Abstract
Purpose
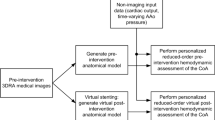
Numerical assessment of the pressure drop across an aortic coarctation using CFD is a promising approach to replace invasive catheter-based measurements. The aim of this study was to investigate and quantify the uncertainty of numerical calculation of the pressure drop introduced during two essential steps of medical image processing: segmentation of the patient-specific geometry and measurement of patient-specific flow rates from 4D-flow-MRI.
Methods
Based on the baseline segmentation, geometries with different stenosis diameters were generated for a sample of ten patients. The pressure drop generated by these geometries was calculated for different volume flow rates using computational fluid dynamics. Based on these simulations, a second order polynomial fit was calculated. Based on these polynomial fits an uncertainty of pressure drop calculation was quantified.
Results
The calculated pressure drop values varied strongly between the patients. In four patients, pressure drops above and below the clinical threshold of 20 mmHg were found. The median standard deviation of the pressure drop was 2.3 mmHg. The sensitivity of the pressure drop toward changes in the volume flow rate or the stenosis geometry varied between patients.
Conclusion
The uncertainty of numerical pressure drop calculation introduced by uncertainties during image segmentation and measurement of volume flow rates was comparable to the uncertainty of pressure drop measurements using invasive catheterization. However, in some patients this uncertainty would have led to different treatment decision. Therefore, patient-specific uncertainty assessment might help to better understand the reliability of a numerically calculated biomarker as the pressure drop across an aortic coarctation.




Similar content being viewed by others
References
Abraham, F., M. Behr, and M. Henkenschloss. Shape optimization in steady blood flow: a numerical study of non-Newtonian effects. Comput. Methods Biomech. Biomed. Eng. 8(2):127–137, 2005.
Andersson, M., J. Lantz, T. Ebbers, and M. Karlsson. Quantitative assessment of turbulence and flow eccentricity in an aortic coarctation: impact of virtual interventions. Cardiovasc. Eng. Technol. 6(3):281–293, 2015.
Bermejo, J., F. Alfonso, and X. Bosch. Imaging techniques in cardiovascular medicine. Rev. Esp. Cardiol. 56:193–194, 2003.
Boccadifuoco, A., A. Mariotti, S. Celi, N. Martini, M. V. Salvetti. Impact of uncertainties in outflow boundary conditions on the predictions of hemodynamic simulations of ascending thoracic aortic aneurysms. Comput. Fluids. 165:96–115. ECCOMAS Congress 2016 Proceedings, 2018.
Boccadifuoco A, Mariotti A, Celi S, Martini N, Salvetti MV. Uncertainty quantification in numerical simulations of the flow in thoracic aortic aneurysms.
Botar, C. C., Á. Á. Tóth, O. R. Klisurić, D. D. Nićiforović, V. A. Vučaj Ćirilović, and V. E. Till. Dynamic simulation and doppler ultrasonography validation of blood flow behavior in abdominal aortic aneurysm. Phys. Med. 37:1–8, 2017.
Bozzi, S., U. Morbiducci, D. Gallo, R. Ponzini, G. Rizzo, C. Bignardi, and G. Passoni. Uncertainty propagation of phase contrast-MRI derived inlet boundary conditions in computational hemodynamics models of thoracic aorta. Comput. Methods Biomech. Biomed. Eng. 20(10):1104–1112, 2017.
Bozzi, S., U. Morbiducci, D. Gallo, R. Ponzini, G. Rizzo, C. Bignardi, and G. Passoni. Uncertainty propagation of phase contrast-MRI derived inlet boundary conditions in computational hemodynamics models of thoracic aorta. Comput. Methods Biomech. Biomed. Eng. 10:1104–1112, 2017.
Bruening, J., F. Hellmeier, P. Yevtushenko, M. Kelm, S. Nordmeyer, S. H. Sündermann, T. Kuehne, and L. Goubergrits. Impact of patient-specific LVOT inflow profiles on aortic valve prosthesis and ascending aorta hemodynamics. J. Comput. Sci. 24:91–100, 2018.
Canniffe, C., P. Ou, K. Walsh, D. Bonnet, and D. Celermajer. Hypertension after repair of aortic coarctation—a systematic review. Int. J. Cardiol. 167(6):2456–2461, 2013.
Celi, S., and S. Berti. Biomechanics and FE modelling of aneurysm: review and advances in computational models, aneurysm. IntechOpen 2012. https://doi.org/10.5772/46030.
Celi, S., and S. Berti. Three-dimensional sensitivity assessment of thoracic aortic aneurysm wall stress: a probabilistic finite-element study. Eur. J. Cardio-Thorac. Surg. 45:467–475, 2014.
Celi, S., N. Martini, L. E. Pastormerlo, V. Positano, and S. Berti. Multimodality imaging for interventional cardiology. Curr. Pharm. Des. 23(22):3285–3300, 2017.
Douglas, P. S., B. De Bruyne, G. Pontone, M. R. Patel, B. L. Norgaard, R. A. Byrne, N. Curzen, I. Purcell, M. Gutberlet, G. Rioufol, U. Hink, H. W. Schuchlenz, G. Feuchtner, M. Gilard, D. Andreini, J. M. Jensen, M. Hadamitzky, K. Chiswell, D. Cyr, A. Wilk, F. Wang, C. Rogers, and M. A. Hlatky. 1-year outcomes of FFRCT-guided care in patients with suspected coronary disease: the PLATFORM study. J. Am. Coll. Cardiol. 68(5):435–445, 2016.
Eck, V. G., W. P. Donders, J. Sturdy, J. Feinberg, T. Delhaas, L. R. Hellevik, and W. Huberts. A guide to uncertainty quantification and sensitivity analysis for cardiovascular applications. Int. J. Numer. Method Biomed. Eng. 21(8):e02755, 2016.
Eck, V. G., J. Sturdy, and L. R. Hellevik. Effects of arterial wall models and measurement uncertainties on cardiovascu-lar model predictions. J. Biomech. 50:188–194, 2017.
Friman, O., A. Hennemuth, A. Harloff, J. Bock, M. Markl, and H. O. Peitgen. Probabilistic 4D blood flow tracking and uncertainty estimation. Med. Image Anal. 15(5):720–728, 2011.
Gallo, D., G. De Santis, F. Negri, D. Tresoldi, R. Ponzini, D. Massai, M. A. Deriu, P. Segers, B. Verhegghe, G. Rizzo, and U. Morbiducci. On the use of in vivo measured flow rates as boundary conditions for image-based hemodynamic models of the human aorta: implications for indicators of abnormal flow. Ann. Biomed. Eng. 40(3):729–741, 2012.
Goubergrits, L., R. Mevert, P. Yevtushenko, J. Schaller, U. Kertzscher, S. Meier, S. Schubert, E. Riesenkampff, and T. Kuehne. The impact of MRI-based inflow for the hemodynamic evaluation of aortic coarctation. Ann. Biomed. Eng. 41:2575–2587, 2013.
Goubergrits, L., E. Riesenkampff, P. Yevtushenko, J. Schaller, U. Kertzscher, F. Berger, and T. Kuehne. Is MRI-based CFD able to improve clinical treatment of coarctations of aorta? Ann. Biomed. Eng. 43(1):168–176, 2015.
Goubergrits, L., E. Riesenkampff, P. Yevtushenko, J. Schaller, U. Kertzscher, A. Hennemuth, F. Berger, S. Schubert, and T. Kuehne. MRI-based computational fluid dynamics for diagnosis and treatment prediction: clinical validation study in patients with coarctation of aorta. J. Magn. Reson. Imaging. 41(4):909–916, 2015.
Hellmeier, F., S. Nordmeyer, P. Yevtushenko, J. Bruening, F. Berger, T. Kuehne, L. Goubergrits, and M. Kelm. Hemodynamic evaluation of a biological and mechanical aortic valve prosthesis using patient-specific MRI-based CFD. Artif. Org. 42(1):49–57, 2018.
Huberts, W., K. Van Canneyt, P. Segers, S. Eloot, J. H. Tordoir, P. Verdonck, F. N. van de Vosse, and E. M. Bosboom. Experimental validation of a pulse wave propagation model for predicting hemodynamics after vascular access surgery. J. Biomech. 45(9):1684–1691, 2012.
International Electrotechnical Commission Standard: IEC 60601-2-34:2011. Medical electrical equipment—Part 2–34: Particular requirements for the basic safety and essential performance of invasive blood pressure monitoring equipment, 2011.
Isaaz, K., J. F. Bruntz, A. Da Costa, D. Winninger, A. Cerisier, C. de Chillou, N. Sadoul, M. Lamaud, G. Ethevenot, and E. Aliot. Noninvasive quantitation of blood flow turbulence in patients with aortic valve disease using online digital computer analysis of doppler velocity data. J. Am. Soc. Echocardiogr. 16(9):965–974, 2003.
Itu, L., P. Sharma, and K. Ralovich. Non-invasive hemodynamic assessment of aortic coarctation: validation with in vivo measurements. Ann. Biomed. Eng. 41:669–681, 2013.
Jager, M. D., J. C. Aldag, and G. G. Deshpande. A presedation fluid bolus does not decrease the incidence of propofol-induced hypotension in pediatric patients. Hosp. Pediatr. 5(2):85–91, 2015.
Karimi, S., M. Dabagh, P. Vasava, M. Dadvar, B. Dabir, and B. Jalali. Effect of rheological models on the hemodynamics within human aorta: CFD study on CT image-based geometry. J. Non-Newton. Fluid Mech. 207:42–52, 2004.
Kousera, C. A., N. B. Wood, W. A. Seed, R. Torii, D. O’Regan, and X. Y. Xu. A numerical study of aortic flow stability and comparison with in vivo flow measurements. J. Biomech. Eng. 135(1):011003, 2013.
Kuprat, A., A. Khamayseh, D. George, and L. Larkey. Volume conserving smoothing for piecewise linear curves, surfaces and triple lines. J. Comput. Phys. 172:99–118, 2001.
Liu, X., Y. Fan, X. Deng, and F. Zhan. Effect of non-Newtonian and pulsatile blood flow on mass transport in the human aorta. J. Biomech. Eng. 44:1123–1131, 2011.
Melero-Ferrer, J. L., R. López-Vilella, H. Morillas-Climent, J. Sanz-Sánchez, I. J. Sánchez-Lázaro, L. Almenar-Bonet, and L. Martínez-Dolz. Novel imaging techniques for heart failure. Card. Fail. Rev. 2(1):27–34, 2016.
Mirzaee, H., T. Henn, M. J. Krause, L. Goubergrits, C. Schumann, M. Neugebauer, T. Kuehne, T. Preusser, and A. Hennemuth. MRI-based computational hemodynamics in patients with aortic coarctation using the lattice Boltzmann methods: clinical validation study. J. Magn. Reson. Imaging. 45(1):139–146, 2017.
Morbiducci, U., R. Ponzini, D. Gallo, C. Bignardi, and G. Rizzo. Inflow boundary conditions for image-based computational hemodynamics: impact of idealized versus measured velocity profiles in the human aorta. J. Biomech. 46(1):102–109, 2013.
Murray, C. D. The physiological principle of minimum work. I. The vascular system and the cost of blood volume. Proc. Natl. Acad. Sci. 12(3):207–214, 1926.
Quarteroni, A., A. Veneziani, and C. Vergara. Geometric multiscale modeling of the cardiovascular system, between theory and practice. Comput. Methods Appl. Mech. Eng. 302:193–252, 2016.
Quicken, S., W. P. Donders, E. M. van Disseldorp, K. Gashi, B. M. Mees, F. N. van de Vosse, R. G. Lopata, T. Delhaas, and W. Huberts. Application of an adaptive polynomial chaos expansion on computationally expensive three-dimensional cardiovascular models for uncertainty quantification and sensitivity analysis. J. Biomech. Eng. 138(12):121010, 2016.
Riesenkampff, E., J. F. Fernandes, S. Meier, L. Goubergrits, S. Kropf, S. Schubert, F. Berger, A. Hennemuth, and T. Kuehne. Pressure fields by flow-sensitive, 4D, velocity-encoded CMR in patients with aortic coarctation. JACC Cardiovasc. Imaging. 7(9):920–926, 2014.
Sankaran, S., L. Grady, and C. A. Taylor. Impact of geometric uncertainty on hemodynamic simulations using ma-chine learning. Comput. Methods Appl. Mech. Eng. 297:167–190, 2015.
Sankaran, S., and A. L. Marsden. A stochastic collocation method for uncertainty quantification and propagation in cardiovascular simulations. J. Biomech. Eng. 133(3):031001, 2011.
Senko, I., A. Shatokhin, I. Bishnoi, Y. Yamada, R. Tanaka, D. Suyama, T. Kawase, and Y. Kato. Intraoperative rupture cerebral aneurysm and computational flow dynamics. Asian J. Neurosurg. 13(2):496–498, 2018.
Tran, J. S., D. E. Schiavazzi, A. B. Ramachandra, A. M. Kahn, and A. L. Marsden. Automated tuning for parameter identification and uncertainty quantification in multi-scale coronary simulations. Comput. Fluids 142:128–138, 2017.
van Bakel, T. M. J., K. D. Lau, J. Hirsch-Romano, S. Trimarchi, A. L. Dorfman, and C. A. Figueroa. Patient-specific modeling of hemodynamics: supporting surgical planning in a fontan circulation correction. J. Cardiovasc. Transl. Res. 11(2):145–155, 2018.
Warnes, C. A., et al. ACC/AHA 2008 guidelines for the management of adults with congenital heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing committee to develop guidelines on the management of adults with congenital heart disease). Circulation 118(23):e714–e833, 2008.
Wyman, R. M., R. D. Safian, V. Portway, J. J. Skillman, R. G. McKay, and D. S. Baim. Current complications of diagnostic and therapeutic cardiac catheterization. J. Am. Coll. Cardiol. 12(6):1400–1406, 1988.
Yevtushenko, P., F. Hellmeier, J. Brüning, T. Kuehne, and L. Goubergrits. Numerical investigation of the impact of branching vessel boundary conditions on aortic hemodynamics. Curr. Dir. Biomed. Eng. 3(2):321–324, 2017.
Zhu, Y., R. Chen, Y. H. Juan, H. Li, J. Wang, Z. Yu, and H. Liu. Clinical validation and assessment of aortic hemodynamics using computational fluid dynamics simulations from computed tomography angiography. Biomed. Eng. Online 17(1):53, 2018.
Conflict of interest
The authors declare that they has no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants and/or their guardians included in the study.
Funding
This work was funded by the German Research Foundation (IDs GO1967/6-1 and KU1329/10-1) and the European Commission (ID 611232).
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editors David A. Steinman, Francesco Migliavacca, and Ajit P. Yoganathan oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Brüning, J., Hellmeier, F., Yevtushenko, P. et al. Uncertainty Quantification for Non-invasive Assessment of Pressure Drop Across a Coarctation of the Aorta Using CFD. Cardiovasc Eng Tech 9, 582–596 (2018). https://doi.org/10.1007/s13239-018-00381-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-018-00381-3