Abstract
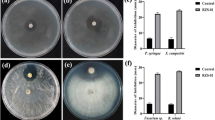
Bacillus spp. have been widely reported with the ability to control plant diseases. In this work, we analyzed the whole genome of LJBS06, which was isolated from grapevine rhizosphere soil. In view of physiological and biochemical characteristics, genome data, and phylogenetic analysis of 16S rRNA, LJBS06 was affiliated with Bacillus stercoris. LJBS06 showed antagonistic activities against a variety of plant pathogens. The inhibition rate of Magnaporthe oryzae was up to 75.05% and the inhibition rates of Colletotrichum gloeosporioides, Coniothyrium diplodiella, and Botrytis cinerea were all above 50% in the plate assays. The genome of LJBS06 had a 4,154,362-bp circular chromosome, with an average GC content of 43.96%, containing an 82,935-bp plasmid with a GC content of 35.18%. The circular chromosome of LJBS06 contained 4231 protein-coding genes, 30 rRNA genes, and 87 tRNA genes, including genes related to the synthesis of plant defense-related enzymes and the promotion of plant growth. Meanwhile, 11 gene clusters involved in biosynthesis of secondary metabolites were present in the genome of LJBS06. In conclusion, our findings indicated that LJBS06 strain had the necessary genetic machinery to control plant pathogens and provided insights for future studies of the biocontrol mechanisms of B. stercoris LJBS06.




Similar content being viewed by others
Availability of data and material
The GenBank accession number for the 16S rRNA gene of Bacillus stercoris LJBS06 is MW940707. The complete sequence of the chromosome of the strain B. stercoris LJBS06 and the sequence of the plasmid have been deposited in GenBank database under the accession number CP070483 and CP070484, respectively. The strain B. stercoris LJBS06 was publicly available from China General Microbiological Culture Collection Center (CGMCC) under the accession number CGMCC No. 21263. General characteristics of B. stercoris LJBS06, effect of LJBS06 on the mycelia of C. gloeosporioides, plate assays for the activities of extracellular enzymes, genome statistics, COG categories of the chromosome and the plasmid of B. stercoris LJBS06, inhibition diameter of B. stercoris LJBS06 against 12 pathogens, genes related to defense-related enzymes in the LJBS06 genome, and genes attributed to plant growth promoting traits in the LJBS06 genome are available as Supplementary Materials.
Code availability
Not applicable.
References
Aboutorabi M (2018) A review on the biological control of plant diseases using various microorganisms. J Res Med Dent Sci 6(4):30–35
Ahemad M, Kibret M (2014) Mechanisms and applications of plant growth promoting rhizobacteria: current perspective. J King Saud Univ Sci 26(1):1–20. https://doi.org/10.1016/j.jksus.2013.05.001
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, Kasarskis A, Lewis S, Matese JC, Richardson JE, Ringwald M, Rubin GM, Sherlock G, Gene Ontology C (2000) Gene ontology: tool for the unification of biology. Nat Genet 25(1):25–29. https://doi.org/10.1038/75556
Bertelli C, Laird MR, Williams KP, Lau BY, Hoad G, Winsor GL, Brinkman FSL, Simon Fraser Univ Res Comp G (2017) IslandViewer 4: expanded prediction of genomic islands for larger-scale datasets. Nucleic Acids Res 45(W1):W30–W35. https://doi.org/10.1093/nar/gkx343
Besemer J, Lomsadze A, Borodovsky M (2001) GeneMarkS: a self-training method for prediction of gene starts in microbial genomes. Implications for finding sequence motifs in regulatory regions. Nucleic Acids Res 29(12):2607–2618. https://doi.org/10.1093/nar/29.12.2607
Blake C, Christensen MN, Kovacs AT (2021) Molecular aspects of plant growth promotion and protection by Bacillus subtilis. Mol Plant Microbe Interact 34(1):15–25. https://doi.org/10.1094/MPMI-08-20-0225-CR
Blin K, Shaw S, Kloosterman AM, Charlop-Powers Z, van Wezel GP, Medema MH, Weber T (2021) antiSMASH 6.0: improving cluster detection and comparison capabilities. Nucleic Acids Res. https://doi.org/10.1093/nar/gkab335
Boeckmann B, Bairoch A, Apweiler R, Blatter MC, Estreicher A, Gasteiger E, Martin MJ, Michoud K, O’Donovan C, Phan I, Pilbout S, Schneider M (2003) The SWISS-PROT protein knowledgebase and its supplement TrEMBL in 2003. Nucleic Acids Res 31(1):365–370. https://doi.org/10.1093/nar/gkg095
Buchfink B, Xie C, Huson DH (2015) Fast and sensitive protein alignment using DIAMOND. Nat Methods 12(1):59–60. https://doi.org/10.1038/nmeth.3176
Byun H-D, Bin HS, Bae S, Han J-M, Hyun C-G (2020) Bacillus subtilis JNUCC Isolated from Galchisokjeot: draft genome sequence and alpha-glucosidase and Tyrosinase inhibitory activities. J Pure Appl Microbiol 14(1):189–193. https://doi.org/10.22207/jpam.14.1.20
Chen YJ, Yu P, Luo JC, Jiang Y (2003) Secreted protein prediction system combining CJ-SPHMM, TMHMM, and PSORT. Mamm Genome 14(12):859–865. https://doi.org/10.1007/s00335-003-2296-6
Chen L, Gu W, Xu HY, Yang GL, Shan XF, Chen G, Wang CF, Qian AD (2018) Complete genome sequence of Bacillus velezensis 157 isolated from Eucommia ulmoides with pathogenic bacteria inhibiting and lignocellulolytic enzymes production by SSF. 3 Biotech 8(2):114. https://doi.org/10.1007/s13205-018-1125-2
Chen MC, Wang JP, Zhu YJ, Liu B, Yang WJ, Ruan CQ (2019) Antibacterial activity against Ralstonia solanacearum of the lipopeptides secreted from the Bacillus amyloliquefaciens strain FJAT-2349. J Appl Microbiol 126(5):1519–1529. https://doi.org/10.1111/jam.14213
Chin C-S, Peluso P, Sedlazeck FJ, Nattestad M, Concepcion GT, Clum A, Dunn C, O’Malley R, Figueroa-Balderas R, Morales-Cruz A, Cramer GR, Delledonne M, Luo C, Ecker JR, Cantu D, Rank DR, Schatz MC (2016) Phased diploid genome assembly with single-molecule real-time sequencing. Nat Methods 13(12):1050. https://doi.org/10.1038/nmeth.4035
Chun J, Oren A, Ventosa A, Christensen H, Arahal DR, da Costa MS, Rooney AP, Yi H, Xu XW, De Meyer S, Trujillo ME (2018) Proposed minimal standards for the use of genome data for the taxonomy of prokaryotes. Int J Syst Evol Microbiol 68(1):461–466. https://doi.org/10.1099/ijsem.0.002516
Dhruw C, Husain K, Kumar V, Sonawane VC (2020) Novel xylanase producing Bacillus strain X2: molecular phylogenetic analysis and its application for production of xylooligosaccharides. 3 Biotech. https://doi.org/10.1007/s13205-020-02322-1
Djordje F, Ivica D, Tanja B, Jelena L, Slaviša S (2018) Biological control of plant pathogens by Bacillus species. J Biotechnol 285:44–55
Dunlap CA, Bowman MJ, Zeigler DR (2020) Promotion of Bacillus subtilis subsp. inaquosorum, Bacillus subtilis subsp. spizizenii and Bacillus subtilis subsp. stercoris to species status. Antonie Van Leeuwenhoek 113(1):1–12. https://doi.org/10.1007/s10482-019-01354-9
Fan H, Ru J, Zhang Y, Wang Q, Li Y (2017) Fengycin produced by Bacillus subtilis 9407 plays a major role in the biocontrol of apple ring rot disease. Microbiol Res 199:89–97. https://doi.org/10.1016/j.micres.2017.03.004
Fira D, Dimkić I, Berić T, Lozo J, Stanković S (2018) Biological control of plant pathogens by Bacillus species. J Biotechnol 285:44–55. https://doi.org/10.1016/j.jbiotec.2018.07.044
Fu L, Penton CR, Ruan Y, Shen Z, Xue C, Li R, Shen Q (2017) Inducing the rhizosphere microbiome by biofertilizer application to suppress banana Fusarium wilt disease. Soil Biol Biochem 104:39–48. https://doi.org/10.1016/j.soilbio.2016.10.008
Gao Z, Zhang B, Liu H, Han J, Zhang Y (2017) Identification of endophytic Bacillus velezensis ZSY-1 strain and antifungal activity of its volatile compounds against Alternaria solani and Botrytis cinerea. Biol Control 105:27–39. https://doi.org/10.1016/j.biocontrol.2016.11.007
Gao H, Li P, Xu X, Zeng Q, Guan W (2018) Research on volatile organic compounds from Bacillus subtilis CF-3: biocontrol effects on fruit fungal pathogens and dynamic changes during fermentation. Front Microbiol. https://doi.org/10.3389/fmicb.2018.00456
Grissa I, Vergnaud G, Pourcel C (2007) CRISPRFinder: a web tool to identify clustered regularly interspaced short palindromic repeats. Nucleic Acids Res 35:W52–W57. https://doi.org/10.1093/nar/gkm360
Guerrero R (2001) Bergey’s manuals and the classification of prokaryotes. Int Microbiol 4(2):103–109
Guo W, Chen Z, Cui P, Chen X (2015) Draft genome of Bacillus sp A053 isolated from the Arctic seawater with antimicrobial activity. Mar Genom 22:19–21. https://doi.org/10.1016/j.margen.2015.03.008
Hakamada Y, Koike K, Kobayashi T, Ito S (1997) Purification and properties of mangano-superoxide dismutase from a strain of alkaliphilic Bacillus. Extremophiles 1(2):74–78. https://doi.org/10.1007/s007920050017
Helisto P, Aktuganov G, Galimzianova N, Melentjev A, Korpela T (2001) Lytic enzyme complex of an antagonistic Bacillus sp., X-b: isolation and purification of components. J Chromatogr B 758(2):197–205. https://doi.org/10.1016/s0378-4347(01)00181-5
Hu Q-P, Xu J-G (2011) A simple double-layered chrome azurol S agar (SD-CASA) plate assay to optimize the production of siderophores by a potential biocontrol agent Bacillus. Afr J Microbiol Res 5(25):4321–4327. https://doi.org/10.5897/ajmr11.238
Jensen LJ, Julien P, Kuhn M, von Mering C, Muller J, Doerks T, Bork P (2008) eggNOG: automated construction and annotation of orthologous groups of genes. Nucleic Acids Res 36:D250–D254. https://doi.org/10.1093/nar/gkm796
Jiang C-H, Liao M-J, Wang H-K, Zheng M-Z, Xu J-J, Guo J-H (2018) Bacillus velezensis, a potential and efficient biocontrol agent in control of pepper gray mold caused by Botrytis cinerea. Biol Control 126:147–157. https://doi.org/10.1016/j.biocontrol.2018.07.017
Kalvari I, Argasinska J, Quinones-Olvera N, Nawrocki EP, Rivas E, Eddy SR, Bateman A, Finn RD, Petrov AI (2018a) Rfam 13.0: shifting to a genome-centric resource for non-coding RNA families. Nucleic Acids Res 46(D1):D335–D342. https://doi.org/10.1093/nar/gkx1038
Kalvari I, Nawrocki EP, Argasinska J, Quinones-Olvera N, Finn RD, Bateman A, Petrov AI (2018b) Non-Coding RNA analysis using the Rfam database. Curr Protoc Bioinform 62(1):e51–e51. https://doi.org/10.1002/cpbi.51
Kaspar F, Neubauer P, Gimpel M (2019) Bioactive secondary metabolites from Bacillus subtilis: a comprehensive review. J Nat Prod 82(7):2038–2053. https://doi.org/10.1021/acs.jnatprod.9b00110
Khan A, Singh P, Srivastava A (2018) Synthesis, nature and utility of universal iron chelator—siderophore: a review. Microbiol Res 212:103–111. https://doi.org/10.1016/j.micres.2017.10.012
Khurshed N, Shakoori AR (2005) Characterization of locally isolated strains of Bacillus and their evaluation as potential biocides against house fly, Musca domestica. Pak J Zool 37(4):249–255
Kim JS, Lee J, Lee CH, Woo SY, Kang H, Seo SG, Kim SH (2015) Activation of pathogenesis-related genes by the rhizobacterium, Bacillus sp. JS, which induces systemic resistance in Tobacco plants. Plant Pathol J 31(2):195–201. https://doi.org/10.5423/PPJ.NT.11.2014.0122
Koren S, Walenz BP, Berlin K, Miller JR, Bergman NH, Phillippy AM (2017) Canu: scalable and accurate long-read assembly via adaptive k-mer weighting and repeat separation. Genome Res 27(5):722–736. https://doi.org/10.1101/gr.215087.116
Krogh A, Brown M, Mian IS, Sjolander K, Haussler D (1994) Hidden Markov models in computational biology: applications to protein modeling. J Mol Biol 235(5):1501–1531. https://doi.org/10.1006/jmbi.1994.1104
Leal C, Fontaine F, Aziz A, Egas C, Clément C, Trotel-Aziz P (2021) Genome sequence analysis of the beneficial Bacillus subtilis PTA-271 isolated from a Vitis vinifera (cv. Chardonnay) rhizospheric soil: assets for sustainable biocontrol. Environ Microbiome. https://doi.org/10.1186/s40793-021-00372-3
Li Y, Heloir M-C, Zhang X, Geissler M, Trouvelot S, Jacquens L, Henkel M, Su X, Fang X, Wang Q, Adrian M (2019) Surfactin and fengycin contribute to the protection of a Bacillus subtilis strain against grape downy mildew by both direct effect and defense stimulation. Mol Plant Pathol 20(8):1037–1050. https://doi.org/10.1111/mpp.12809
Liang TW, Chen YY, Pan PS, Wang SL (2014) Purification of chitinase/chitosanase from Bacillus cereus and discovery of an enzyme inhibitor. Int J Biol Macromol 63:8–14. https://doi.org/10.1016/j.ijbiomac.2013.10.027
Lowe TM, Eddy SR (1997) tRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res 25(5):955–964. https://doi.org/10.1093/nar/25.5.955
Mayende L, Wilhelmi B, Pletschke B (2006) Cellulases (CMCases) and polyphenol oxidases from thermophilic Bacillus spp. isolated from compost. Soil Biol Biochem 38(9):2963–2966. https://doi.org/10.1016/j.soilbio.2006.03.019
Meier-Kolthoff JP, Auch AF, Klenk H-P, Goeker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform. https://doi.org/10.1186/1471-2105-14-60
Miljakovic D, Marinkovic J, Balesevic-Tubic S (2020) The significance of Bacillus spp. in disease suppression and growth promotion of field and vegetable crops. Microorganisms. https://doi.org/10.3390/microorganisms8071037
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428. https://doi.org/10.1021/ac6014a030
Milner JL, Stohl EA, Handelsman J (1996) Zwittermicin a resistance gene from Bacillus cereus. J Bacteriol 178(14):4266–4272. https://doi.org/10.1128/jb.178.14.4266-4272.1996
Mohammad FG, Alireza T (2007) Isolation and characterization of polyphenol oxidase- and peroxidase-producing Bacillus strains from fully fermented tea (Camellia sinensis). World J Microbiol Biotechnol 23(9):1327–1332. https://doi.org/10.1007/s11274-007-9363-1
Nicholson WL, Munakata N, Horneck G, Melosh HJ, Setlow P (2000) Resistance of Bacillus endospores to extreme terrestrial and extraterrestrial environments. Microbiol Mol Biol Rev 64(3):548. https://doi.org/10.1128/mmbr.64.3.548-572.2000
Ogata H, Goto S, Sato K, Fujibuchi W, Bono H, Kanehisa M (1999) KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res 27(1):29–34. https://doi.org/10.1093/nar/27.1.29
Olishevska S, Nickzad A, Deziel E (2019) Bacillus and Paenibacillus secreted polyketides and peptides involved in controlling human and plant pathogens. Appl Microbiol Biotechnol 103(3):1189–1215. https://doi.org/10.1007/s00253-018-9541-0
Ongena M, Jourdan E, Adam A, Paquot M, Brans A, Joris B, Arpigny J-L, Thonart P (2007) Surfactin and fengycin lipopeptides of Bacillus subtilis as elicitors of induced systemic resistance in plants. Environ Microbiol 9(4):1084–1090. https://doi.org/10.1111/j.1462-2920.2006.01202.x
Paudel YP, Qin W (2015) Characterization of novel cellulase-producing bacteria isolated from rotting wood samples. Appl Biochem Biotechnol 177(5):1186–1198. https://doi.org/10.1007/s12010-015-1806-9
Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8(10):785–786. https://doi.org/10.1038/nmeth.1701
Qiao J, Yu X, Liang X, Liu Y, Borriss R, Liu Y (2017) Addition of plant-growth-promoting Bacillus subtilis PTS-394 on tomato rhizosphere has no durable impact on composition of root microbiome. BMC Microbiol 17:1–12. https://doi.org/10.1186/s12866-017-1039-x
Rajput S, Tiwari E (2012) Study the effect of various pH on the protease producing capacity of Bacillus. J Pure Appl Microbiol 6(1):501–503
Richter M, Rossello-Mora R (2009) Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci USA 106(45):19126–19131. https://doi.org/10.1073/pnas.0906412106
Santana MA, Moccia-V CC, Gillis AE (2008) Bacillus thuringiensis improved isolation methodology from soil samples. J Microbiol Methods 75(2):357–358. https://doi.org/10.1016/j.mimet.2008.06.008
Sari G, Trihadiningrum Y, Ni’matuzahroh N (2019) Isolation and Identification of native bacteria from total petroleum hydrocarbon polluted soil in wonocolo public oilfields, Indonesia. J Ecol Eng 20(8):60–64
Shafi J, Tian H, Ji M (2017) Bacillus species as versatile weapons for plant pathogens: a review. Biotechnol Biotechnol Equip 31(3):446–459. https://doi.org/10.1080/13102818.2017.1286950
Thipyapong P, Steffens JC (1997) Tomato polyphenol oxidase—differential response of the polyphenol oxidase F promoter to injuries and wound signals. Plant Physiol 115(2):409–418. https://doi.org/10.1104/pp.115.2.409
Valenzuela-Ruiz V, Robles-Montoya RI, Parra-Cota FI, Santoyo G, Del Carmen O-MM, Rodriguez-Ramirez R, de Los S-VS (2019) Draft genome sequence of Bacillus paralicheniformis TRQ65, a biological control agent and plant growth-promoting bacterium isolated from wheat (Triticum turgidum subsp. durum) rhizosphere in the Yaqui Valley, Mexico. 3 Biotech 9(11):436. https://doi.org/10.1007/s13205-019-1972-5
Walker BJ, Abeel T, Shea T, Priest M, Abouelliel A, Sakthikumar S, Cuomo CA, Zeng Q, Wortman J, Young SK, Earl AM (2014) Pilon: an integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS One. https://doi.org/10.1371/journal.pone.0112963
Wu L, Wu H, Chen L, Lin L, Borriss R, Gao X (2015a) Bacilysin overproduction in Bacillus amyloliquefaciens FZB42 markerless derivative strains FZBREP and FZBSPA enhances antibacterial activity. Appl Microbiol Biotechnol 99(10):4255–4263. https://doi.org/10.1007/s00253-014-6251-0
Wu L, Wu H, Chen L, Yu X, Borriss R, Gao X (2015b) Difficidin and bacilysin from Bacillus amyloliquefaciens FZB42 have antibacterial activity against Xanthomonas oryzae rice pathogens. Sci Rep. https://doi.org/10.1038/srep12975
Xu L, Ning X, Chai H, Fei G (2020a) Preliminary evaluation of irradiated medium and the optimization of conditions for a catalase produced by Bacillus firmus GL3. J Food Meas Charact 14(2):1073–1084. https://doi.org/10.1007/s11694-019-00357-4
Xu S, Xie X, Zhao Y, Shi Y, Chai A, Li L, Li B (2020b) Whole-genome analysis of Bacillus velezensis ZF2, a biocontrol agent that protects cucumis sativus against corynespora leaf spot diseases. 3 Biotech 10(4):186. https://doi.org/10.1007/s13205-020-2165-y
Yanez-Mendizabal V, Falconi CE (2021) Bacillus subtilis CtpxS2-1 induces systemic resistance against anthracnose in Andean lupin by lipopeptide production. Biotech Lett 43(3):719–728. https://doi.org/10.1007/s10529-020-03066-x
Yang J-H, Kim I-S, Oh C-H, Oh N-S (2012) Protease and γ-glutamyltranspeptidase activities of Bacillus spp. isolated from rice straw. Food Sci Biotechnol 21(1):291–294. https://doi.org/10.1007/s10068-012-0039-8
Yang J, Zhu X, Cao M, Wang C, Zhang C, Lu Z, Lu F (2016) Genomics-inspired discovery of three antibacterial active metabolites, aurantinins B, C, and D from compost-associated Bacillus subtilis fmb60. J Agric Food Chem 64(46):8811–8820. https://doi.org/10.1021/acs.jafc.6b04455
Zhang M, Li J, Shen A, Tan S, Yan Z, Yu Y, Xue Z, Tan T, Zeng L (2016) Isolation and Identification of Bacillus amyloliquefaciens IBFCBF-1 with potential for biological control of phytophthora blight and growth promotion of pepper. J Phytopathol 164(11–12):1012–1021. https://doi.org/10.1111/jph.12522
Zhang X, Zhou Y, Li Y, Fu X, Wang Q (2017) Screening and characterization of endophytic Bacillus for biocontrol of grapevine downy mildew. Crop Prot 96:173–179. https://doi.org/10.1016/j.cropro.2017.02.018
Funding
This work was supported by Shanghai Municipal Commission for Science and Technology (18391900400), National Key R & D Program of China (2019YFD1002501), and China Postdoctoral Science Foundation (18Z102060107 and 20Z102060031).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by BW, HP, WW, BY, YC, FX, YP, PF. The first draft of the manuscript was written by BW and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
All authors approved the manuscript.
Consent for publication
Written informed consent for publication was obtained from all participants.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, B., Peng, H., Wu, W. et al. Genomic insights into biocontrol potential of Bacillus stercoris LJBS06. 3 Biotech 11, 458 (2021). https://doi.org/10.1007/s13205-021-03000-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-03000-6