Abstract
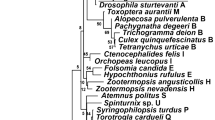
Phage WO is a bacteriophage found in Wolbachia. Herein, we represent the first phylogenetic study of WOs that infect spiders (Araneae). Seven species of spiders (Araneus alternidens, Nephila clavata, Hylyphantes graminicola, Prosoponoides sinensis, Pholcus crypticolens, Coleosoma octomaculatum, and Nurscia albofasciata) from six families were infected by Wolbachia and WO, followed by comprehensive sequence analysis. Interestingly, WO could be only detected Wolbachia-infected spiders. The relative infection rates of those seven species of spiders were 75, 100, 88.9, 100, 62.5, 72.7, and 100 %, respectively. Our results indicated that both Wolbachia and WO were found in three different body parts of N. clavata, and WO could be passed to the next generation of H. graminicola by vertical transmission. There were three different sequences for WO infected in A. alternidens and two different WO sequences from C. octomaculatum. Only one sequence of WO was found for the other five species of spiders. The discovered sequence of WO ranged from 239 to 311 bp. Phylogenetic tree was generated using maximum likelihood (ML) based on the orf7 gene sequences. According to the phylogenetic tree, WOs in N. clavata and H. graminicola were clustered in the same group. WOs from A. alternidens (WAlt1) and C. octomaculatum (WOct2) were closely related to another clade, whereas WO in P. sinensis was classified as a sole cluster.

Similar content being viewed by others
References
Ahantarig A, Trinachartvanit W, Chauvatcharin N, Kittayapong P, Baimai V (2008a) Wolbachia and bacteriophage WO-B density of Wolbachia A-infected Aedes albopictus mosquito. Folia Microbiol 53(6):547–550
Ahantarig A, Khumlthong R, Kittayapong P, Baimai V (2008b) Relative densities of bacteriophage WO and Wolbachia bacteria of Aedes albopictus mosquito during development. Ann Microbiol 58:189–193
Ahantarig A, Chauvatcharin N, Ruang-areerate T, Baimai V, Kittayapong P (2011) Infection incidence and relative density of the bacteriophage WO-B in Aedes albopictus mosquitoes from fields in Thailand. Curr Microbiol 62:816–820
Augustinos AA, Santos-Garcia D, Dionyssopoulou E, Moreira M, Papapanagiotou A, Scarvelakis M, Doudoumis V, Ramos S, Aguiar AF, Borges PAV, Khadem M, Latorre A, Tsiamis G, Bourtzis K (2011) Detection and characterization of Wolbachia infections in natural populations of aphids: is the hidden diversity fully unraveled? PLoS ONE 6(12), e28695
Bordenstein SR, Wernegreen JJ (2004) Bacteriophage flux in endosymbionts (Wolbachia): frequency, lateral transfer, and recombination rates. Mol Biol Evol 21:1981–1991
Braig HR, Zhou W, Dobson SL, O’Neill SL (1998) Cloning and characterization of a gene encoding the major surface protein of the bacterial endosymbiont Wolbachia pipientis. J Bacteriol 180:2773–2778
Breeuwer JA, Werren JH (1990) Microorganisms associated with chromosome destruction and reproductive isolation between two insect species. Nature 346:558–560
Chauvatcharin N, Ahantarig A, Baimai V, Kittayapong P (2006) Bacteriophage WO-B and Wolbachia in natural mosquito hosts: infection incidence, transmission mode and relative density. Mol Ecol 15:2451–2461
Dobson SL, Fox CW, Jiggins FM (2002) The effect of Wolbachia-induced cytoplasmic incompatibility on host population size in natural and manipulated systems. Proc Biol Sci 269:437–445
Fujii Y, Kubo T, Ishikawa H, Sasaki T (2004) Isolation and characterization of the bacteriophage WO from Wolbachia, an arthropod endosymbiont. Biochem Biophys Res Commun 317:1183–1188
Gavotte L, Vavre F, Heri H, Ravallec M, Sthouthamer R, Boulétreau M (2004) Diversity, distribution and specificity of WO phage infection in Wolbachia of four insect species. Insect Mol Biol 13(2):147–153
Gavotte L, Henri H, Stouthamer R, Charif D, Charlat S, Boulétreau M, Vavre F et al (2007) A survey of the bacteriophage WO in the endosymbiotic bacteria Wolbachia. Mol Biol Evol 24:427–435
Kent BN, Bordenstein SR (2010) Phage WO of Wolbachia: lambda of the endosymbiont world. Trends Microbiol 18:173–181
Louis C, Nigro L (1989) Ultrastructural evidence of Wolbachia rickettsiales in Drosophila simulans and their relationships with unidirectional cross-incompatibility. J Invertebr Pathol 54:39–44
Lu MH, Zhang KJ, Hong XY (2012) Tripartite associations among bacteriophage WO, Wolbachia, and host affected by temperature and age in Tetranychus urticae. Exp Appl Acarol 58:207–220
Masui S, Kamoda S, Sasaki T, Ishikawa H (2000) Distribution and evolution of bacteriophage WO in Wolbachia, the endosymbiont causing sexual alterations in arthropods. J Mol Evol 51:491–497
Ravikumar H, Prakash BM, Sampathkumar S, Puttaraju HP (2011) Molecular subgrouping of Wolbachia and bacteriophage WO infection among some Indian Drosophila species. J Genet 90:507–510
Rousset F, Bouchon D, Juchault P, Pintureau B, Solignac M (1992) Wolbachia endosymbionts responsible for various alterations of sexuality in arthropods. Proc Biol Sci 250:91–98
Rowley SM, Raven RJ, McGraw EA (2004) Wolbachia pipientis in Australian spiders. Curr Microbiol 49:208–214
Sinkins SP (2004) Wolbachia and cytoplasmic incompatibility in mosquitoes. Insect Biochem Mol Biol 34:723–729
Song DX, Zhu MS, Chen J (1999) The spiders of China. Hebei Science Technological Publishing House, China
Turley AP, Moreira LA, O’Neill SL, McGraw EA (2009) Wolbachia infection reduces blood-feeding success in the dengue fever mosquito, Aedes aegypti. PLoS Negl Trop Dis 3, e516
Wang ZY, Deng C, Yun YL, Chen J, Peng Y (2010) Molecular detection and the phylogenetics of Wolbachia in Chinese spiders (Araneae). J Arachnol 38:237–241
Wright JD (1979) The etiology and biology of cytoplasmic incompatibility in the Aedes scutellaris group. PhD thesis, Univ Calif, Los Angeles. 199 pp
Zhang D, Zhan X, Wu X, Yang X, Li Z (2014) A field survey for Wolbachia and phage WO infections of Aedes albopictus in Guangzhou city, China. Parasitol Res 113:399–404
Acknowledgments
This study was supported by the National Natural Science Fund of China (31401982) and the Key Scientific and Technological Projects of Hubei (2013BEC035) and Wuhan (2014020101010074). We thank Dr. Janet W. Reid (JWR Associates, New York, USA) for editing the English text.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yan, Q., Qiao, H., Gao, J. et al. Detection and phylogenetic analysis of bacteriophage WO in spiders (Araneae). Folia Microbiol 60, 497–503 (2015). https://doi.org/10.1007/s12223-015-0393-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-015-0393-z