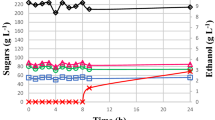
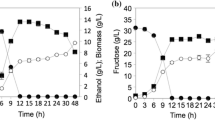
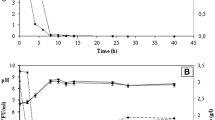
Abstract
The aim of this study was to evaluate the kinetic parameters and the production of metabolites of 13 novel yeasts isolated from a distillery and fruits, and Saccharomyces cerevisiae CAT-1, cultivated in fructose-based medium. The yeasts with the highest µmax were obtained from must, Pichia kudriavzevii BB2, P. kudriavzevii BB1, and S. cerevisiae BB9 (0.47–0.49 h−1). S. cerevisiae CAT-1 (3.02 g gDCM−1 h−1), S. cerevisiae BB9 (3.01 g gDCM−1 h−1), and Candida glabrata Recol 41 (2.52 g gDCM−1 h−1) stood out in terms of µS. C. parapsilosis Recol 29, and Rhodotorula mucilaginosa Recol 03 strains showed the highest YX/S (0.30 and 0.28 gDCM g−1, respectively). C. glabrata Recol 10 and S. cerevisiae BB9 strains stood out for their higher substrate conversion rates into ethanol (0.44 and 0.41 gEth gS−1, respectively). R. mucilaginosa Recol 03 presented the poorest performance in substrate consumption (0.87 g gDCM−1 h−1), while the strains isolated from must and C. glabrata Recol 10 showed the highest ethanol production and the C. parapsilosis Recol 29 showed the highest biomass conversion.



Similar content being viewed by others
Abbreviations
- q S :
-
Specific substrate uptake rate
- µ max :
-
Maximum specific growth rate
- Y X/S :
-
Biomass yield
- Y P/S :
-
Product yield
- X max :
-
Maximum cell concentration
References
Albertyn J, Pohl CH, Viljoen BC (2014) Rhodotorula. In: Batt CA, Tortorello ML (eds) Encyclopedia of Food Microbiology, 2nd edn. Academic Press, Cambridge, pp 291–295
Andersen D, Renshaw JC, Wiebe MG (2003) Rhodotorulic acid production by Rhodotorula mucilaginosa. Mycol Res 107:949–956. https://doi.org/10.1017/S0953756203008220
Anjos J, De-Sousa HR, Roca C, Cássio F, Luttik M, Pronk JT, Salema-Oom M, Gonçalves P (2013) Fsy1, the sole hexose-proton transporter characterized in Saccharomyces yeasts, exhibits a variable fructose: H+ stoichiometry. Biochim Biophys Acta 1828:201–207. https://doi.org/10.1016/j.bbamem.2012.08.011
Basso LC, Basso TO, Rocha SN (2011) Ethanol production in Brazil: the industrial process and its impact on yeast fermentation. Bernardes MAS -recent developments biofuel productionand prospects. Intech, Rijeka, pp 85–100
Batistote M, Cardoso CAL, Ramos DD, Ernandes JR (2010) Desempenho de leveduras obtidas em indústrias de Mato Grosso do Sul na produção de etanol em mosto a base de cana de açúcar. Ciên Nat 32:83–95
Bellaver LH, De Carvalho NMB, Abrahão-Neto J, Gombert AK (2004) Ethanol formation and enzyme activities around glucose-6-phosphate in Kluyveromyces marxianus CBS 6556 exposed to glucose or lactose excess. FEMS Yeast Res 4:691–698. https://doi.org/10.1016/j.femsyr.2004.01.004
Berthels NJ, Otero RRC, Bauer FF, Thevelein JM, Pretorius IS (2004) Discrepancy in glucose and fructose utilization during fermentation by Saccharomyces cerevisiae wine yeast strains. FEMS Yeast Res 4:683–689. https://doi.org/10.1016/j.femsyr.2004.02.005
Bura R, Vajzovic A, Doty SL (2012) Novel endophytic yeast Rhodotorula mucilaginosa strain PTD3 I: production of xylitol and ethanol. J Ind Microbiol Biotechnol 39:1003–1011. https://doi.org/10.1007/s10295-012-1109-x
Camargo JZ, Nascimento VM, Stefanello I, Silva CAA, Gonçalves FA, Perdomo IC, Fonseca GG (2018) Biochemical evaluation, molecular characterization and identification of novel yeast strains isolated from Brazilian savannah fruits, chicken litter and a sugar and alcohol mill with biotechnological potential for biofuel and food industries. Biocatal Agric Biotechnol 16:390–399. https://doi.org/10.1016/j.bcab.2018.09.006
Cardona CA, Sánchez OJ (2007) Fuel ethanol production: process design trends and integration opportunities. Bioresour Technol 98:2415–2457. https://doi.org/10.1016/j.biortech.2007.01.002
Castrillo JI, Ugalde UO (1993) Patterns of energy metabolism and growth kinetics of Kluyveromyces marxianus in whey chemostat culture. Appl Microbiol Biotechnol 40:386–393. https://doi.org/10.1007/BF00170398
Chan GF, Gan HM, Ling HL, Rashid NAA (2012) Genome sequence of Pichia kudriavzevii M12, a potential producer of bioethanol and phytase. Eukaryot Cell 11:1300–1301. https://doi.org/10.1128/EC.00229-12
Contreras A, Hidalgo C, Schmidt S, Henschke PA, Curtin C, Varela C (2015) The application of non-Saccharomyces yeast in fermentations with limited aeration as a strategy for the production of wine with reduced alcohol content. Int J Food Microbiol 205:7–15. https://doi.org/10.1016/j.ijfoodmicro.2015.03.027
Cordero-Bueso G, Esteve-Zarzoso B, Cabellos JM, Gil-Díaz M, Arroyo T (2013) Biotechnological potential of non-Saccharomyces yeasts isolated during spontaneous fermentations of Malvar (Vitis vinifera cv. L.). Eur Food Res Technol 236:193–207. https://doi.org/10.1007/s00217-012-1874-9
Della-Bianca BE, de Hulster E, Pronk JT, van Maris AJ, Gombert AK (2014) Physiology of the fuel ethanol strain Saccharomyces cerevisiae PE-2 at low pH indicates a context-dependent performance relevant for industrial applications. FEMS Yeast Res 14:1196–1205. https://doi.org/10.1111/1567-1364.12217
Dhaliwal SS, Oberoi HS, Sandhu SK, Nanda D, Kumar D, Uppal SK (2011) Enhanced ethanol production from sugarcane juice by galactose adaptation of a newly isolated thermotolerant strain of Pichia kudriavzevii. Bioresour Technol 102:5968–5975. https://doi.org/10.1016/j.biortech.2011.02.015
Fonseca GG, De Carvalho NMB, Gombert AK (2013) Growth of the yeast Kluyveromyces marxianus CBS 6556 on different sugar combinations as sole carbon and energy source. Appl Microbiol Biotechnol 97:5055–5067. https://doi.org/10.1007/s00253-013-4748-6
Freire AL, Ramos CL, De Almeida EG, Duarte WF, Schwan RF (2014) Study of the physicochemical parameters and spontaneous fermentation during the traditional production of yakupa, an indigenous beverage produced by Brazilian Amerindians. World J Microbiol Biotechnol 30:567–577. https://doi.org/10.1007/s11274-013-1476-0
Giannattasio S, Guaragnella N, Ždralević M, Marra E (2013) Molecular mechanisms of Saccharomyces cerevisiae stress adaptation and programmed cell death in response to acetic acid. Front Microbiol 4:33. https://doi.org/10.3389/fmicb.2013.00033
Gomes FCO, Silva CLC, Marini MM, Oliveira ES, Rosa CA (2007) Use of selected indigenous Saccharomyces cerevisiae strains for the production of the traditional cachaça in Brazil. J Appl Microbiol 103:2438–2447. https://doi.org/10.1111/j.1365-2672.2007.03486.x
Gomez-Flores M, Nakhla G, Hafez H (2015) Microbial kinetics of Clostridium termitidis on cellobiose and glucose for biohydrogen production. Biotechnol Lett 37:1965–1971. https://doi.org/10.1111/j.1365-2672.2007.03486.x
Jasman J, Prijambada ID, Hidayat C, Widianto D (2012) Selection of yeast strains for ethanol fermentation of glucose-fructose-sucrose mixture. Indones J Biotechnol 17:114–120. https://doi.org/10.22146/ijbiotech.16001
Jones GV, White MA, Cooper OR, Storchmann K (2005) Climate change and global wine quality. Clim Chang 73:319–343. https://doi.org/10.1007/s10584-005-4704-2
Kitagawa T, Tokuhiro K, Sugiyama H, Kohda K, Isono N, Hisamatsu M, Takahashi H, Imaeda T (2010) Construction of a β-glucosidase expression system using the multistress-tolerant yeast Issatchenkia orientalis. Appl Microbiol Biotechnol 87:1841–1853. https://doi.org/10.1007/s00253-010-2629-9
Kogan G, Kocher A (2007) Role of yeast cell wall polysaccharides in pig nutrition and health protection. Livest Sci 109:161–165. https://doi.org/10.1016/j.livsci.2007.01.134
Kumdam H, Murthy SN, Gummadi SN (2013) Production of ethanol and arabitol by Debaryomyces nepalensis: influence of process parameters. AMB Expr 3:1–13. https://doi.org/10.1186/2191-0855-3-23
Kurtzman CP, Suzuki M (2010) Meyerozyma. In: Kurtzman C, Fell JW, Boekhout T (eds) The yeasts: a taxonomic study, 5th edn. Elsevier, Amsterdam, pp 621–624
Limtong S, Yongmanitchai W, Kawasaki H, Fujiyama K (2009) Wickerhamomyces edaphicus sp. nov. and Pichia jaroonii sp. nov., two ascomycetous yeast species isolated from forest soil in Thailand. FEMS Yeast Res 9:504–510. https://doi.org/10.1111/j.1567-1364.2009.00488.x
Nascimento VM, Silva LF, Gomez JGC, Fonseca GG (2016) Growth of Burkholderia sacchari LFM 101 cultivated in glucose, sucrose and glycerol. Sci Agric 73:429–433. https://doi.org/10.1590/0103-9016-2015-0196
Nolleau V, Preziosi-Belloy L, Navarro JM (1995) The reduction of xylose to xylitol by Candida guilliermondii and Candida parapsilosis: incidence of oxygen and pH. Biotechnol Lett 17:417–422. https://doi.org/10.1007/BF00130800
Nyanga LK, Nout MJ, Smid EJ, Boekhout T, Zwietering MH (2012) Yeasts preservation: alternatives for lyophilisation. World J Microbiol Biotechnol 28:3239–3244. https://doi.org/10.1007/s11274-012-1118-y
Oliveira APA, Silvestre MA, Alves-Prado HF, Rodrigues A, Paz MF, Fonseca GG, Leite RSR (2015) Bioprospecting of yeasts for amylase production in solid state fermentation and evaluation of the catalytic properties of enzymatic extracts. Afr J Biotechnol 14:1215–1223. https://doi.org/10.5897/AJB2014.14062
Orlowski JH, Barford JP (1991) Direct uptake of sucrose by Saccharomyces cerevisiae in batch and continuous culture. J Gen Appl Microbiol 37:215–218. https://doi.org/10.2323/jgam.37.215
Papon N, Savini V, Lanoue A, Simkin AJ, Crèche J, Giglioli-Guivarc’h N, Clastre M, Courdavault V, Sibirny AA (2013) Candida guilliermondii: biotechnological applications, perspectives for biological control, emerging clinical importance and recent advances in genetics. Curr Genet 59:73–90. https://doi.org/10.1007/s00294-013-0391-0
Postma E, Verduyn C, Scheffers WA, Van Dijken JP (1989) Enzymic analysis of the Crabtree effect in glucose-limited chemostat cultures of Saccharomyces cerevisiae. Appl Environ Microbiol 55:468–477
Pronk JT, Yde Steensma H, Van Dijken JP (1996) Pyruvate metabolism in Saccharomyces cerevisiae. Yeast 12:1607–1633. https://doi.org/10.1002/(SICI)1097-0061(199612)12:16%3C1607:AID-YEA70%3E3.0.CO;2-4
Ramos CL, Duarte WF, Freire AL, Dias DR, Eleutherio ECA, Schwan RF (2013) Evaluation of stress tolerance and fermentative behavior of indigenous Saccharomyces cerevisiae. Braz J Microbiol 44:935–944. https://doi.org/10.1590/S1517-83822013005000051
Romani A, Pereira F, Johansson B, Domingues L (2015) Metabolic engineering of Saccharomyces cerevisiae ethanol strains PE-2 and CAT-1 for efficient lignocellulosic fermentation. Bioresour Technol 179:150–158. https://doi.org/10.1016/j.biortech.2014.12.020
Shafaghat H, Najafpour GD, Reazaei PS, Sharifzadeh M (2009) Growth kinetics and ethanol productivity of Saccharomyces cerevisiae PTCC 24860 on various carbon sources. World Appl Sci J 7:140–144
Silva CAA, Fonseca GG (2016) Brazilian savannah fruits: characteristics, properties, and potential applications. Food Sci Biotechnol 25:1225–1232. https://doi.org/10.1007/s10068-016-0195-3
Silva RO, Batistote M, Cereda MP (2011) Wild strains of fermenting yeast isolated of sugar cane juice from an alcohol distillery from Mato Grosso, Brazil. J Biotechnol Biodivers 2:22–27
Silva S, Negri M, Henriques M, Oliveira R, Williams DW, Azeredo J (2012) Candida glabrata, Candida parapsilosis and Candida tropicalis: biology, epidemiology, pathogenicity and antifungal resistance. FEMS Microbiol Rev 36:288–305. https://doi.org/10.1111/j.1574-6976.2011.00278.x
Silva-Filho EA, Santos SKB, Resende AM, Morais JOF, Morais Júnior MA, Simões DA (2005) Yeast population dynamics on industrial fuel ethanol fermentation processes assessed by PCR finger printing. Anton Leew 88:13–23. https://doi.org/10.1007/s10482-004-7283-8
Sonnleitner B, Kappeli O (1986) Growth of Saccharomyces cerevisiae is controlled by its limited respiratory capacity: formulation and verification of a hypothesis. Biotechnol Bioeng 28:927–937. https://doi.org/10.1002/bit.260280620
Teixeira MC, Raposo LR, Mira NP, Lourenço AB, Sá-Correia I (2009) Genome-wide identification of Saccharomyces cerevisiae genes required for maximal tolerance to ethanol. Appl Environ Microbiol 75:5761–5772. https://doi.org/10.1128/AEM.00845-09
Tronchoni J, Gamero A, Arroyo-López FN, Barrio E, Querol A (2009) Differences in the glucose and fructose consumption profiles in diverse Saccharomyces wine species and their hybrids during grape juice fermentation. Int J Food Microbiol 134:237–243. https://doi.org/10.1016/j.ijfoodmicro.2009.07.004
Verduyn C, Postma E, Scheffers WA, Van Dijken JP (1992) Effect of benzoic acid on metabolic fluxes in yeasts: a continuous-culture study on the regulation of respiration an alcoholic fermentation. Yeast 8:501–517. https://doi.org/10.1002/yea.320080703
Wang D, Xu Y, Hu J, Zhao G (2004) Fermentation kinetics of different sugars by the apple wine yeast Saccharomyces cerevisiae. J Inst Brew 110:340–346. https://doi.org/10.1002/j.2050-0416.2004.tb00630.x
Wang X, Ike M, Shiroma R, Tokuyasu K, Sakakibara Y (2013) Expression of neutral β-glucosidase from Scytalidium thermophilum in Candida glabrata for ethanol production from alkaline-pretreated rice straw. J Biosci Bioeng 116:362–365. https://doi.org/10.1016/j.jbiosc.2013.03.007
Wang XC, Li AH, Dizy M, Ullah N, Sun WX, Tao YS (2017) Evaluation of aroma enhancement for ‘‘Ecolly” dry white wines by mixed inoculation of selected Rhodotorula mucilaginosa and Saccharomyces cerevisiae. Food Chem 228:550–555. https://doi.org/10.1016/j.foodchem.2017.01.11
Acknowledgements
The authors gratefully acknowledge the Pro-rectory of Research and Post-graduation of the Federal University of Grande Dourados and the Brazilian research funding agencies CAPES, CNPq, and FUNDECT for their financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Andrade Silva, C.A., Oka, M.L. & Fonseca, G.G. Physiology of yeast strains isolated from Brazilian biomes in a minimal medium using fructose as the sole carbon source reveals potential biotechnological applications. 3 Biotech 9, 191 (2019). https://doi.org/10.1007/s13205-019-1721-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-019-1721-9