Abstract
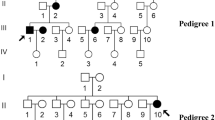
Mitochondrial single nucleotide polymorphisms (SNPs) are significantly correlated with the development of diabetes mellitus . However, these polymorphisms are emerging to be considerably diverse among different population and regions. Sivaganga district is recently being given importance for genetic studies due to its rigid family structure. Hence, the present study is planned to investigate the correlation between mitochondrial SNP of ND1 gene with type II diabetes (T2D) of Karaikudi population of Sivaganga district. For this study, 206 samples including 105 T2D and 101 healthy volunteers were collected and screened for ND1 gene mutation. Genotyping for the ND1 gene was characterized by polymerase chain reaction, Restriction fragment length polymorphism (RFLP) followed by sequencing. In addition, clinical and biochemical variables were analyzed. RFLP and sequencing results showed G>A transition at 3316 position of ND1 gene in both T2D (6.67 %) and in control subjects (1.9 %). Since the frequency of the mutation was found high in T2D patients, we screened for the mutation in their family members and identified its maternal mode of inheritance. The result warrants for the detailed study to identify population based mitochondrial mutation association with disease development.



Similar content being viewed by others
References
Alcolado JC, Majid A, Brockington M, Sweeney MG, Morgan R, Rees A, Harding AE, Barnett AH (1994) Mitochondrial gene defects in patients with NIDDM. Diabetologia 37:372–376
Andrews RM, Kubacka I, Chinnery PF (1999) Reanalysis and revision of the Cambridge reference sequence for human mitochondrial DNA. Nat Genet 23:147
Bartel SH (1972) Serum creatinine estimation by Jaffe Kinetic method. Clin Chem 37:193
Brandon MC, Lott MT, Nguyen KC, Spolim S, Navathe SB, Baldi P, Wallace DC (2005) MITOMAP: a human mitochondrial genome database-2004 update. Nucleic Acids Res 33:611–613
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414:813–820
Chinnery PF, Johnson MA, Wardell TM, Singh Kler R, Hayes C, Brown DT, Taylor RW, Bindoff LA, Turnbull DM, Turnbull DM (2000) The epidemiology of pathogenic mitochondrial DNA mutations. Ann Neurol 48:188–193
Choo Kang AT, Lynn S, Taylor GA, Daly ME, Sihota SS, Wardell TM, Chinnery PF, Turnbull DM, Walker M (2002) Defining the importance of mitochondrial gene defects in maternally inherited diabetes by sequencing the entire mitochondrial genome. Diabetes 51:2317–2320
Das M, Pal S, Ghosh A (2012) Family history of type 2 diabetes and prevalence of metabolic syndrome in adult Asian Indians. J Cardiovasc Dis Res 3:104–108
Deng GH, Zhou X, Pang ZY, Liu SM, Xie Y (2009) Study on mitochondrial function of ND1 gene with 3316 G-A mutation in human diabetes. Zhonghua Yi Xue Za Zhi 89:2822–2826
Devi K, Nisha RG, Prabhu NM, Manikandan R, Karthikeyan M (2013) Awareness on type II diabetes and its complication among sivaganga district population in Tamilnadu: a cross section survey. J Adv Sci Res 4:38–42
Devi K, Santhini E, Manikandan R, Prabhu NM (2015) The prevalence, awareness and potential of complementary alternative medicine in type 2 diabetics living in Madurai, India. Eur J Integr Med (in press)
Duraisamy P, Santhini E, Vijaya Padma V, Balamurugan R (2010) Prevalence of mitochondrial tRNA gene mutations and their association with specific clinical phenotypes in type-II diabetes mellitus patients of Coimbatore. Genet Test Mol Biomark 14:49–55
Genetics and Diabetes: what’s your risk? Joslin Diabetes Center (2013) Stay healthy with diabetes. (http://www.joslin.org/info/genetics_and_diabetes.html)
Genetics and Diabetes. (http://www.who.int/genomics/about/Diabetis-fin.pdf)
Gerbitz KD, Gempel K, Brdiczka D (1996) Mitochondria and diabetes: genetic, biochemical, and clinical implications of the cellular energy circuit. Diabetes 45:113–126
Giles RE, Blanc H, Cann HM, Wallace DC (1980) Maternal inheritance of human mitochondrial DNA. Proc Natl Acad Sci 77:6715–6719
Hattori Y, Nakajima K, Eizawa T, Ehara T, Koyama M, Hirai T, Fukuda Y, Kinoshita M (2003) Heteroplasmic mitochondrial DNA 3310 mutation in NADH dehydrogenase subunit one associated with type two diabetes, hypertrophic cardiomyopathy, and mental retardation in a single patient. Diabetes Care 26:952–953
Hirai M, Suzuki S, Onoda M, Hinokio Y, Ai L, Hirai A, Ohtomo M, Komatsu K, Kasuga S, Satoh Y, Akai H, Toyota T (1996) Mitochondrial DNA 3394 mutation in the NADH dehydrogenase subunit 1 associated with non-insulin-dependent diabetes mellitus. Biochem Biophys Res Commun 219:951–955
Jeppson JO (2002) Approved IFCC reference method for the measurement of HbA1c in human blood. Clin Chem Lab Med 40:78–89
Ji L, Hou X, Han X (2001) Prevalence and clinical characteristics of mitochondrial tRNAleu(UUR) mt 3243A>G and ND1 gene mt 3316 G>A mutations in Chinese patients with type 2 diabetes. Chin Med J 114:1205–1207
Kalinin VN, Schmidt W, Poller W, Olek K (1995) A new point mutation in the mitochondrial gene ND1, detected in a patient with type II diabetes. Genetika 31:1180–1182
Lam C, Yang T, Tsang M, Pang C (2001) Homoplasmic 3316G→A in the ND1 gene of the mitochondrial genome: a pathogenic mutation or a neutral polymorphism. J Med Genet 38:10
Larsson NG, Clayton DA (1995) Molecular genetic aspects of human mitochondrial disorders. Annu Rev Genet 29:151–178
Liu SM, Zhou X, Zheng F, Li X, Liu F, Zhang HM, Xie Y (2007) Novel mutations found in mitochondrial diabetes in Chinese Han population. Diabetes Res Clin Pract 76:425–435
Mezghani N, Mkaouar Rebai E, Mnif M, Charfi N, Rekik N, Youssef S, Abid M, Fakhfash F (2010) The heteroplasmic m.14709T > C mutation in the tRNAGlu gene in two Tunisian families with mitochondrial diabetes. J Diabetes Complicat 24:270–277
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215
Nakano S, Fukuda M, Hotta F, Ito T, Ishii T, Kitazawa M, Nishizawa M, Kiqoshi T, Kakinuma H, Takahashi H, Uchida K (1998) Mitochondrial DNA point mutation at nucleotide pair 3316 in a Japanese family with heterogeneous phenotypes of diabetes. Endocr J 45:625–630
Pranoto A (2005) The association of mitochondrial DNA mutation G3316A and T3392C with diabetes mellitus. Folia Med Indonesia 41:3–8
Richterich R, Kuffer H (1973) The determination of urea in plasma and serum by a urease/Berthelot method. Klin Biochem 11:553–564
Rieder MJ, Taylor SL, Tobe VO, Nickerson DA (1998) Automating the identification of DNA variations using quality-based fluorescence re-sequencing: analysis of the human mitochondrial genome. Nucleic Acids Res 26:967–973
Sasakuma F, Shimizu T, Ishikawa O, Hasegawa K, Kitamura H, Matsumiya K, Doi K, Kojima J (1994) An improved method for determination of C-peptide levels in serum: pretreatment of test samples with anti-insulin antibody insolubilized with magnetic particles. Rinsho Byori 42:183–188
Seneca S, Lissens W, Liebaers I, Van den Bergh P, Nassogne MC, Benatar A, De Meirleir L (1998) Pitfalls in the diagnosis of mtDNA mutations. J Med Genet 35:963–964
Shin CS, Kim SK, Park KS, Kim WB, Kim SY, Cho BY, Lee HK, Koh CS, Shin CH, Lee JB (1998) A new point mutation (3426, A to G) in mitochondrial NADH dehydrogenase gene in Korean diabetic patients which mimics 3243 mutation by restriction fragment length polymorphism pattern. Endocr J 45:105–110
Trinder P (1969) Blood sugar estimation by GOD–POD method. Ann Clin Biochem 6:24–27
Van den Ouweland JM, Lemkes HH, Trembath RC, Ross R, Velho G, Cohen D, Froguel P, Maassen JA (1994) Maternally inherited diabetes and deafness is a distinct subtype of diabetes and associates with a single point mutation in the mitochondrial tRNALeu (UUR) gene. Diabetes 43:746–751
Vijaya Padma V, Anitha S, Santhini E, Pradeepa D, Tresa D, Ganesan P, Ishwarya P, Balakrishan R (2010) Mitochondrial and nuclear gene mutations in the type 2 diabetes patients of Coimbatore population. Mol Cell Biochem 345:223–229
Weng SW, Lin TK, Wang PW, Chen SD, Chuang YC, Liou CW (2013) Single nucleotide polymorphisms in the mitochondrial control region are associated with metabolic phenotypes and oxidative stress. Gene 531:370–376
Yu P, Yu D, Liu D, Wang K, Tang X (2004) Relationship between mutations of mitochondrial DNA ND1 gene and type 2 diabetes. Chin Med J 117:985–989
Zhao Q, Zhang L (2001) Relationship between mitochondrial DNA nt3394T→C mutation in the ND1 gene region and DM. Chin J Med Genet 18:229–230
Acknowledgments
We would like to thank Dr. M. Manivannan (Diabetologist), volunteers, patients and their family members for their co-operation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical compliance
This study was carried out with ethical approval from the Alagappa University Institutional ethical committee (Human Research), Reference number IEC/AU/2013/2.
Conflict of interest
The authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Devi, K., Santhini, E., Ramanan, D. et al. Mitochondrial ND1 gene mutation analysis in type II diabetes of Karaikudi population. Genes Genom 38, 37–43 (2016). https://doi.org/10.1007/s13258-015-0337-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-015-0337-7