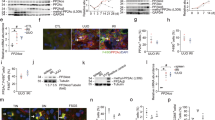
Abstract
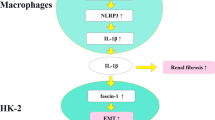
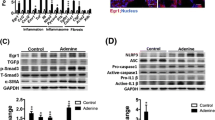
Renal fibrosis is defined by excessive extracellular matrix (ECM) accumulation and is associated with a decreased kidney function. Increased inflammation and infiltration of inflammatory cells are the key features of renal fibrosis development; however, the mechanism of how inflammation starts is still un-known. Here, we show that the activation of epithelial Protease-activating receptor 2 (PAR2) signaling plays an important role in the initiation of inflammation via increased chemokine expression and inflammatory cell induction. In the adenine diet-induced renal fibrosis mouse model, PAR2 expression was significantly increased in the renal tubule region. Kidneys from PAR2-knockout mice were protected from adenine diet-induced renal fibrosis, kidney dysfunction, and inflammation. Using NRK52E kidney epithelial cells, we further elucidated the mechanisms underlying these processes. Activation of PAR2 signaling pathway by PAR2 agonist specifically increased the levels of chemokines, including MCP1 and MCP3, via the MAPK-NF-κB signaling pathway. Inhibition of the MAPK signaling pathway attenuated PAR2 agonist-induced NF-κB activation, chemokine expression, and macrophage cell induction. Furthermore, PAR2 activation directly increased mesenchymal cell markers in epithelial cells. Taken together, we found that increased PAR2 expression and the PAR2/MAPK signaling pathway promote renal fibrosis by increasing the inflammatory responses and promoting EMT process.






Similar content being viewed by others
References
Allison SJ (2015) Fibrosis: Targeting EMT to reverse renal fibrosis. Nat Rev Nephrol 11:565. https://doi.org/10.1038/nrneph.2015.133
Buchtler S, Grill A, Hofmarksrichter S, Stockert P, Schiechl-Brachner G, Rodriguez Gomez M, Neumayer S, Schmidbauer K, Talke Y, Klinkhammer BM, Boor P, Medvinsky A, Renner K, Castrop H, Mack M (2018) Cellular origin and functional relevance of collagen I production in the kidney. J Am Soc Nephrol 29:1859–1873. https://doi.org/10.1681/ASN.2018020138
Chung H, Ramachandran R, Hollenberg MD, Muruve DA (2013) Proteinase-activated receptor-2 transactivation of epidermal growth factor receptor and transforming growth factor-beta receptor signaling pathways contributes to renal fibrosis. J Biol Chem 288:37319–37331. https://doi.org/10.1074/jbc.M113.492793
Denic A, Glassock RJ, Rule AD (2016) Structural and functional changes with the aging kidney. Adv Chronic Kidney Dis 23:19–28. https://doi.org/10.1053/j.ackd.2015.08.004
Derynck R, Zhang YE (2003) Smad-dependent and Smad-independent pathways in TGF-beta family signalling. Nature 425:577–584. https://doi.org/10.1038/nature02006
Du CY, Zhang T, Xiao X, Shi YH, Duan HJ, Ren YZ (2017) Protease-activated receptor-2 promotes kidney tubular epithelial inflammation by inhibiting autophagy via the PI3K/Akt/mTOR signalling pathway. Biochem J 474:2733–2747. https://doi.org/10.1042/Bcj20170272
Duffield JS (2014) Cellular and molecular mechanisms in kidney fibrosis. J Clin Invest 124:2299–2306. https://doi.org/10.1172/JCI72267
Farris AB, Colvin RB (2012) Renal interstitial fibrosis: mechanisms and evaluation. Curr Opin Nephrol Hypertens 21:289–300. https://doi.org/10.1097/MNH.0b013e3283521cfa
Frigo DE, Tang Y, Beckman BS, Scandurro AB, Alam J, Burow ME, Mclachlan JA (2004) Mechanism of AP-1-mediated gene expression by select organochlorines through the p38 MAPK pathway. Carcinogenesis 25:249–261. https://doi.org/10.1093/carcin/bgh009
Gonzalez J, Mouttalib S, Delage C, Calise D, Maoret JJ, Pradere JP, Klein J, Buffin-Meyer B, Van Der Veen B, Charo IF, Heeringa P, Duchene J, Bascands JL, Schanstra JP (2013) Dual effect of chemokine CCL7/MCP-3 in the development of renal tubulointerstitial fibrosis. Biochem Biophys Res Commun 438:257–263. https://doi.org/10.1016/j.bbrc.2013.07.025
Grandaliano G, Pontrelli P, Cerullo G, Monno R, Ranieri E, Ursi M, Loverre A, Gesualdo L, Schena FP (2003) Protease-activated receptor-2 expression in IgA nephropathy: a potential role in the pathogenesis of interstitial fibrosis. J Am Soc Nephrol 14:2072–2083. https://doi.org/10.1097/01.asn.0000080315.37254.a1
Hayashi S, Oe Y, Fushima T, Sato E, Sato H, Ito S, Takahashi N (2017) Protease-activated receptor 2 exacerbates adenine-induced renal tubulointerstitial injury in mice. Biochem Biophys Res Commun 483:547–552. https://doi.org/10.1016/j.bbrc.2016.12.108
Heuberger DM, Schuepbach RA (2019) Protease-activated receptors (PARs): mechanisms of action and potential therapeutic modulators in PAR-driven inflammatory diseases. Thromb J 17:4. https://doi.org/10.1186/s12959-019-0194-8
Hilliard LM, Sampson AK, Brown RD, Denton KM (2013) The “his and hers” of the renin-angiotensin system. Curr Hypertens Rep 15:71–79. https://doi.org/10.1007/s11906-012-0319-y
Kashyap S, Osman M, Ferguson CM, Nath MC, Roy B, Lien KR, Nath KA, Garovic VD, Lerman LO, Grande JP (2018) Ccl2 deficiency protects against chronic renal injury in murine renovascular hypertension. Sci Rep 8:8598. https://doi.org/10.1038/s41598-018-26870-y
Kim DH, Lee B, Lee J, Kim ME, Lee JS, Chung JH, Yu BP, Dong HH, Chung HY (2019) FoxO6-mediated IL-1beta induces hepatic insulin resistance and age-related inflammation via the TF/PAR2 pathway in aging and diabetic mice. Redox Biol 24:101184. https://doi.org/10.1016/j.redox.2019.101184
Lemos CC, Mandarim-De-Lacerda CA, Dorigo D, Coimbra TM, Bregman R (2005) Chronic renal failure in male and female rats. J Nephrol 18:368–373
Levin A, Tonelli M, Bonventre J, Coresh J, Donner JA, Fogo AB, Fox CS, Gansevoort RT, Heerspink HJL, Jardine M, Kasiske B, Kottgen A, Kretzler M, Levey AS, Luyckx VA, Mehta R, Moe O, Obrador G, Pannu N, Parikh CR, Perkovic V, Pollock C, Stenvinkel P, Tuttle KR, Wheeler DC, Eckardt KU, Participants ISNGKHS (2017) Global kidney health 2017 and beyond: a roadmap for closing gaps in care, research, and policy. Lancet 390:1888–1917. https://doi.org/10.1016/S0140-6736(17)30788-2
Lv W, Booz GW, Wang Y, Fan F, Roman RJ (2018) Inflammation and renal fibrosis: Recent developments on key signaling molecules as potential therapeutic targets. Eur J Pharmacol 820:65–76. https://doi.org/10.1016/j.ejphar.2017.12.016
Mansour SG, Puthumana J, Coca SG, Gentry M, Parikh CR (2017) Biomarkers for the detection of renal fibrosis and prediction of renal outcomes: a systematic review. BMC Nephrol 18:72. https://doi.org/10.1186/s12882-017-0490-0
Marnett LJ, Rowlinson SW, Goodwin DC, Kalgutkar AS, Lanzo CA (1999) Arachidonic acid oxygenation by COX-1 and COX-2. Mechanisms of catalysis and inhibition. J Biol Chem 274:22903–22906. https://doi.org/10.1074/jbc.274.33.22903
Meng XM, Nikolic-Paterson DJ, Lan HY (2014) Inflammatory processes in renal fibrosis. Nat Rev Nephrol 10:493–503. https://doi.org/10.1038/nrneph.2014.114
Meng XM, Tang PM, Li J, Lan HY (2015) TGF-beta/Smad signaling in renal fibrosis. Front Physiol 6:82. https://doi.org/10.3389/fphys.2015.00082
Morgan MJ, Liu ZG (2011) Crosstalk of reactive oxygen species and NF-kappaB signaling. Cell Res 21:103–115. https://doi.org/10.1038/cr.2010.178
Oe Y, Hayashi S, Fushima T, Sato E, Kisu K, Sato H, Ito S, Takahashi N (2016) Coagulation factor Xa and protease-activated Receptor 2 as novel therapeutic targets for diabetic nephropathy. Arterioscler Thromb Vasc Biol 36:1525–1533. https://doi.org/10.1161/ATVBAHA.116.307883
Pan MH, Lai CS, Dushenkov S, Ho CT (2009) Modulation of inflammatory genes by natural dietary bioactive compounds. J Agric Food Chem 57:4467–4477. https://doi.org/10.1021/jf900612n
Peng X, Zhang J, Xiao Z, Dong Y, Du J (2015) CX3CL1-CX3CR1 interaction increases the population of Ly6C(-)CX3CR1(hi) macrophages contributing to unilateral ureteral obstruction-induced fibrosis. J Immunol 195:2797–2805. https://doi.org/10.4049/jimmunol.1403209
Pichler R, Afkarian M, Dieter BP, Tuttle KR (2017) Immunity and inflammation in diabetic kidney disease: translating mechanisms to biomarkers and treatment targets. Am J Physiol Renal Physiol 312:F716–F731. https://doi.org/10.1152/ajprenal.00314.2016
Qi R, Yang C (2018) Renal tubular epithelial cells: the neglected mediator of tubulointerstitial fibrosis after injury. Cell Death Dis 9:1126. https://doi.org/10.1038/s41419-018-1157-x
Ramachandran R, Hollenberg MD (2008) Proteinases and signalling: pathophysiological and therapeutic implications via PARs and more. Br J Pharmacol 153(Suppl 1):S263–S282. https://doi.org/10.1038/sj.bjp.0707507
Richter K, Konzack A, Pihlajaniemi T, Heljasvaara R, Kietzmann T (2015) Redox-fibrosis: Impact of TGFbeta1 on ROS generators, mediators and functional consequences. Redox Biol 6:344–352. https://doi.org/10.1016/j.redox.2015.08.015
Romagnani P, Remuzzi G, Glassock R, Levin A, Jager KJ, Tonelli M, Massy Z, Wanner C, Anders HJ (2017) Chronic kidney disease. Nat Rev Dis Primers 3:17088. https://doi.org/10.1038/nrdp.2017.88
Rothmeier AS, Ruf W (2012) Protease-activated receptor 2 signaling in inflammation. Semin Immunopathol 34:133–149. https://doi.org/10.1007/s00281-011-0289-1
Sallusto F, Baggiolini M (2008) Chemokines and leukocyte traffic. Nat Immunol 9:949–952. https://doi.org/10.1038/ni.f.214
Sanz AB, Sanchez-Nino MD, Ramos AM, Moreno JA, Santamaria B, Ruiz-Ortega M, Egido J, Ortiz A (2010) NF-kappaB in renal inflammation. J Am Soc Nephrol 21:1254–1262. https://doi.org/10.1681/ASN.2010020218
Shuiai Z, Huijun S, Weizhong G, Aimin L, Jianhua M (2017) Evaluation of TGF-beta1 and MCP-1 expression and tubulointerstitial fibrosis in children with Henoch-Schonlein purpura nephritis and IgA nephropathy: A clinical correlation. Clinics (sao Paulo) 72:95–102. https://doi.org/10.6061/clinics/2017(02)05
Sureshbabu A, Muhsin SA, Choi ME (2016) TGF-beta signaling in the kidney: profibrotic and protective effects. Am J Physiol Renal Physiol 310:F596–F606. https://doi.org/10.1152/ajprenal.00365.2015
Tesch GH (2008) MCP-1/CCL2: a new diagnostic marker and therapeutic target for progressive renal injury in diabetic nephropathy. Am J Physiol Renal Physiol 294:F697-701. https://doi.org/10.1152/ajprenal.00016.2008
Tsou CL, Peters W, Si Y, Slaymaker S, Aslanian AM, Weisberg SP, Mack M, Charo IF (2007) Critical roles for CCR2 and MCP-3 in monocyte mobilization from bone marrow and recruitment to inflammatory sites. J Clin Invest 117:902–909. https://doi.org/10.1172/JCI29919
Vesey DA, Suen JY, Seow V, Lohman RJ, Liu L, Gobe GC, Johnson DW, Fairlie DP (2013) PAR2-induced inflammatory responses in human kidney tubular epithelial cells. Am J Physiol Renal Physiol 304:F737–F750. https://doi.org/10.1152/ajprenal.00540.2012
Watanabe M, Oe Y, Sato E, Sekimoto A, Sato H, Ito S, Takahashi N (2019) Protease-activated receptor 2 exacerbates cisplatin-induced nephrotoxicity. Am J Physiol Renal Physiol 316:F654–F659. https://doi.org/10.1152/ajprenal.00489.2018
Xiong J, Zhu Z, Liu J, Wang Y, Li Z (2005) Role of protease activated receptor-2 expression in renal interstitial fibrosis model in mice. J Huazhong Univ Sci Technolog Med Sci 25:523–526. https://doi.org/10.1007/BF02896006
Yan S, Ding H, Peng J, Wang X, Pang C, Wei J, Wei J, Chen H (2020) Down-regulation of protease-activated receptor 2 ameliorated osteoarthritis in rats through regulation of MAPK/NF-kappaB signaling pathway in vivo and in vitro. Biosci Rep. https://doi.org/10.1042/BSR20192620
Yang L, Qu M, Wang Y, Duan H, Chen P, Wang Y, Shi W, Danielson P, Zhou Q (2013) Trichostatin A inhibits transforming growth factor-beta-induced reactive oxygen species accumulation and myofibroblast differentiation via enhanced NF-E2-related factor 2-antioxidant response element signaling. Mol Pharmacol 83:671–680. https://doi.org/10.1124/mol.112.081059
Yu M, Ryu DR, Kim SJ, Choi KB, Kang DH (2010) Clinical implication of metabolic syndrome on chronic kidney disease depends on gender and menopausal status: results from the Korean National Health and Nutrition Examination Survey. Nephrol Dial Transplant 25:469–477. https://doi.org/10.1093/ndt/gfp483
Zeeh F, Witte D, Gadeken T, Rauch BH, Grage-Griebenow E, Leinung N, Fromm SJ, Stolting S, Mihara K, Kaufmann R, Settmacher U, Lehnert H, Hollenberg MD, Ungefroren H (2016) Proteinase-activated receptor 2 promotes TGF-beta-dependent cell motility in pancreatic cancer cells by sustaining expression of the TGF-beta type I receptor ALK5. Oncotarget 7:41095–41109. https://doi.org/10.18632/oncotarget.9600
Zlotnik A, Yoshie O (2000) Chemokines: a new classification system and their role in immunity. Immunity 12:121–127. https://doi.org/10.1016/s1074-7613(00)80165-x
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (NRF-2018R1A2A3075425, NRF-2020R1C1C1004839, and NRF-2021R1I1A1A0105215311). This study was financially supported by the 2021 Post-Doc. Development Program of Pusan National University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ha, S., Chung, K.W., Lee, J. et al. Renal tubular PAR2 promotes interstitial fibrosis by increasing inflammatory responses and EMT process. Arch. Pharm. Res. 45, 159–173 (2022). https://doi.org/10.1007/s12272-022-01375-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-022-01375-5