaruso V, Xiruochakis E, et al. TACE versus TAE as therapy for hepatocellular carcinoma. Expert Rev Anticancer Ther. 2008;8:1623–41.
Massari ME, Murre C. Helix–loop–helix proteins: regulators of transcription in eucaryotic organisms. Mol Cell Biol. 2000;20(2):429–40.
Grau R, Punzon C, Fresno M, Iniguez MA. Peroxisome-proliferator-activated receptor alpha agonists inhibit cyclo-oxygenase 2 and vascular endothelial growth factor transcriptional activation in human colorectal carcinoma cells via inhibition of activator protein-1. Biochem J. 2006;395(1):81–8.
Atchley WR, Fitch WM. A natural classification of the basic helix–loop–helix class of transcription factors. Proc Natl Acad Sci USA. 1997;94(10):5172–6.
Lee SU, Song HO, Lee W, Singaravelu G, Yu JR, Park WY. Identification and characterization of a putative basic helix–loop–helix (bHLH) transcription factor interacting with calcineurin in C. elegans. Mol Cells. 2009;28(5):455–61.
Mermod N, Williams TJ, Tjian R. Enhancer binding factors AP-4 and AP-1 act in concert to activate SV40 late transcription in vitro. Nature. 1988;332(6164):557–61.
Hu YF, Luscher B, Admon A, Mermod N, Tjian R. Transcription factor AP-4 contains multiple dimerization domains that regulate dimer specificity. Genes Dev. 1990;4(10):1741–52.
Jung P, Menssen A, Mayr D, Hermeking H. AP4 encodes a c-MYC-inducible repressor of p21. Proc Natl Acad Sci USA. 2008;105(39):15046–51.
Jung P, Hermeking H. The c-MYC–AP4–p21 cascade. Cell Cycle. 2009;8(7):982–9.
Cui Y, Narayanan CS, Zhou J, Kumar A. Exon-I is involved in positive as well as negative regulation of human angiotensinogen gene expression. Gene. 1998;224(1–2):97–107.
Wang R, Zhang YW, Zhang X, Liu R, Hong S, Xia K, et al. Transcriptional regulation of APH-1A and increased gamma-secretase cleavage of APP and Notch by HIF-1 and hypoxia. FASEB J. 2006;20(8):1275–7.
Tsujimoto K, Ono T, Sato M, Nishida T, Oguma T, Tadakuma T. Regulation of the expression of caspase-9 by the transcription factor activator protein-4 in glucocorticoid-induced apoptosis. J Biol Chem. 2005;280(30):27638–44.
Kim MY, Jeong BC, Lee JH, Kee HJ, Kook H, Kim NS, et al. A repressor complex, AP4 transcription factor and geminin, negatively regulates expression of target genes in nonneuronal cells. Proc Natl Acad Sci USA. 2006;103(35):13074–9.
Unk I, Kiss-Toth E, Boros I. Transcription factor AP-4 participates in activation of bovine leukemia virus long terminal repeat by p34 tax. Nucleic Acids Res. 1994;22(23):4872–5.
Glahder JA, Hansen CN, Vinther J, Madsen BS, Norrild B. A promoter within the E6 ORF of human papillomavirus type 16 contributes to the expression of the E7 oncoprotein from a monocistronic mRNA. J Gen Virol. 2003;84(Pt 12):3429–41.
Fodor E, Weinrich SL, Meister A, Mermod N, Rutter WJ. A pancreatic exocrine cell factor and AP4 bind overlapping sites in the amylase 2A enhancer. Biochemistry. 1991;30(33):8102–8.
Cao J, Tang M, Li WL, Xie J, Du H, Tang WB, et al. Upregulation of activator protein-4 in human colorectal cancer with metastasis. Int J Surg Pathol. 2009;17(1):16–21.
Lin TX, Huang J, Huang H, Cai QQ, Xu KW, Yin XB, et al. Identification of response element gene sequence for non-steroid hormone transcription factors for the activation and up-regulation of L-plastin expression in prostate cancer. Zhonghua Nan Ke Xue. 2005;11(10):731–4.
Fang W, Li X, Jiang Q, Liu Z, Yang H, Wang S, Xie S, Liu Q, Liu T, Huang J, Xie W, Li Z, Zhao Y, Wang E, Marincola FM, Yao K. Transcriptional patterns, biomarkers and pathways characterizing nasopharyngeal carcinoma of Southern China. J Transl Med. 2008;6:32. doi:10.1186/1479-5876-6-32.
Tu L, Liu Z, He X, He Y, Yang H, Jiang Q, Xie S, Xiao G, Li X, Yao K, Fang W. Over-expression of eukaryotic translation initiation factor 4 gamma 1 correlates with tumor progression and poor prognosis in nasopharyngeal carcinoma. Mol Cancer. 2010;9:78. doi:10.1186/1476-4598-9-78.
Liu Z, Li L, Yang Z, Luo W, Li X, Yang H, Yao K, Wu B, Fang W. Increased expression of MMP9 is correlated with poor prognosis of nasopharyngeal carcinoma. BMC Cancer. 2010;10:270. doi:10.1186/1471-2407-10-270.
Zhu X-D, Zhang J-B, Zhuang P-Y, Zhu H-G, Zhang W, Xiong Y-Q, Wei-Zhong Wu L, Wang Z-YT, Sun H-C. High expression of macrophage colony-stimulating factor in peritumoral liver tissue is associated with poor survival after curative resection of hepatocellular carcinoma. J Clin Oncol. 2008;26:2707–16. doi:10.1200/JCO.2007.15.6521.
Xu MZ, Yao TJ, Lee NP, Ng IO, Chan YT, Zender L, Lowe SW, Poon RT, Luk JM. Yes-associated protein is an independent prognostic marker in hepatocellular carcinoma. Cancer. 2009;115(19):4576–85. doi:10.1002/cncr.24495. PMID: 19551889.
Kelly K, Crowley J, Bunn Jr PA, Presant CA, Grevstad PK, Moinpour CM, Ramsey SD, Wozniak AJ, Weiss GR, Moore DF, Israel VK, Livingston RB, Gandara DR. Randomized phase III trial of paclitaxel plus carboplatin versus vinorelbine plus cisplatin in the treatment of patients with advanced non-small cell lung cancer: a Southwest Oncology Group trial. J Clin Oncol. 2001;19:3210–8.
Hennessy BT, Hanrahan EO, Daly PA. Non-Hodgkin lymphoma: an update. Lancet Oncol. 2004;5:341–53.
Peng SY, Lai PL, Chu JS, Lee PH, Tsung PT, Chen DS, et al. Expression and hypomethylation of alpha-fetoprotein gene in unicentric and multicentric human hepatocellular carcinomas. Hepatology. 1993;17:35–41.
Hsu HC, Jeng YM, Mao TL, Chu JS, Lai PL, Peng SY. β-Catenin mutations are associated with a subset of low stage hepatocellular carcinoma negative for hepatitis B virus and with favorable prognosis. Am J Pathol. 2000;157:763–70.
Tuck AB, O’Malley FP, Singhal H, Harris JF, Tonkin KS, Kerkvliet N, et al. Osteopontin expression in a group of lymph node negative breast cancer patients. Int J Cancer. 1998;79:502–8.
Ue T, Yokozaki H, Kitadai Y, Yamamoto S, Yasui W, Ishikawa T, et al. Co-expression of osteopontin and CD44v9 in gastric cancer. Int J Cancer. 1998;79:127–32.
Hwang YH, Choi JY, Kim S, et al. Over-expression of c-raf-1 proto-oncogene in liver cirrhosis and hepatocellular carcinoma. Hepatol Res. 2004;29:113–21.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Ben-Shun Hu, Gang Zhao, and Hai-Feng Yu contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Hu, BS., Zhao, G., Yu, HF. et al. High expression of AP-4 predicts poor prognosis for hepatocellular carcinoma after curative hepatectomy. Tumor Biol. 34, 271–276 (2013). https://doi.org/10.1007/s13277-012-0547-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-012-0547-4