Abstract
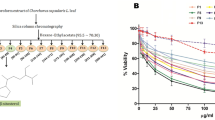
Looming drug resistance cases of leishmaniasis infection are an undeniably serious danger to worldwide public health, also jeopardize the efficacy of available drugs. Besides this, no successful vaccine is available till date. Since the ancient era, many plants and their parts have been used as medicines against various ailments. Hence, the importance of drug development for new molecules against Leishmania infection is significant that is a cost-effective and safer drug preferably from the natural herbal resources. We evaluated the GC–MS screening and efficacy of Putranjiva roxburghii (PR) against the sensitive and resistant promastigotes of L. donovani. GC–MS profiling revealed that the extract was rich in myo-inositol-4-C-methyl, azulene and desulphosinigrin. Quantitative investigation of phytoconstituents confirmed that PR was rich in phenols, flavonoids and terpenoids. We found an IC50 25.61 ± 0.57 µg/mL and 29.02 ± 1.21 µg/mL of PR against sodium stibogluconate sensitive and resistant strain respectively. It was found to be safer in cytotoxicity assay and generated ROS mediated oxidative stress in the parasitic cells which was evidenced by the increased and decreased levels of superoxide radicals, lipid peroxidation products, lipid bodies and levels of thiol, plasma membrane integrity respectively. Therefore, our results support the importance of P. roxburghii as a medicinal plant against L. donovani and showed potential for exploration as an antileishmanial agent.








Similar content being viewed by others
References
Abdel Moneim AE (2016) Indigofera oblongifolia prevents lead acetate-induced hepatotoxicity, oxidative stress, fibrosis and apoptosis in rats. PLoS ONE 11:1–18. https://doi.org/10.1371/journal.pone.0158965
Akya A, Chegene Lorestani R, Zhaleh H et al (2020) Effect of Vigna radiata, Tamarix ramosissima and Carthamus lanatus extracts on Leishmania major and Leishmania tropica: an in vitro study. Chin Herb Med 12:171–177. https://doi.org/10.1016/j.chmed.2019.12.006
Ayukekbong JA, Ntemgwa M, Atabe AN (2017) The threat of antimicrobial resistance in developing countries: causes and control strategies. Antimicrob Resist Infect Control 6:1–8. https://doi.org/10.1186/s13756-017-0208-x
Baba SP, Bhatnagar A (2018) Role of thiols in oxidative stress. Curr Opin Toxicol 7:133–139. https://doi.org/10.1016/j.cotox.2018.03.005
Balkrishna A, Sharma VK, Das SK et al (2020) Characterization and anti-cancerous effect of putranjiva roxburghii seed extract mediated silver nanoparticles on human colon (HCT-116), pancreatic (PANC-1) and breast (MDA-MB 231) cancer cell lines: a comparative study. Int J Nanomed 15:573–585. https://doi.org/10.2147/IJN.S230244
Britta EA, Scariot DB, Falzirolli H et al (2014) Cell death and ultrastructural alterations in Leishmania amazonensis caused by new compound 4-Nitrobenzaldehyde thiosemicarbazone derived from S-limonene. BMC Microbiol 14:1–12. https://doi.org/10.1186/s12866-014-0236-0
Bruno F, Castelli G, Migliazzo A et al (2015) Cytotoxic screening and in vitro evaluation of pentadecane against leishmania infantum promastigotes and amastigotes. J Parasitol 101:701–705. https://doi.org/10.1645/15-736
Bryceson A (2001) A policy for leishmaniasis with respect to the prevention and control of drug resistance. Trop Med Int Heal 6:928–934. https://doi.org/10.1046/j.1365-3156.2001.00795.x
Cataneo AHD, Tomiotto-Pellissier F, Miranda-Sapla MM et al (2019) Quercetin promotes antipromastigote effect by increasing the ROS production and anti-amastigote by upregulating Nrf2/HO-1 expression, affecting iron availability. Biomed Pharmacother 113:108745. https://doi.org/10.1016/j.biopha.2019.108745
Chakravarty J, Sundar S (2010) Drug resistance in leishmaniasis. J Glob Infect Dis 2:167. https://doi.org/10.4103/0974-777x.62887
Chandrasekaran S, Dayakar A, Veronica J et al (2013) An in vitro study of apoptotic like death in Leishmania donovani promastigotes by withanolides. Parasitol Int 62:253–261. https://doi.org/10.1016/j.parint.2013.01.007
Chouhan G, Islamuddin M, Want MY et al (2015) Apoptosis mediated leishmanicidal activity of Azadirachta indica bioactive fractions is accompanied by Th1 immunostimulatory potential and therapeutic cure in vivo. Parasit Vectors 8:1–24. https://doi.org/10.1186/s13071-015-0788-3
Cragg GM, Newman DJ (2013) Natural products: a continuing source of novel drug leads. Biochim Biophys Acta Gen Subj 1830:3670–3695. https://doi.org/10.1016/j.bbagen.2013.02.008
Croft SL, Sundar S, Fairlamb AH (2006) Drug resistance in leishmaniasis. Clin Microbiol Rev 19:111–126
da Silva Rodrigues JH, Miranda N, Volpato H et al (2019) The antidepressant clomipramine induces programmed cell death in Leishmania amazonensis through a mitochondrial pathway. Parasitol Res 118:977–989. https://doi.org/10.1007/s00436-018-06200-x
da Silva RRP, da Silva BJM, Rodrigues APD et al (2015) In vitro biological action of aqueous extract from roots of Physalis angulata against Leishmania (Leishmania) amazonensis. BMC Complement Altern Med 15:1–10. https://doi.org/10.1186/s12906-015-0717-1
Dantas AP, Barbosa HS, De Castro SL (2003) Biological and ultrastructural effects of the anti-microtubule agent taxol against Trypanosoma cruzi. J Submicrosc Cytol Pathol 35:287–294
Dar P, Faisal M, Dar A, Waqas U (2018) Journey describing biological activities and chemical constituents in the leaves, stem bark and seed of putranjiva roxburghii. Curr Tradit Med 4:263–278. https://doi.org/10.2174/2215083805666181206104450
Das A, Das MC, Das N, Bhattacharjee S (2017) Evaluation of the antileishmanial potency, toxicity and phytochemical constituents of methanol bark extract of Sterculia villosa. Pharm Biol 55:998–1009. https://doi.org/10.1080/13880209.2017.1285946
Das R, Roy A, Dutta N, Majumder HK (2008) Reactive oxygen species and imbalance of calcium homeostasis contributes to curcumin induced programmed cell death in Leishmania donovani. Apoptosis 13:867–882. https://doi.org/10.1007/s10495-008-0224-7
Dayakar A, Chandrasekaran S, Veronica J et al (2015) In vitro and in vivo evaluation of anti-leishmanial and immunomodulatory activity of Neem leaf extract in Leishmania donovani infection. Exp Parasitol 153:45–54. https://doi.org/10.1016/j.exppara.2015.02.011
De MacEdo Silva ST, Visbal G, Godinho JLP et al (2018) In vitro antileishmanial activity of ravuconazole, a triazole antifungal drug, as a potential treatment for leishmaniasis. J Antimicrob Chemother 73:2360–2373. https://doi.org/10.1093/jac/dky229
Monte-Neto de RL, Coelho AC, Raymond F et al (2011) Gene expression profiling and molecular characterization of antimony resistance in Leishmania amazonensis. PLoS Negl Trop Dis. https://doi.org/10.1371/journal.pntd.0001167
El-Khadragy M, Alolayan EM, Metwally DM et al (2018) Clinical efficacy associated with enhanced antioxidant enzyme activities of silver nanoparticles biosynthesized using moringa oleifera leaf extract, against cutaneous leishmaniasis in a murine model of leishmania major. Int J Environ Res Public Health 15:1037
Essid R, Rahali FZ, Msaada K et al (2015) Antileishmanial and cytotoxic potential of essential oils from medicinal plants in Northern Tunisia. Ind Crops Prod 77:795–802. https://doi.org/10.1016/j.indcrop.2015.09.049
Forni C, Facchiano F, Bartoli M et al (2019) Beneficial role of phytochemicals on oxidative stress and age-related diseases. Biomed Res Int. https://doi.org/10.1155/2019/8748253
Gannavaram S, Bhattacharya P, Ismail N et al (2016) Modulation of innate immune mechanisms to enhance Leishmania vaccine-induced immunity: role of coinhibitory molecules. Front Immunol 7:1–10. https://doi.org/10.3389/fimmu.2016.00187
Ghorai N, Ghorai N, Chakraborty S et al (2012) Estimation of total Terpenoids concentration in plant tissues using a monoterpene, Linalool as standard reagent. Protoc Exch. https://doi.org/10.1038/protex.2012.055
Goyal DK, Keshav P, Kaur S (2021a) Adjuvanted vaccines driven protection against visceral infection in BALB/c mice by Leishmania donovani. Microb Pathog. https://doi.org/10.1016/j.micpath.2021.104733
Goyal DK, Keshav P, Kaur S (2021b) Immune induction by adjuvanted Leishmania donovani vaccines against the visceral leishmaniasis in BALB/c mice. Immunobiology. https://doi.org/10.1016/j.imbio.2021.152057
Guerin PJ, Olliaro P, Sundar S, et al (2002) Visceral leishmaniasis: Current status of control, diagnosis, and treatment, and a proposed research and development agenda. Lancet Infect Dis 2:494–501. https://doi.org/10.1016/S1473-3099(02)00347-X
Gunjan S, Singh SK, Sharma T et al (2016) Mefloquine induces ROS mediated programmed cell death in malaria parasite: plasmodium. Apoptosis 21:955–964. https://doi.org/10.1007/s10495-016-1265-y
Gupta M (2016) A review of pharmacological properties, pharmacognosy and therapeutic actions of Putranjiva roxburghii Wall. (Putranjiva). Int J Herb Med 4:104–108
Hammi KM, Essid R, Tabbene O et al (2020) Antileishmanial activity of Moringa oleifera leaf extracts and potential synergy with amphotericin B. South Afr J Bot 129:67–73. https://doi.org/10.1016/j.sajb.2019.01.008
Harborne J (1973) Phytochemical methods in: a guide to modern techniques of plant analysis. Chapman and Hall Ltd, London
Hefnawy A, Berg M, Dujardin JC, Muylder GD (2017) Exploiting knowledge on Leishmania Drug resistance to support the quest for new drugs. Trend Parasitol 33:167–174
Islamuddin M, Sahal D, Afrin F (2013) Apoptosis-like death in Leishmania donovani promastigotes induced by eugenol-rich oil of Syzygium aromaticum. J Med Microbiol 63:74–85. https://doi.org/10.1099/jmm.0.064709-0
Kathuria M, Bhattacharjee A, Sashidhara KV et al (2014) Induction of mitochondrial dysfunction and oxidative stress in leishmania donovani by orally active clerodane diterpene. Antimicrob Agents Chemother 58:5916–5928. https://doi.org/10.1128/AAC.02459-14
Kaye PM, Cruz I, Picado A et al (2020) Leishmaniasis immunopathology—impact on design and use of vaccines, diagnostics and drugs. Semin Immunopathol 42:247–264. https://doi.org/10.1007/s00281-020-00788-y
Kumar N (2020) Phytochemistry and medicinal value of putranjiva roxburghii wall. In: Advances in pharmaceutical biotechnology: recent progress and future applications, p. 133–144.https://doi.org/10.1007/978-981-15-2195-9_11
Lee SJ, Zhang J, Choi AMK, Kim HP (2013) Mitochondrial dysfunction induces formation of lipid droplets as a generalized response to stress. Oxid Med Cell Longev. https://doi.org/10.1155/2013/327167
Magalhães LS, Bomfim LGS, Mota SG et al (2018) Increased thiol levels in antimony-resistant leishmania infantum isolated from treatment-refractory visceral leishmaniasis in Brazil. Mem Inst Oswaldo Cruz 113:119–125. https://doi.org/10.1590/0074-02760170289
Misra P, Kumar A, Khare P et al (2009) Pro-apoptotic effect of the landrace Bangla Mahoba of Piper betle on Leishmania donovani may be due to the high content of eugenol. J Med Microbiol 58:1058–1066. https://doi.org/10.1099/jmm.0.009290-0
Molnár J, Engi H, Hohmann J et al (2010) Reversal of multidrug resitance by natural substances from plants. Curr Top Med Chem 10:1757–1768
Montrieux E, Perera WH, García M et al (2014) In vitro and in vivo activity of major constituents from Pluchea carolinensis against Leishmania amazonensis. Parasitol Res 113:2925–2932. https://doi.org/10.1007/s00436-014-3954-1
Monzote L, Perera Córdova WH, García M et al (2016) In-vitro and in-vivo activities of phenolic compounds against cutaneous leishmaniasis. Rec Nat Prod 10:269–276
Mukherjee D, Singh CB, Dey S et al (2016) Induction of apoptosis by zerumbone isolated from Zingiber zerumbet (L.) Smith in protozoan parasite Leishmania donovani due to oxidative stress. Brazil J Infect Dis 20:48–55. https://doi.org/10.1016/j.bjid.2015.10.002
Ogungbe IV, Erwin WR, Setzer WN (2014) Antileishmanial phytochemical phenolics: molecular docking to potential protein targets. J Mol Graph Model 48:105–117. https://doi.org/10.1016/j.jmgm.2013.12.010
Onal G, Kutlu O, Gozuacik D, Emre SD (2017) Lipid droplets in health and disease. Lipids Health Dis. https://doi.org/10.1186/s12944-017-0521-7
Owczarczyk-Saczonek A, Lahuta LB, Ligor M et al (2018) The healing-promoting properties of selected cyclitols—a review. Nutrients 10:1–20. https://doi.org/10.3390/nu10121891
Park YS, Jung ST, Kang SG et al (2008) Antioxidants and proteins in ethylene-treated kiwifruits. Food Chem 107:640–648. https://doi.org/10.1016/j.foodchem.2007.08.070
Pintaudi B, Di Vieste G, Bonomo M (2016) The Effectiveness of Myo-Inositol and D-Chiro inositol treatment in type 2 diabetes. Int J Endocrinol. https://doi.org/10.1155/2016/9132052
Ponte-Sucre A, Gamarro F, Dujardin JC et al (2017) Drug resistance and treatment failure in leishmaniasis: a 21st century challenge. PLoS Negl Trop Dis 11:1–24. https://doi.org/10.1371/journal.pntd.0006052
Pramanik PK, Paik D, Pramanik A, Chakraborti T (2019) White jute (Corchorus capsularis L.) leaf extract has potent leishmanicidal activity against Leishmania donovani. Parasitol Int 71:41–45. https://doi.org/10.1016/j.parint.2019.03.012
Purkait B, Kumar A, Nandi N et al (2012) Mechanism of amphotericin B resistance in clinical isolates of Leishmania donovani. Antimicrob Agents Chemother 56:1031–1041. https://doi.org/10.1128/AAC.00030-11
Reanmongkol W, Noppapan T, Subhadhirasakul S (2009) Antinociceptive, antipyretic, and anti-inflammatory activities of Putranjiva roxburghii Wall. leaf extract in experimental animals. J Nat Med 63:290–296. https://doi.org/10.1007/s11418-009-0336-6
Redza-Dutordoir M, Averill-Bates DA (2016) Activation of apoptosis signalling pathways by reactive oxygen species. Biochim Biophys Acta Mol Cell Res 1863:2977–2992. https://doi.org/10.1016/j.bbamcr.2016.09.012
Rezvan H, Moafi M (2015) An overview on Leishmania vaccines: a narrative review article. Vet Res forum an Int Q J 6:1–7
Rohloff J, Hymete A, Tariku Y (2013) Plant-derived natural products for the treatment of leishmaniasis, 1st edn. Copyright © 2013 Elsevier B.V. All rights reserved.
Roy S, Dutta D, Satyavarapu EM et al (2017) Mahanine exerts in vitro and in vivo antileishmanial activity by modulation of redox homeostasis. Sci Rep 7:1–16. https://doi.org/10.1038/s41598-017-03943-y
Sales MDC, Costa HB, Fernandes PMB et al (2016) Antifungal activity of plant extracts with potential to control plant pathogens in pineapple. Asian Pac J Trop Biomed 6:26–31. https://doi.org/10.1016/j.apjtb.2015.09.026
Sen N, Banerjee B, Das BB et al (2007) Apoptosis is induced in leishmanial cells by a novel protein kinase inhibitor withaferin A and is facilitated by apoptotic topoisomerase I-DNA complex. Cell Death Differ 14:358–367. https://doi.org/10.1038/sj.cdd.4402002
Sharma U, Velpandian T, Sharma P, Singh S (2009) Evaluation of anti-leishmanial activity of selected Indian plants known to have antimicrobial properties. Parasitol Res 105:1287–1293. https://doi.org/10.1007/s00436-009-1554-2
Strober W (2001) Trypan blue exclusion test of cell viability. Curr Protoc Immunol Appendix 3:2–3. https://doi.org/10.1002/0471142735.ima03bs21
Sundar S, Singh A, Singh OP (2014) Strategies to overcome antileishmanial drugs unresponsiveness. J Trop Med. https://doi.org/10.1155/2014/646932
Tan H, Wang N, Li S et al (2016) The reactive oxygen species in macrophage polarization: human diseases. Oxid Med Cell Longev 2016:1–16. https://doi.org/10.1155/2016/2795090
Tomiotto-Pellissier F, Alves DR, Miranda-Sapla MM et al (2018) Caryocar coriaceum extracts exert leishmanicidal effect acting in promastigote forms by apoptosis-like mechanism and intracellular amastigotes by Nrf2/HO-1/ferritin dependent response and iron depletion: leishmanicidal effect of Caryocar coriaceum leaf ex. Biomed Pharmacother 98:662–672. https://doi.org/10.1016/j.biopha.2017.12.083
Varma A, Alok S, Jain SK (2011) Hypoglycemic activity of Putranjiva roxburghii Wall. in alloxan induced diabetic rats. Int J Pharm Sci Res 2:160–164. https://doi.org/10.13040/IJPSR.0975-8232.2(1).160-64
Zhou SH, Fang ZX, Lü Y et al (2009) Phenolics and antioxidant properties of bayberry (Myrica rubra Sieb. et Zucc.) pomace. Food Chem 112:394–399. https://doi.org/10.1016/j.foodchem.2008.05.104
Acknowledgements
The authors are thankful to the Department of Botany, Panjab University Chandigarh for identifying the plant material. Sophisticated Analytical Instrument Facilities (SAIF)/Central Instrumentation Laboratory (CIL), Panjab University, Chandigarh is acknowledged for GC-MS analysis.
Funding
This work was financially supported by University Grants Commission, New Delhi under UGC-JRF/SRF Fellowship (3643/NET-DEC.2012) and UGC-CAS (F.4–28/2015/CAS-II) (SAP-II).
Author information
Authors and Affiliations
Contributions
PK and DKG carried out the experimental work and wrote the manuscript. PK, DKG, and SK designed the study. SK supervised the experimental study and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors hereby declare that they have no conflicts of interest.
Ethics approval
Ethics committee approval was received for this study from the scientific committee of the Faculty of Science, Panjab University, Chandigarh.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Keshav, P., Goyal, D.K. & Kaur, S. GC–MS screening and antiparasitic action of Putranjiva roxburghii leaves against sensitive and resistant strains of Leishmania donovani. J Parasit Dis 45, 1002–1013 (2021). https://doi.org/10.1007/s12639-021-01388-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-021-01388-9