Abstract
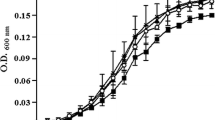
Quorum sensing or cell to cell communication which includes inter- and intra-cellular communication has been implicated in the production of virulence factor and formation of biofilm in food-borne pathogens. In the present study, the effect of quorum sensing signals on the biofilms of food-borne pathogens has been elucidated. N-butryl homoserine lactone and N-hexanoyl homoserine lactone belonging to acyl homoserine lactone (AHL) family of signaling molecules were investigated for their effect on the biofilm formation (attachment and exopolymeric substance production) in the food-borne pathogens Escherichia coli, Salmonella enterica serovar Typhimurium and Vibrio parahemolyticus. The signaling molecules at a concentration of 1 µM were capable of increasing biofilm formation in all the tested pathogens. There was an increase in the attachment of the bacterial cells and biomass as observed by microtiter plate assay and exopolymeric substances production in the biofilms in presence of the AHLs. Further, it needs to be elucidated if the effect of AHLS on the biofilms of E. coli and S. enterica serovar Typhimurium is SdiA dependent.

Similar content being viewed by others
References
Bai JA, Rai VR (2014) Quorum sensing inhibitory and anti-biofilm activity of essential oils and their in vivo efficacy in food systems. Food Biotech 28:269–292
Bridier A, Sanchea-Vizuete P, Guilbaud M, Piard JC, Naitali M, Briandet R (2015) Biofilm-associated persistence of food-borne pathogens. Food Microbiol 45:167–178
Davies DG, Parsek MR, Pearson JP, Iglewski BH, Costerton JW, Greenberg EP (1998) The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science 280:295–298
Dyszel JL, Soares JA, Swearingen MC, Lindsay A, Smith JN et al (2010) E coli K-12 and EHEC Genes regulated by SdiA. PLoS ONE 5:e8946
Dziva F, van Diemen PM, Stevens MP, Smith AJ, Wallis TS (2004) Identification of Escherichia coli O157: H7 genes influencing colonization of the bovine gastrointestinal tract using signature-tagged mutagenesis. Microbiology 150:3631–3645
Flemming HC, Wingender J (2010) The biofilm matrix. Nat Rev Microbiol 8:623–633
Gade-Portratz CJ, McCarter LL (2011) Quorum Sensing and Silencing in Vibrio parahaemolyticus. J Bacteriol 193:4224–4237
Giaouris E, Chorianopoulos N, Skandamia P, Nychas GJ (2012) Attachment and biofilm formation by salmonella in food processing environments. In: Mahmoud BSM (ed) Salmonella—A Dangerous Foodborne Pathogen. InTech, pp. 157–180
González Barrios AF, Zuo R, Hashimoto Y, Yang L, Bentley WE, Wood TK (2006) Autoinducer 2 controls biofilm formation in Escherichia coli through a novel motility quorum sensing regulator (MqsR, B3022). J Bacteriol 188:305–316
Hammer BK, Bassler BL (2003) Quorum sensing controls biofilm formation in Vibrio cholerae. Mol Microbiol 50:101–104
Jaglic Z, Desvaux M, Weiss A, Nesse LL, Meyer RL, Demnerova K, Schimdt H, Giaouris E, Sipailiene A, Teixeira P, Kacaniova M, Riedel CU, Knochel S (2014) Surface adhesins and exopolymers of selected foodborne pathogens. Microbiol 160:2561–2582
Janssens JCA, Steenackers H, Robijns S, Gellens E, Levin J, Zhao H, Hermans K, De Coster D, Verhoeven TL, Marchal K, Vanderleyden J, De Vos DE, De Keersmaecker SCJ (2008) Brominated Furanones Inhibit Biofilm Formation by Salmonella enterica serovar Typhimurium. Appl Environ Microbiol 74:6639–6648
Lee J, Page R, Garcia-Contreras R, Palermino JM, Zhang XS, Doshi O, Wood TK, Peti W (2007) Structure and function of the Escherichia coli protein YmgB: a protein critical for biofilm formation and acid-resistance. J Mol Biol 373:11–26
Lee J, Maeda T, Hong SH, Wood TK (2009) Reconfiguring the quorum sensing regulator SdiA of Escherichia coli to control biofilm formation via indole and N-acylhomoserine lactones. Appl Environ Microbiol 75:1703–1716
Ng WL, Bassler BL (2009) Bacterial quorum sensing network architectures. Annu Rev Genet 43:197–222
O’Toole GA, Kolter R (1998) Initiation of biofilm formation in Pseudomonas fluorescens CS365 proceeds via multiple, convergent signaling pathways: a genetic analysis. Mol Microbiol 28:449–461
Patankar AV, Gonzalez JE (2009) Orphan luxr regulators of quorum sensing. FEMS Microbiol Rev 33:739–756
Rosselin M, Virlogeux-Payant I, Roy C, Bottreau E, Sizaret PY, Mijouin L, Germon P, Caron E, Velge P, Wiedemann A (2010) Rck of Salmonella enterica, subspecies enterica serovar enteritidis, mediates zipper-like internalization. Cell Res 20:647–664
Rutherford ST, van Kessel JC, Shao Y, Bassler BL (2011) AphA and LuxR/HapR reciprocally control quorum sensing in vibrios. Genes Dev 25:397–408
Samudrala R, Heffron F, McDermott JE (2009) Accurate prediction of secreted substrates and identification of a conserved putative secretion signal for type III secretion systems. PLOS Pathogen 5:1000375
Sharma VK, Bearson SM (2013) Evaluation of the impact of quorum sensing transcriptional regulator SdiA on long-term persistence and fecal shedding of Escherichia coli O157:H7 in weaned calves. Microb Pathog 57:21–26
Sharma VK, Bearson SM, Bearson BL (2010) Evaluation of the effects of sdiA, a luxR homologue, on adherence and motility of Escherichia coli O157: H7. Microbiol 156:1303–1312
Smith JM, Ahmer BM (2003) Detection of other microbial species by Salmonella: expression of the sdia regulon. J Bacteriol 185:1357–1366
Smith JL, Fratamico PM, Yan X (2011) Eavesdropping by bacteria: the role of SdiA in Escherichia coli and Salmonella enterica serovar Typhimurium quorum sensing. Foodborne Pathogen Dis 8:169–178
Soares JA, Ahmer BMM (2011) Detection of acyl-homoserine lactones by Escherichia and Salmonella. Curr Opin Microbiol 14:188–193
Steenackers H, Hermans K, Vanderleyden J, De Keersmaecker SCJ (2012) Salmonella biofilms: an overview on occurrence, structure, regulation and eradication. Food Res Int 45:502–531
Van Houdt R, Aertsen A, Moons P, Vanoirbeek K, Michiels CW (2006) N-acyl-l-homoserine lactone signal interception by Escherichia coli. FEMS Microbiol 256:83–89
Wang L, Ling Y, Jiang H, Qiu Y, Qiu J, Chen H, Yang R, Zhou D (2013) AphA is required for biofilm formation, motility, and virulence in pandemic Vibrio parahemolyticus. Int J Food Microbiol 160:245–251
Winkelstroter LK, Teixeira FBR, Silva EP, Alves VF, De Martinis ECP (2014) Unraveling microbial biofilms of importance for food microbiology. Microbial Eco. 68:35–46
Yao Y, Martinez-Yamout MA, Dickerson TJ, Brogan AP, Wright PE, Dyson HJ (2006) Structure of the Escherichia coli Quorum Sensing Protein SdiA: activation of the folding switch by Acyl Homoserine Lactones. J Mol Biol 355(2):262–273
Yeom J, Lee Y, Park W (2012) Effects of non-ionic solute stresses on biofilm formation and lipopolysaccharide production in Escherichia coli O157:H7. Res Microbiol 163:258–267
Yildiz FH, Visick KL (2009) Vibrio biofilms: so much the same yet so different. Trends Microbiol 17:109–118
Zhang Y, Qiu Y, Tan Y, Guo Z, Yang R, Zhou D (2012) Transcriptional regulation of opaR, qrr2-4and aphA by the master quorum sensing regulator OpaR in Vibrio parahaemolyticus. PLoS ONE 7:34622
Acknowledgments
This work was supported by the Indian Council of Medical Research through the Senior Research Fellowship (F.No. 80/709/2011-ECD-I dated 21/06/2012).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
A., J.B., V., R.R. Effect of small chain N acyl homoserine lactone quorum sensing signals on biofilms of food-borne pathogens. J Food Sci Technol 53, 3609–3614 (2016). https://doi.org/10.1007/s13197-016-2346-1
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-016-2346-1