Abstract
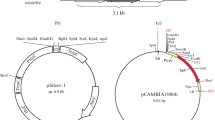
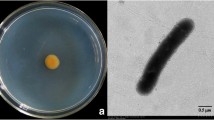
Streptomyces lydicus A01 and Trichoderma harzianum P1 are potential biocontrol agents of fungal diseases in plants. S. lydicus A01 produces natamycin to bind the ergosterol of the fungal cell membrane and inhibits the growth of Botrytis cinerea. T. harzianum P1, on the other hand, features high chitinase activity and decomposes the chitin in the cell wall of B. cinerea. To obtain the synergistic biocontrol effects of chitinase and natamycin on Botrytis cinerea, this study transformed the chit42 gene from T. harzianum P1 to S. lydicus A01. The conjugal transformant (CT) of S. lydicus A01 with the chit42 gene was detected using polymerase chain reaction (PCR). Associated chitinase activity and natamycin production were examined using the 3, 5-dinitrosalicylic acid (DNS) method and ultraviolet spectrophotometry, respectively. The S. lydicus A01-chit42 CT showed substantially higher chitinase activity and natamycin production than its wild type strain (WT). Consequently, the biocontrol effects of S. lydicus A01-chit42 CT on B. cinerea, including inhibition to spore germination and mycelial growth, were highly improved compared with those of the WT. Our research indicates that the biocontrol effect of Streptomyces can be highly improved by transforming the exogenous resistance gene, i.e. chit42 from Trichoderma, which not only enhances the production of antibiotics, but also provides a supplementary function by degrading the cell walls of the pathogens.
Similar content being viewed by others
References
Alfonso, C., Raposo, R., and Melgarejo, P. 2000. Genetic diversity in Botrytis cinerea populations on vegetable crops in greenhouses in south-eastern Spain. Plant Pathol. 49, 243–251.
Chen, Z., Wen, J., Song, Y., Wen, Y., and Li, J.L. 2007. Enhancement and selective production of avermectin B by recombinants of Streptomyces avermitilis via intraspecific protoplast fusion. Chinese Sci. Bull. 52, 616–622.
Cortes, C., Gutierrez, A., Olmedo, V., Inbar, J., Chet, I., and Herrera-Estrella, A. 1998. The expression of genes involved in parasitism by Trichoderma harzianum is triggered by a diffusible factor. Mol. Gen. Genet. 260, 218–225.
Distefano, G., Malfa, S.L., Vitale, A., Lorito, M., Deng, Z.N., and Gentile, A. 2008. Defence-related gene expression in transgenic lemon plants producing an antimicrobial Trichoderma harzianum endochitinase during fungal infection. Transgenic Res. 17, 873–879.
Elad, Y., Chet, I., Boyle, P., and Henis, Y. 1983. Parasitism of Trichoderma spp. on Rhizoctonia solani and Sclerotium rolfsii-scanning electron microscopy and fluorescence microscopy. Phytopathology 73, 85–88.
Elad, Y. and Kapat, A. 1999. The role of Trichoderma harzianum protease in the biocontrol of Botrytis cinerea. Eur. J. Plant Pathol. 105, 177–189.
Harman, G.E., Hayes, C.K., Lorito, M., Broadway, R.M., Di Pietro, A., Peterbauer, C., and Tronsmo, A. 1993. Chitinolytic enzymes of Trichoderma harzianum: Purification of chitobiosidase and endochitinase. Phytopathology 83, 313–318.
Hong, B., Wu, B.Y., and Li, Y. 2003. Production of C-terminal amidated recombinant salmon calcitonin in Streptomyces lividans. Appl. Biochem. Biotechnol. 110, 113–123.
Huang, X.Q., Chen, L.H., Ran, W., Shen, Q.R., and Yang, X.M. 2011. Trichoderma harzianum strain SQR-T37 and its bio-organic fertilizer could control Rhizoctonia solani damping-off disease in cucumber seedlings mainly by the mycoparasitism. Appl. Microbiol. Biotechnol. 91, 741–755.
Ishikawa, R., Shirouzu, K., Nakashita, H., Lee, H.Y., Motoyama, T., Yamaguchi, I., Teraoka, T., and Arie, T. 2005. Foliar spray of validamycin A or validoxylamine A controls tomato Fusarium wilt. Phytopathology 95, 1209–1216.
Kieser, T., Bibb, M.J., Buttner, M.J., Chater, K.F., and Hopwood, D.A. 2000. Practical Streptomyces genetics, pp. 249–250. The John Innes Foundation, Norwich, England.
Kim, K.J., Yang, Y.J., and Kim, J.G. 2003. Purification and characterization of chitinase from Streptomyces sp. M-20. J. Biochem. Mol. Biol. 36, 185–189.
Limón, M.C., Chacón, M.R., Mejías, R., Delgado-Jarana, J., Rincón, A.M., Codón, A.C., and Benítez, T. 2004. Increased antifungal and chitinase specific activities of Trichoderma harzianum CECT 2413 by addition of a cellulose binding domain. Appl. Microbiol. Biotechnol. 64, 675–685.
Liu, Y.S., Shibu, M.A., Jhan, H.J., Lo, C.T., and Peng, K.C. 2010. Purification and characterization of novel glucanases from Trichoderma harzianum ETS 323. J. Agr. Food Chem. 58, 10309–10314.
Lorito, M., Hayes, C.K., Di Pietro, A., Woo, S.L., and Harman, G.E. 1994. Purification, characterization and synergistic activity of a glucan 1, 3-β-glucosidase and an N-acetyl-β-glucosaminidase from Trichoderma harzianum. Phytopathology 84, 398–405.
Lorito, M., Peterbauer, C., Sposato, P., Mach, R.L., Strauss, J., and Kubicek, C.P. 1996. Mycoparasitic interaction relieves binding of the Cre1 carbon catabolite repressor protein to promoter sequences of the ech42 (endochitinase-encoding) gene in Trichoderma harzianum. Proc. Natl. Acad. Sci. USA 93, 14868–14872.
Lu, C.G., Liu, W.C., Qiu, J.Y., Wang, H.M., Liu, T., and Liu, D.W. 2008. Identification of an antifungal metabolite produced by a potential biocontrol Actinomyces strain A01. Braz. J. Microbiol. 39, 701–707.
Mach, R.L., Peterbauer, C.K., Payer, K., Jaksits, S., Woo, S.L., Zeilinger, S., Kullnig, C.M., Lorito, M., and Kubicek, C.P. 1999. Expression of two major chitinase genes of Trichoderma atroviride (T. harzianum P1) is triggered by different regulatory signals. Appl. Environ. Microbiol. 65, 1858–1863.
Monte, E. 2010. Understanding Trichoderma: Between biotechnology and microbial ecology. Int. Microbiol. 4, 1–4.
Nampoothiri, K.M., Baijua, T.V., Sandhya, C., Sabu, A., Szakacs, G., and Pandey, A. 2004. Process optimization for antifungal chitinase production by Trichoderma harzianum. Process Biochem. 39, 1583–1590.
Nawani, N.N. and Kapadnis, B.P. 2005. Optimization of chitinase production using statistics based experimental designs. Process Biochem. 40, 651–660.
Pintado, C.M.B.S., Ferreira, M.A.S.S., and Sousa, I. 2010. Control of pathogenic and spoilage microorganisms from cheese surface by whey protein films containing malic acid, nisin and natamycin. Food Control. 21, 240–246.
Reissig, J.L., Storminger, J.L., and Leloir, L.F. 1955. A modified colorimetric method for the estimation of N-acetylamino sugars. J. Biol. Chem. 217, 959–966.
Steyaert, J.M. 2004. Co-expression of two genes, a chitinase (chit42) and proteinase (prb1), implicated in mycoparasitism by Trichoderma hamatum. Mycologia 96, 1245–1252.
te Welscher, Y.M., ten Napel, H.H., Balagué, M.M., Souza, C.M., Riezman, H., de Kruijff, B., and Breukink, E. 2008. Natamycin blocks fungal growth by binding specifically to ergosterol without permeabilizing the membrane. J. Biol. Chem. 283, 6393–6401.
te Welscher, Y.M., Jones, L., van M.R., Dijksterhuis, J., de Kruijff, B., Eitzen, G., and Breukink, E. 2010. Natamycin inhibits vacuole fusion at the priming phase via a specific interaction with ergosterol. Antimicrob. Agents Chemother. 54, 2618–2625.
Viterbo, A., Haran, S., Friesem, D., Ramot, O., and Chet, I. 2001. Antifungal activity of a novel endochitinase gene (chit36) from Trichoderma harzianum Rifai TM. FEMS Microbiol. Lett. 200, 169–174.
Wang, X.J., Wang, X.C., and Xiang, W.S. 2009. Improvement of milbemycin-producing Streptomyces bingchenggensis by rational screening of ultraviolet-and chemically induced mutants. World J. Microbiol. Biotechnol. 25, 1051–1056.
Woo, S.L., Donzelli, B., Scala, F., Mach, R., Harman, G.E., Kubicek, C.P., Del Sorbo, G., and Lorito, M. 1999. Disruption of the ech42 (endochitinase-encoding) gene affects biocontrol activity in Trichoderma harzianum P1. MPMI. 12, 419–429.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, Q., Bai, L., Liu, W. et al. Construction of a Streptomyces lydicus A01 transformant with a chit42 gene from Trichoderma harzianum P1 and evaluation of its biocontrol activity against Botrytis cinerea . J Microbiol. 51, 166–173 (2013). https://doi.org/10.1007/s12275-013-2321-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-013-2321-8