Abstract
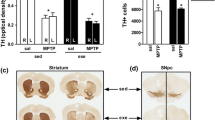
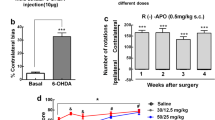
Two experiments were performed to investigate the effects of physical exercise upon the hypokinesia induced by two different types of MPTP administration to C57/BL6 mice. In the first, mice were administered either the standard MPTP dose (2 × 20 or 2 × 40 mg/kg, 24-h interval) or vehicle (saline, 5 ml/kg); and over the following 3 weeks were given daily 30-min period of wheel running exercise over five consecutive days/week or placed in a cage in close proximity to the running wheels. Spontaneous motor activity testing in motor activity test chambers indicated that exercise attenuated the hypokinesic effects of both doses of MPTP upon spontaneous activity or subthreshold l-Dopa-induced activity. In the second experiment, mice were either given wheel running activity on four consecutive days (30-min period) or placed in a cage nearby and on the fifth day, following motor activity testing over 60 min, injected with either MPTP (1 × 40 mg/kg) or vehicle. An identical procedure was maintained over the following 4 weeks with the exception that neither MPTP nor vehicle was injected after the fifth week. The animals were left alone (without either exercise or MPTP) and tested after 2- and 4-week intervals. Weekly exercise blocked, almost completely, the progressive development of severe hypokinesia in the MPTP mice and partially restored normal levels of activity after administration of subthreshold l-Dopa, despite the total absence of exercise following the fifth week. In both experiments, MPTP-induced loss of dopamine was attenuated by the respective regime of physical exercise with dopamine integrity more effectively preserved in the first experiment. The present findings are discussed in the context of physical exercise influences upon general plasticity and neuroreparative propensities as well as those specific for the nigrostriatal pathway.






Similar content being viewed by others
References
Ahmad AO, Park J-H, Stenho-Bittel L, Lau YS (2009) Effects of endurance exercise on ventral tegmental area neurons in the chronic 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine and probenecid-treated mice. Neurosci Lett 450:102–105
Aksu I, Topcu A, Camsari UM, Acikgoz O (2009) Effect of acute and chronic exercise on oxidant-antioxidant equilibrium in rat hippocampus, prefrontal cortex and striatum. Neurosci Lett 452:281–285
Al-Jarrah M, Pothakos K, Novikova L, Smirnova IV, Kurz MJ, Stenho-Bittel L, Lau YS (2007) Endurance exercise promotes cardiorespiratory rehabilitation without neurorestoration in the chronic mouse model of Parkinsonism with severe neurodegeneration. Neuroscience 149:28–37
Archer T, Fredriksson A (2003) An antihypokinesic action of α2-adrenoceptors upon MPTP-induced behavior deficits in mice. J Neural Transm 110:183–200
Archer T, Fredriksson A (2006) Influence of noradrenaline denervation upon MPTP-induced deficts in mice. J Neural Transm 113:1119–1129
Archer T, Fredriksson A (2007) Functional consequences of iron overload in catecholaminergic interactions: the Youdim factor. Neurochem Res 32:1625–1639
Archer T, Fredriksson A, Jonsson G, Lewander T, Mohammed AK, Ross SB, Söderberg U (1986) Central noradrenaline depletion antagonizes aspects of d-amphetamineinduced hyperactivity in the rat. Psychopharmacology 88:141–146
Balthazar CH, Leite LH, Rodrigues AG, Coimbra CC (2009) Performance-enhancing and thermoregulatory effects of intracerebroventricular dopamine in running rats. Pharmacol Biochem Behav 93:465–469
Belke W (1996) Investigating the reinforcing properties of running. In: Epling WF, Pierce WD (eds) Activity anorexia: theory, research, and treatment. Erlbaum, Mahwah, pp 45–55
Bilowit DS (1956) Establishing physical objectives in rehabilitation of patients with Parkinson’s disease. Phys Ther Rev 36:176–178
Björk L, Lindgren S, Hacksell U, Lewander T (1991) (S)-UH-301 antagonizes (R)-8-OH-DPAT-induced cardiovascular effects in the rat. Eur J Pharmacol 199:367–370
Cambras T, Vilaplana J, Campuzano A, Canal-Corretger MM, Carulla M, Diez-Noguera A (2000) Entrainment of the rat motor activity rhythm: effects of light-dark cycle and physical exercise. Physiol Behav 70:227–232
Challet E, Malan A, Pévet P (1996) Daily hypocaloric feeding entrains circadian rhythms of wheel-running and body temperature in rats kept at constant darkness. Neurosci Lett 211:1–4
Ciucci MR, Ma ST, Kane JR, Ahrens AM, Schallert T (2008) Limb use and complex ultrasonic vocalization in a rat model of Parkinson’s disease: deficit-targeted training. Parkinsonism Relat Disord 14(Suppl. 2):S172–S175
Cohen AD, Tillerson JL, Smith AD, Schallert T, Zigmond MJ (2003) Neuroprotective effects of prior limb use in 6-hydroxydopamine-treated rats: possible role of GDNF. J Neurochem 85:299–305
Cotman CW, Berchtold NC (2002) Exercise: a behavioural intervention to enhance brain health and plasticity. Trends Neurosci 25:295–301
de Visser L, van der Bos R, Spruijt BM (2005) Automated home cage observations as a tool to measure the effects of wheel running on cage floor locomotion. Behav Brain Res 160:382–388
de Visser L, van der Bos R, Stoker AK, Kas MJH, Spruijt BM (2007) Effects of genetic background and environmental novelty on wheel running as a rewarding behaviour in mice. Behav Brain Res 177:290–297
Decamp E, Schneider JS (2004) Attention and executive function deficits in chronic low-dose MPTP-treated non-human primates. Eur J Neurosci 20:1371–1378
Döbrössy MD, Dunnett SB (2001) The influence of environment and experience on neural grafts. Nat Rev Neurosci 2:871–879
Döbrössy MD, Dunnett SB (2003) Motor training effects on recovery of function after striatal lesions and striatal grafts. Exp Neurol 184:274–284
Dunn AL, Trivedi MH, Kampert JB, Clark CG, Chambliss HO (2005) Exercise treatment for depression: efficacy and dose response. Am J Prev Med 28:1–8
Fisher BE, Petzinger GM, Nixon K, Hogg E, Bremmer S, Meshul CK, Jakowec MW (2004) Exercise-induced behavioral recovery and neuroplasticity in the 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine-lesioned mouse basal ganglia. J Neurosci Res 77:378–390
Foley TE, Fleshner M (2008) Neuroplasticity of dopamine circuits after exercise: implications for central fatigue. Neuromol Med 10:67–80
Fox CM, Ramig LO, Ciucci MR, Sapir S, McFarland DH, Farley BG (2006) The science and practice of LSVT/LOUD: neural plasticity approach to treating individuals with Parkinson’s disease and other neurological disorders. Semin Speech Lang 27:283–299
Fredriksson A, Archer T (1994) MPTP-induced behavioural and biochemical deficits: a parametric analysis. J Neural Transm Dis Dement Sect 7:123–132
Fredriksson A, Archer T (2003) Effect of postnatal iron administration on MPTP-induced behavioural deficits and neurotoxicity: behavioural enhancement by L-Dopa-MK-801 co-administration. Behav Brain Res 139:31–46
Fredriksson A, Archer T (2007) Postnatal iron overload destroys NA-DA functional interactions. J Neural Transm 114:195–203
Fredriksson A, Gentsch C, Archer T (1994) Synergistic interactions between NMDA-antagonists and L-Dopa on activity in MPTP-treated mice. J Neural Transm [Gen Sect] 97:197–209
Fredriksson A, Plaznik A, Sundström E, Jonsson G, Archer T (1990) MPTP-induced hypoactivity in mice: reversal by L-Dopa. Pharmacol Toxicol 67:295–301
Fredriksson A, Palomo T, Chase TN, Archer T (1999) Tolerance to a suprathreshold dose of L-Dopa in MPTP mice: effects of glutamate antagonists. J Neural Transm 106:283–300
Fredriksson A, Schröder N, Eriksson P, Izquierdo I, Archer T (2001) Neonatal iron potentiates adult MPTP-induced neurodegenerative and functional deficits. Parkinsonism Relat Dis 7:97–105
German DC, Nelson EL, Liang CL, Speciale SG, Sinton CM, Sonsalla PK (1996) The neurotoxin MPTP causes degeneration of specific nucleus A8, A9 and A10 dopaminergic neurons in the mouse. Neurodegeneration 5:299–312
Gomez-Pinilla F, Ying Z, Roy RR, Molteni R, Edgerton VR (2002) Voluntary exercise induces a BDNF-mediated mechanism that promotes neuroplasticity. J Neurophysiol 88:2187–2195
Goodwin VA, Richards SH, Taylor RS, Taylor AH, Campbell JL (2008) The effectiveness of exercise interventions for people with Parkinson’s disease: a systematic review and meta-analysis. Mov Dis 23:631–640
Harri M, Lindblom J, Malinen H, Hyttinen M, Lapvetelainen T, Escola S, Helminen HJ (1999) Effect of access to a running wheel on behaviour of C57Bl/6 J mice. Lab Anim Sci 49:401–405
Heikkila RE, Sieber B-A, Manzino L, Sonsalla PK (1989) Some features of the nigrostriatal dopaminergic neurotoxin 1-methyl-4-phenyl-1, 2, 3, 6-tetrohydropyridine (MPTP) in the mouse. Mol Chem Neuropathol 10:171–183
Hurwitz A (1989) The benefit of a home exercise regime for ambulatory Parkinson’s disease patients. J Neurosci Nurs 21:180–184
Jackson-Lewis V, Jakowec M, Burke RE, Przedborski S (1995) Time course and morphology of dopaminergic neuronal death cauesed by the neurotoxin 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine. Neurodegeneration 4:257–269
Jahanshahi M, Jenkins IH, Brown RG, Marsden CD, Passingham RE, Brooks DJ (1995) Self-initiated versus externally triggered movements. I. An investigation using measurement of regional cerebral blood flow with PET and movement-related potentials in normal and Parkinson’s disease subjects. Brain 118:913–933
Kirk R (1995) Experimental design: procedures for the behavioral sciences. Brooks/Cole, Belmont
Kurz MJ, Pothakos K, Jamaluddon S, Scott-Pandorf M, Arellano C, Lau Y-S (2007) A chronic mouse model of Parkinson’s disease has a reduced gait pattern certainty. Neurosci Lett 429:39–42
Langston JW (1985) MPTP neurotoxicity: an overview and characterization of phases of toxicity. Life Sci 36:201–206
Lau YS (2005) Progressive neurodegeneration in the chronic MPTP/probenecid model of Parkinson’s disease. In: Ebadi M, Pfeiffer R (eds) Parkinson’s disease. CRC Press, Boca Raton, pp 109–115
Laurin D, Verreault R, Lindsay J, MacPherson K, Rockwood K (2001) Physical activity and risk of cognitive impairment and dementia in elderly persons. Arch Neurol 58:498–504
Lett BT, Grant VL, Byrne MJ, Koh MT (2000) Pairings of a distinctive chamber with the after effect of wheel running produce conditioned place preference. Appetite 34:87–94
Lightfoot JT, Turner MJ, Daves M, Vordermark A, Kleeberger SR (2004) Genetic influence on daily wheel running activity level. Physiol Genomics 19:270–276
Liu Y, Yu H, Mohell N, Nordvall G, Lewander T, Hacksell U (1995) Derivatives of cis-2-amino-8-hydroxy-1-methyltetralin: mixed 5-HT1A-receptor agonists and dopamine D2-receptor antagonists. J Med Chem 38:150–160
Meek TH, Lonquich BP, Hannon RM, Garland T (2009) Endurance capacity of mice selectively bred for high voluntary wheel running. J Exp Biol 212:2908–2917
Miyai I, Fujimoto Y, Ueda Y, Yamamoto H, Nozaki S, Saito T, Kang J (2000) Treadmill training with body weight support: its effect on Parkinson’s disease. Arch Phys Med Rehabil 81:849–852
Morris M, Schoo A (2004) Optimizing exercise and physical activity in older adults. Butterworth Heinemann, Edinburgh
Muhlack S, Welnic J, Woitalla D, Müller T (2007) Exercise improves efficacy of levodopa in patients with Parkinson’s disease. Mov Dis 22:427–430
Novikova L, Garris BL, Garris DR, Lau YS (2006) Early signs of neuronal apoptosis in the substantia nigra pars compacta of the progressive neurodegenerative mouse 1.methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine/probenecid model of Parkinson’s disease. Neuroscience 140:67–76
O’Dell SJ, Gross NB, Fricks AN, Casiano BD, Nguyen TB, Marshall JF (2007) Running wheel exercise enhances recovery from nigrostriatal dopamine injury without inducing neuroprotection. Neuroscience 144:1141–1151
Palmer SS, Mortimer JA, Webster DD, Bistevins R, Dickinson GL (1986) Exercise therapy for Parkinson’s disease. Arch Phys Med Rehab 67:741–745
Petroske E, Meredith GE, Callen S, Totterdell S, Lau YS (2001) Mouse model of Parkinsonism: a comparison between subacute MPTP and chronic MPTP/probenecid treatment. Neuroscience 106:589–601
Petzinger GM, Walsh JP, Akopian G, Hogg E, Abernathy A, Arevalo P, Turnquist P, Vuckovic M, Fisher BE, Togasaki DM, Jakowec MW (2007) Effects of treadmill exercise on dopaminergic transmission in the 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine-lesioned mouse model of basal ganglia injury. J Neurosci 27:52291–52300
Pohl M, Rockstroh G, Ruckreim S, Mrass G, Mehrholz J (2003) Immediate effects of speed-dependent treadmill training on gait parameters in early Parkinson’s disease. Arch Phys Med Rehabil 84:1760–1766
Pothakos K, Kurz MJ, Lau YS (2009) Restorative effect of endurance exercise on behavioral deficits in the chronic mouse model of Parkinson’s disease with severe neurodegeneration. BMC Neurosci 10:1–14
Ridgel AL, Vitek JL, Alberts JL (2009) Forced, not voluntary, exercise improves motor function in Parkinson’s disease patients. Neurorehabil Neural Repair 23:600–608
Sabatini U, Boulanouar K, Fabre N et al (2000) Cortical motor reorganisation in akinetic patients with Parkinson’s disease: a functional MRI study. Brain 123:394–403
Sherwin CM (1998a) The use and perceived importance of three resources which provide caged laboratory mice the opportunity for extended locomotion. Appl Anim Behav Sci 55:353–367
Sherwin CM (1998b) Voluntary wheel running: a review and novel interpretation. Anim Behav 56:11–27
Smith AD, Zigmond MJ (2003) Can the brain be protected through exercise? Lessons from an animal model of parkinsonism. Exp Neurol 184:31–39
Sonsalla PK, Heikkila RE (1986) The influence of dose and dosing interval on MPTP-induced dopaminergic neurotoxicity in mice. Eur J Pharmacol 129:339–345
Sundström E, Fredriksson A, Archer T (1990) Chronic neurochemical and behavioural changes in MPTP-lesioned C57 BL/6 mice: a model for Parkinson’s disease. Brain Res 528:181–188
Swallow JG, Carter PA, Garland T (1998a) Artificial selection for increased wheel-running behavior in house mice. Behav Genet 28:227–237
Swallow JG, Carter PA, Zhan WZ, Sieck GC (1998b) Effects of voluntary activity and genetic selection on aerobic capacity in house mice (Mus domesticus). J Appl Physiol 84:69–76
Teixeira AM, Trevizol F, Colpo G, Garcia SC, Charão M, Pereira RP, Fachinetto R, Rocha JB, Bürger ME (2008) Influence of chronic exercise on reserpine-induced oxidative stress in rats: behavioral and antioxidant evaluations. Pharmacol Biochem Behav 88:465–472
Tetrud JW, Langston JW, Redmond DE, Roth RH, Sladek JR, Angel RW (1986) MPTP-induced tremor in human and non-human primates. Neurology 36(Suppl. 1):308
Tillerson JL, Cohen AD, Philpower J, Miller GW, Zigmond MJ, Schallert T (2001) Forced limb-use effects on the behavioral and neurochemical effects of 6-hydroxydopamine. J Neurosci 21:4427–4435
Tillerson JL, Caudle WM, Reveron ME, Miller GW (2002) Detection of behavioral impairments correlated to neurochemical deficits in mice treated with moderate doses of 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine. Exp Neurol 178:80–90
Tillerson JL, Caudle WM, Reveron ME, Miller GW (2003) Exercise induces behavioral recovery and attenuates neurochemical deficits in rodent models of Parkinson’s disease. Neuroscience 119:899–911
Toole T, Maitland CG, Warren E, Hubmann MF, Panton L (2005) The effects of loading and unloading treadmill walking on balance, gait, fall risk, and daily function in Parkinsonism. NeuroRehabilitation 20:307–322
Turner MJ, Kleeberger SR, Lightfoot JT (2005) Influence of genetic background on daily running-wheel activity differs with aging. Physiol Genomics 22:76–85
Van Eijkeren FJM, Reijmers RSJ, Kleinveld MJ, Minten A, ter Bruggen JP, Bloem BR (2008) Nordic walking improves mobility in Parkinson’s disease. Mov Dis 23:2239–2243
Van Gelder BM, Tijhuis MA, Kalmijn S, Giampeoli S, Nissinen A, Kromhout D (2004) Physical activity in relation to cognitive decline in elderly men: the FINE study. Neurology 63:2316–2321
Vargas-Perez H, Borelli E, Diaz J-L (2004) Wheel running use in dopamine D2L receptor knockout mice. Neurosci Lett 366:172–175
Vucković MG, Wood RI, Holschneider DP, Abernathy A, Togasaki DM, Smith A, Petzinger GM, Jakowec MW (2008) Memory, mood, dopamine, and serotonin in the 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine-lesioned mouse model of basal ganglia injury. Neurobiol Dis 32:319–327
Werme M, Thoren P, Olson L, Brene S (2000) Running and cocaine both upregulate dynorphin mRNA in medial caudate putamen. Eur J Neurosci 12:2967–2974
Wichmann T, DeLong MR (1996) Functional and pathophysiological models of the basal ganglia. Curr Opin Neurobiol 6:751–758
Yoon MC, Shin MS, Kim TS, Kim BK, Ko IG, Sung YH, Kim SE, Lee HH, Kim YP, Kim CJ (2007) Treadmill exercise suppresses nigrostriatal dopaminergic neuronal loss in 6-hydroxydopamine-induced Parkinson’s rats. Neurosci Lett 423:12–17
Yousefi B, Tadibi V, Khoei AF and Montazeri A (2009) Exercise therapy, quality of life, and activities of daily living in patients with Parkinson disease: a small scale quasi-randomised trial. Trials 10:67
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Archer, T., Fredriksson, A. Physical Exercise Attenuates MPTP-Induced Deficits in Mice. Neurotox Res 18, 313–327 (2010). https://doi.org/10.1007/s12640-010-9168-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-010-9168-0