Abstract
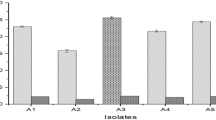
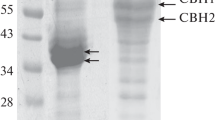
A tannase (E.C. 3.1.1.20) producing fungal strain was isolated from soil and identified as Aspergillus heteromorphus MTCC 8818. Maximum tannase production was achieved on Czapek Dox minimal medium containing 1% tannic acid at a pH of 4.5 and 30°C after 48 h incubation. The crude enzyme was purified by ammonium sulfate precipitation and ion exchange chromatography. Diethylaminoethyl-cellulose column chromatography led to an overall purification of 39.74-fold with a yield of 19.29%. Optimum temperature and pH for tannase activity were 50°C and 5.5 respectively. Metal ions such as Ca2+, Fe2+, Cu1+, and Cu2+ increased tannase activity, whereas Hg2+, Na1+, K1+, Zn2+, Ag1+, Mg2+, and Cd2+ acted as enzyme inhibitors. Various organic solvents such as isopropanol, isoamyl alcohol, benzene, methanol, ethanol, toluene, and glycerol also inhibited enzyme activity. Among the surfactants and chelators studied, Tween 20, Tween 80, Triton X-100, EDTA, and 1, 10-o-phenanthrolein inhibited tannase activity, whereas sodium lauryl sulfate enhanced tannase activity at 1% (w/v).
Similar content being viewed by others
References
Zhong, X., L. Peng, S. Zheng, Z. Sun, Y. Ren, M. Dong, and A. Xu (2004) Secretion, purification and characterization of a recombinant Aspergillus oryzae tannase in Pichia pastoris. Protein Expr. Purif. 36: 165–169.
Lekha, P. K. and B. K. Lonsane (1997) Production and application of tannin acyl hydralose: State of the art. Adv. Appl. Microbiol. 44: 215–260.
Chhokar, V., M. Sangwan, V. Beniwal, K. Nehra, and K. S. Nehra (2009) Effect of additives on the activity of tannase from Aspergillus awamori MTCC9299. Appl. Biochem. Biotechnol. 160: 2256–2264.
Mukherjee, G. and R. Banerjee (2005) Effects of temperature, pH and additives on the activity of tannase produced by a co-culture of Rhizopus oryzae and Aspergillus foetidus. World J. Microbiol. Biotech. 22: 207–211.
Garca-Conesa, M. T., P. Ostergaard, S. Kauppinen, and G. Williamson (2001) Hydrolysis of diethyl diferulates by a tannase from Aspergillus oryzae. Carbohydrate Polymers 44: 319–324.
Madhavakrishna, W., S. M. Bose, and Y. Nayudamma (1960) Estimation of tannase and certain oxidizing enzymes in Indian vegetables and stuffs. The Bull. Central Leather Res. Institute 7: 1–11.
Nichaus, J. U. and G. G. Gross (1997) A gallatannin degrading esterase from the leaves of Penduculate Oak. Phytochem. 45: 1555–1560.
Bhat, T. K., B. Singh, and O. P. Sharma (1998) Microbial degradation of tannins: A current perspective. Biodegradation. 9: 343–357.
Pandey, A., C. R. Soccol, and D. Mitchell (2000) New developments in solid state fermentation:1. Bioprocesses and products. Proc. Biochem. 35: 1153–1169.
Bradoo, S., R. Gupta, and R. K. Saxena (1996) Screening for extracellular tannase producing fungi: Development of a rapid and simple plate assay. J. Gen. Appl. Microb. 42: 325–329.
Mondal, K. C., D. Banerjee, M. Jana, and B. R. Pati (2001) Colorimetric assay method for determination of the tannase activity. Anal. Biochem. 295: 168–171.
Bradford, M. M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Kumar, R., J. Sharma, and R. Singh (2007) Production of tannase from Aspergillus ruber under solid-state fermentation using jamun (Syzygium cumini) leaves. Microbiol. Res. 162: 384–390.
Kar, B. and R. Banerjee (2000) Biosynthesis of tannin acyl hydrolase from tannin-rich forest residue under different fermentation conditions. J. Ind. Microbiol. Biotechnol. 25: 29–38.
Battestin, V. and G. A. Macedo (2007) Tannase production by Paecilomyces variotii. Biores. Technol. 98: 1832–1837.
Bradoo, S., R. Gupta, and R. K. Saxena (1997) Parametric optimization and biochemical regulation of extracellular tannase from Aspergillus japonicus. Proc. Biochem. 32: 135–139.
Marco, M. -G., L. V. RodrÍguez, E. L. Ramos, J. Renovato, M. A. Cruz-Hernández., R. Rodríguez, J. Contreras, and C. N. Aguilar (2009) A novel tannase from the xerophilic fungus Aspergillus niger GH1. J. Microbiol. Biotechnol. 19: 987–996.
Pinto, G. A. S., G. F. S. Leite, C. S. Terzi, and S. Couri (2001) Selection of tannase producing Aspergillus Niger strains. Braz. J. Microbiol. 32: 24–26.
Mondal, K. C., D. Banerjee, R. Banerjee, and B. R. Pati (2001) Production and characterization of tannase from Bacillus cereus KBR 9. J. Gen. Appl. Microbiol. 47: 263–267.
Sharma, S., L. Agarwal, and R. K. Saxena (2008) Purification, immobilization and characterization of tannase from Penicillium variable. Biores. Technol. 99: 2544–2551.
Mahendran, B., N. Raman, and D. J. Kim (2006) Purification and characterization of tannase from Paecilomyces variotii: Hydrolysis of tannic acid using immobilized tannase. Appl. Microbiol. Biotechnol. 70: 444–450.
Sabu, A., S. G. Kiran, and A. Pandey (2005) Purification and characterization of tannin acyl hydrolose from Aspergillus niger ATCC 16620. J. Food Technol. Biotechnol. 2: 133–138.
Sharma, S., T. K. Bhat, and R. K. Dawra (1999) Isolation, purification and properties of tannase from A. niger van Tieghem. World J. Microbiol. Biotechnol. 15: 673–677.
Ramirez-Coronel, M. A., G. Viniegra-Gonzalez, A. Darvill, and C. Augur (2003) A novel tannase from Aspergillus niger with α-glucosidase activity. Microbiol. 149: 2941–2946.
Saborowski, R., G. Sahling, M. A. Navarette del Toro, I. Walter, and F. L. Garcya-Carreno (2004) Stability and effects of organic solvents on endopeptidases from thegastric fluid of the marine crab Cancer pagurus. J. Mol. Catal. B: Enzymatic 30: 109–118.
Nakamura, T., H. Iwahashi, and Y. Eguchi (1984) Enzymatic proof for the identity of the S-sulfocysteine synthase and cysteine synthase B of Salmonella typhimurium. J. Bacteriol. 158: 1122–1127.
Kar, B., R. Banerjee, and B. C. Bhattacharyya (2003) Effect of additives on the behavioural properties of tannin acyl hydrolase. Proc. Biochem. 38: 1285–1293.
Kasieczka-Burnecka, M., K. Karina, H. Kalinowska, M. Knap, and M. Turkiewicz (2007) Purification and characterization of two cold-adapted extracellular tannin acyl hydrolases from an Antarctic strain Verticillium sp. P9. Appl. Microbiol. Biotechnol. 77: 77–89.
Peek, K., R. M. Daniel, C. Monk, L. Parker, and T. Coolbear (1992) Purification and characterization of a thermostable proteinase isolated from Thermus sp strain Rt 41A. Eur. J. Biochem. 207: 1035–1044.
Belmares, R., J. C. C. Esquivel, R. R. Herrera, A. R. Coronel, and C. N. Aguilar (2004) Microbial production of tannase: An enzyme with potential use in food industry. Lebensm. Wiss. Technol. 37: 857–864.
Barthomeuf, C., F. Regerat, and H. Pourrat (1994) Production, purification and characterization of a tannase from Aspergillus niger LCF 8. J. Ferment. Bioeng. 77: 320–323.
Libuchi, S., Y. Minoda, and K. Yamada (1968) Studies on tannin acyl hydrolase of microorganisms Part III. Purification of the enzyme and some properties of it. Agric. Biol. Chem. 32: 803–809.
Yamada, H., O. Adachi, M. Watanabe, and N. Sato (1968) Studies on fungal tannase Part I. Formation, purification and catalytic properties of tannase of Aspergillus flavus. Agric. Biol. Chem. 32: 1070–1078.
Aoki, K., R. Shinke, and H. Nishira (1976) Purification and some properties of yeast tannase. Agric. Biol. Chem. 40: 79–85.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chhokar, V., Seema, Beniwal, V. et al. Purification and characterization of extracellular tannin acyl hydrolase from Aspergillus heteromorphus MTCC 8818. Biotechnol Bioproc E 15, 793–799 (2010). https://doi.org/10.1007/s12257-010-0058-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-010-0058-3