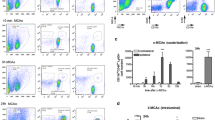
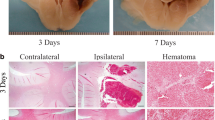
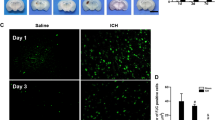
Abstract
Polymorphonuclear neutrophils (PMNs) infiltration into brain parenchyma after cerebrovascular accidents is viewed as a key component of secondary brain injury. Interestingly, a recent study of ischemic stroke suggests that after ischemic stroke, PMNs do not enter brain parenchyma and as such may cause no harm to the brain. Thus, the present study was designed to determine PMNs’ behavior after intracerebral hemorrhage (ICH). Using the autologous blood injection model of ICH in rats and immunohistochemistry for PMNs and vascular components, we evaluated the temporal and spatial PMNs distribution in the ICH-affected brain. We found that, similar to ischemia, there is a robust increase in presence of PMNs in the ICH-injured tissue that lasts for at least 1 to 2 weeks. However, in contrast to what was suggested for ischemia, besides PMNs that stay in association with the vasculature, after ICH, we found abundance of intraparenchymal PMNs (with no obvious association with vessels) in the ICH core and hematoma border, especially between 1 and 7 days after the ictus. Interestingly, the increased presence of intraparenchymal PMNs after ICH coincided with the massive loss of microvascular integrity, suggesting vascular disruption as a potential cause of PMNs presence in the brain parenchyma. Our study indicates that in contrast to ischemic stroke, after ICH, PMNs target not only vascular compartment but also brain parenchyma in the affected brain. As such, it is possible that the pathogenic role and therapeutic implications of targeting PMNs after ICH could be different from these after ischemic stroke. Our work suggests the needs for more studies addressing the role of PMNs in ICH.





Similar content being viewed by others
References
Qureshi AI, Mendelow AD, Hanley DF. Intracerebral haemorrhage. Lancet. 2009;373:1632–44.
Aronowski J, Zhao X. Molecular pathophysiology of cerebral hemorrhage: secondary brain injury. Stroke. 2011;42:1781–6.
Keep RF, Hua Y, Xi G. Intracerebral haemorrhage: mechanisms of injury and therapeutic targets. Lancet Neurol. 2012;11:720–31.
Ravichandran KS. Find-me and eat-me signals in apoptotic cell clearance: progress and conundrums. J Exp Med. 2010;207:1807–17.
Elliott MR, Ravichandran KS. Clearance of apoptotic cells: implications in health and disease. J Cell Biol. 2010;189:1059–70.
Chekeni FB, Ravichandran KS. The role of nucleotides in apoptotic cell clearance: implications for disease pathogenesis. J Mol Med (Berlin). 2011;89:13–22.
Aronowski J, Hall CE. New horizons for primary intracerebral hemorrhage treatment: experience from preclinical studies. Neurol Res. 2005;27:268–79.
del Zoppo GJ, Schmid-Schonbein GW, Mori E, Copeland BR, Chang CM. Polymorphonuclear leukocytes occlude capillaries following middle cerebral artery occlusion and reperfusion in baboons. Stroke. 1991;22:1276–83.
Gong C, Hoff JT, Keep RF. Acute inflammatory reaction following experimental intracerebral hemorrhage in rat. Brain Res. 2000;871:57–65.
Weston RM, Jones NM, Jarrott B, Callaway JK. Inflammatory cell infiltration after endothelin-1-induced cerebral ischemia: histochemical and myeloperoxidase correlation with temporal changes in brain injury. J Cereb Blood Flow Metab. 2007;27:100–14.
Jin R, Yang G, Li G. Inflammatory mechanisms in ischemic stroke: role of inflammatory cells. J Leukoc Biol. 2010;87:779–89.
Soehnlein O. An elegant defense: how neutrophils shape the immune response. Trends Immunol. 2009;30:511–2.
Garcia JH, Liu KF, Yoshida Y, Lian J, Chen S, del Zoppo GJ. Influx of leukocytes and platelets in an evolving brain infarct (Wistar rat). Am J Pathol. 1994;144:188–99.
Matsuo Y, Kihara T, Ikeda M, Ninomiya M, Onodera H, Kogure K. Role of neutrophils in radical production during ischemia and reperfusion of the rat brain: effect of neutrophil depletion on extracellular ascorbyl radical formation. J Cereb Blood Flow Metab. 1995;15:941–7.
Kalimo H, del Zoppo GJ, Paetau A, Lindsberg PJ. Polymorphonuclear neutrophil infiltration into ischemic infarctions: myth or truth? Acta Neuropathol. 2013;125:313–6.
Dawson DA, Ruetzler CA, Carlos TM, Kochanek PM, Hallenbeck JM. Polymorphonuclear leukocytes and microcirculatory perfusion in acute stroke in the SHR. Keio J Med. 1996;45:248–52. discussion 252–243.
Hallenbeck JM, Dutka AJ, Tanishima T, Kochanek PM, Kumaroo KK, Thompson CB, et al. Polymorphonuclear leukocyte accumulation in brain regions with low blood flow during the early postischemic period. Stroke. 1986;17:246–53.
Sansing LH, Harris TH, Kasner SE, Hunter CA, Kariko K. Neutrophil depletion diminishes monocyte infiltration and improves functional outcome after experimental intracerebral hemorrhage. Acta Neurochir Suppl. 2011;111:173–8.
Loftspring MC, Johnson HL, Johnson AJ, Clark JF. Depletion of gr-1-positive cells is associated with reduced neutrophil inflammation and astrocyte reactivity after experimental intracerebral hemorrhage. Transl Stroke Res. 2012;3:147–54.
Investigators EAST. Use of anti-icam-1 therapy in ischemic strokes: results of the enlimomab acute stroke trial. Neurology. 2001;57:1428–34.
Becker KJ. Anti-leukocyte antibodies: Leukarrest (hu23f2g) and enlimomab (r6.5) in acute stroke. Curr Med Res Opin. 2002;18(2):s18–22.
Krams M, Lees KR, Hacke W, Grieve AP, Orgogozo JM, Ford GA. Acute stroke therapy by inhibition of neutrophils (astin): an adaptive dose-response study of UK-279,276 in acute ischemic stroke. Stroke. 2003;34:2543–8.
Beray-Berthat V, Palmier B, Plotkine M, Margaill I. Neutrophils do not contribute to infarction, oxidative stress, and no synthase activity in severe brain ischemia. Exp Neurol. 2003;182:446–54.
Emerich DF, Dean 3rd RL, Bartus RT. The role of leukocytes following cerebral ischemia: pathogenic variable or bystander reaction to emerging infarct? Exp Neurol. 2002;173:168–81.
Fassbender K, Ragoschke A, Kuhl S, Szabo K, Fatar M, Back W, et al. Inflammatory leukocyte infiltration in focal cerebral ischemia: unrelated to infarct size. Cerebrovasc Dis. 2002;13:198–203.
Hayward NJ, Elliott PJ, Sawyer SD, Bronson RT, Bartus RT. Lack of evidence for neutrophil participation during infarct formation following focal cerebral ischemia in the rat. Exp Neurol. 1996;139:188–202.
Takeshima R, Kirsch JR, Koehler RC, Gomoll AW, Traystman RJ. Monoclonal leukocyte antibody does not decrease the injury of transient focal cerebral ischemia in cats. Stroke. 1992;23:247–52.
Enzmann G, Mysiorek C, Gorina R, Cheng YJ, Ghavampour S, Hannocks MJ, et al. The neurovascular unit as a selective barrier to polymorphonuclear granulocyte (PMN) infiltration into the brain after ischemic injury. Acta Neuropathol. 2013;125:395–412.
Moxon-Emre I, Schlichter LC. Neutrophil depletion reduces blood–brain barrier breakdown, axon injury, and inflammation after intracerebral hemorrhage. J Neuropathol Exp Neurol. 2011;70:218–35.
Patel S, de la Fuente J, Atra A, Senevirathne S, Cale CM. Where have all the neutrophils gone? Arch Dis Child Educ Pract Ed. 2009;94:74–7.
Yang S, Nakamura T, Hua Y, Keep RF, Younger JG, He Y, et al. The role of complement C3 in intracerebral hemorrhage-induced brain injury. J Cereb Blood Flow Metab. 2006;26:1490–5.
Xue M, Del Bigio MR. Intracerebral injection of autologous whole blood in rats: time course of inflammation and cell death. Neurosci Lett. 2000;283:230–2.
Zhao X, Sun G, Zhang J, Strong R, Song W, Gonzales N, et al. Hematoma resolution as a target for intracerebral hemorrhage treatment: role for peroxisome proliferator-activated receptor gamma in microglia/macrophages. Ann Neurol. 2007;61:352–62.
Hickenbottom SL, Grotta JC, Strong R, Denner LA, Aronowski J. Nuclear factor-kappab and cell death after experimental intracerebral hemorrhage in rats. Stroke. 1999;30:2472–7. discussion 2477–2478.
Felberg RA, Grotta JC, Shirzadi AL, Strong R, Narayana P, Hill-Felberg SJ, et al. Cell death in experimental intracerebral hemorrhage: the “black hole” model of hemorrhagic damage. Ann Neurol. 2002;51:517–24.
Zhao X, Zhang Y, Strong R, Grotta JC, Aronowski J. 15d-prostaglandin j2 activates peroxisome proliferator-activated receptor-gamma, promotes expression of catalase, and reduces inflammation, behavioral dysfunction, and neuronal loss after intracerebral hemorrhage in rats. J Cereb Blood Flow Metab. 2006;26:811–20.
Pinkus GS, Pinkus JL. Myeloperoxidase: a specific marker for myeloid cells in paraffin sections. Mod Pathol. 1991;4:733–41.
Gotoh S, Itoh M, Fujii Y, Arai S, Sendo F. Enhancement of the expression of a rat neutrophil-specific cell surface antigen by activation with phorbol myristate acetate and concanavalin a. J Immunol. 1986;137:643–50.
Skrajnar S, Anzur Lasnik M, Bedina ZA. A flow cytometric method for determination of the blood neutrophil fraction in rats. J Am Assoc Lab Anim Sci. 2009;48:152–6.
Arkema JM, Schadee-Eestermans IL, Beelen RH, Hoefsmit EC. A combined method for both endogenous myeloperoxidase and acid phosphatase cytochemistry as well as immunoperoxidase surface labelling discriminating human peripheral blood-derived dendritic cells and monocytes. Histochemistry. 1991;95:573–8.
de Mendez I, Young Jr KR, Bignon J, Lambre CR. Biochemical characteristics of alveolar macrophage-specific peroxidase activities in the rat. Arch Biochem Biophys. 1991;289:319–23.
Lifshitz V, Benromano T, Weiss R, Blanga-Kanfi S, Frenkel D. Insulin-degrading enzyme deficiency accelerates cerebrovascular amyloidosis in an animal model. Brain Behav Immun. 2012
Tu YF, Tsai YS, Wang LW, Wu HC, Huang CC, Ho CJ. Overweight worsens apoptosis, neuroinflammation and blood–brain barrier damage after hypoxic ischemia in neonatal brain through JNK hyperactivation. J Neuroinflammation. 2011;8:40.
Wang LW, Tu YF, Huang CC, Ho CJ. JNK signaling is the shared pathway linking neuroinflammation, blood–brain barrier disruption, and oligodendroglial apoptosis in the white matter injury of the immature brain. J Neuroinflammation. 2012;9:175.
Williams KC, Zhao RW, Ueno K, Hickey WF. Pecam-1 (CD31) expression in the central nervous system and its role in experimental allergic encephalomyelitis in the rat. J Neurosci Res. 1996;45:747–57.
Wang J, Tsirka SE. Tuftsin fragment 1–3 is beneficial when delivered after the induction of intracerebral hemorrhage. Stroke. 2005;36:613–8.
Wasserman JK, Schlichter LC. Neuron death and inflammation in a rat model of intracerebral hemorrhage: effects of delayed minocycline treatment. Brain Res. 2007;1136:208–18.
Amulic B, Cazalet C, Hayes GL, Metzler KD, Zychlinsky A. Neutrophil function: from mechanisms to disease. Annu Rev Immunol. 2012;30:459–89.
Acknowledgments
This work was partially supported by the National Institutes of Health, NINDS grants NS060768 and NS064109.
Compliance with Ethics Requirements
All institutional and national guidelines for the care and use of laboratory animals were followed.
Conflict of Interest
All authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, X., Sun, G., Zhang, H. et al. Polymorphonuclear Neutrophil in Brain Parenchyma After Experimental Intracerebral Hemorrhage. Transl. Stroke Res. 5, 554–561 (2014). https://doi.org/10.1007/s12975-014-0341-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-014-0341-2