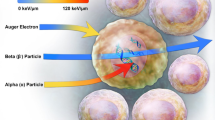
Abstract
Cancer is the second cause of death and morbidity in Europe. Unfortunately, currently available treatments cannot permanently cure most cancers, especially when metastatic. New therapy approaches are, therefore, urgently needed. Radionuclide therapy deposits cytotoxic radiation by means of energetic particles (alfa, beta, and auger) labeled to a carrier that specifically targets cancer cells. Targeted Alpha Therapy is very promising, because alpha particles deliver high energy (i.e., cytotoxic effect) in a small range, binding a target cell population without significant harm to healthy tissues. The high linear energy transfer typical of alpha particles determines irreversible double-strand DNA breaks with per-unit absorbed doses of acute biologic effects three-to-seven times greater than the damage produced by external beam with photons or beta radiation. As consequence, cells—not equipped to efficiently repair this type of damage—typically undergo death. Therefore, Targeted Alpha Therapy is such a new approach to treat tumors. This article aimed to provide an overview (five “W”s and “How”) on Targeted Alpha Therapy.
Graphical Abstract

Similar content being viewed by others

References
https://clinicaltrials.gov/ct2/results?term=alpharadin&cond=“Prostatic+Neoplasms”. Accessed 6 Jan 2020
Study of Radium-223 dichloride versus placebo and hormonal treatment as background therapy in subjects with bone predominant her2 (human epidermal growth factor receptor 2) negative hormone receptor positive metastatic breast cancer. https://clinicaltrials.gov/ct2/show/NCT02258464. Accessed 30 Dec 2019
Lintuzumab-Ac225 in Older Acute Myeloid Leukemia (AML) Patients. https://clinicaltrials.gov/ct2/show/NCT02575963. Accessed 16 Dec 2019
Study Testing Radium-223 Dichloride in Relapsed Multiple Myeloma. https://clinicaltrials.gov/ct2/show/NCT02928029. Accessed 16 Dec 2019
Phase I Dose Escalation of Monthly Intravenous Ra-223 Dichloride in Osteosarcoma. https://clinicaltrials.gov/ct2/show/record/NCT01833520?term=NCT01833520. Accessed 9 Jan 2020
A Phase I Study of Lintuzumab-Ac225 in Patients With Refractory Multiple Myeloma. https://clinicaltrials.gov/ct2/show/record/NCT02998047. Accessed 9 Dec 2019
Targeted Atomic Nano-Generators (Actinium-225-Labeled Humanized Anti-CD33 Monoclonal Antibody HuM195) in Patients With Advanced Myeloid Malignancies. https://clinicaltrials.gov/show/NCT00672165. Accessed 9 Dec 2019
211At-BC8-B10 Before donor stem cell transplant in treating patients with high-risk acute myeloid leukemia, acute lymphoblastic leukemia, myelodysplastic syndrome, or mixed-phenotype acute leukemia. https://clinicaltrials.gov/ct2/show/NCT03128034. Accessed 23 Dec 2019
211At-BC8-B10 and donor stem cell transplant in treating relapsed or refractory AML, ALL, or myelodysplastic syndrome. https://clinicaltrials.gov/ct2/show/NCT03670966?term=astatine&cond=Leukemia&draw=2&rank=2. Accessed 14 Jan 2020
A first in human study of BAY2701439 to look at safety, how the body absorbs, distributes and excretes the drug, and how well the drug works in participants with advanced cancer expressing the HER2 protein. https://clinicaltrials.gov/ct2/show/NCT04147819?term=alpha-particle&draw=2&rank=11. Accessed 18 Dec 2019
A Phase 1 study of [225Ac]-FPI-1434 injection. https://clinicaltrials.gov/ct2/show/NCT03746431?term=actinium&draw=2&rank=8. Accessed 14 Jan 2020
A study of radium-223 in combination with tasquinimod in bone-only metastatic castration-resistant prostate cancer. https://clinicaltrials.gov/ct2/show/NCT02396368?term=alpha-particle&draw=2&rank=18. Accessed 18 Dec 2019
Ahmadzadehfar H, Rahbar K, Kürpig S et al (2015) Early side effects and first results of radioligand therapy with 177Lu-DKFZ-617 PSMA of castrate-resistant metastatic prostate cancer: a two-centre study. EJNMMI Res 5:36. https://doi.org/10.1186/s13550-015-0114-2
Ahmadzadehfar H, Zimbelmann S, Yordanova A et al (2017) Radioligand therapy of metastatic prostate cancer using 177Lu-PSMA-617 after radiation exposure to 223Ra-dichloride. Oncotarget 8:6–7. https://doi.org/10.18632/oncotarget.15698
Allan BJ (2013) Systemic targeted alpha radiotherapy for cancer. J Biomed Phys Eng 3:67–80
Allen BJ (2012) Systemic targeted alpha radiotherapy for cancer. J Proc R Soc New South Wales 145:19–33. https://doi.org/10.1358/dnp.2008.21.6.1246832.disrupting
Allen BJ, Raja C, Rizvi S et al (2005) Intralesional targeted alpha therapy for metastatic melanoma. Cancer Biol Ther 4:1318–1324
Allen BJ, Singla AA, Rizvi SMA et al (2011) Analysis of patient survival in a Phase I trial of systemic targeted α-therapy for metastatic melanoma. Immunotherapy 3:1041–1050. https://doi.org/10.2217/imt.11.97
Andersson H, Lindegren S, Bäck T et al (2000) The curative and palliative potential of the monoclonal antibody MOv18 labelled with 211At in nude mice with intraperitoneally growing ovarian cancer xenografts—a long-term study. Acta Oncol 39:741–745. https://doi.org/10.1080/028418600750063820
Andersson H, Cederkrantz E, Back T et al (2009) Intraperitoneal-particle radioimmunotherapy of ovarian cancer patients: pharmacokinetics and dosimetry of 211At-MX35 F(ab’)2—a phase I study. J Nucl Med 50:1153–1160. https://doi.org/10.2967/jnumed.109.062604
Armstrong DK, Bundy B, Wenzel L et al (2006) Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N Engl J Med 354:34–43. https://doi.org/10.1056/nejmoa052985
Atallah E, Berger M, Jurcic J et al (2019) A phase 2 study of actinium-225 (225Ac)-lintuzumab in older patients with untreated acute myeloid leukemia (AML). J Med Imaging Radiat Sci 50:S37. https://doi.org/10.1016/j.jmir.2019.03.113
Baidoo KE, Yong K, Brechbiel MW (2013) Molecular pathways: targeted α-particle radiation therapy. Clin Cancer Res 19:530–537. https://doi.org/10.1158/1078-0432.ccr-12-0298
Ballal S, Yadav MP, Bal C et al (2019) Broadening horizons with 225Ac-DOTATATE targeted alpha therapy for gastroenteropancreatic neuroendocrine tumour patients stable or refractory to 177Lu-DOTATATE PRRT: first clinical experience on the efficacy and safety. Eur J Nucl Med Mol Imaging. https://doi.org/10.1007/s00259-019-04567-2
Bander N, Milosky M, Nanus D et al (2005) Phase I trial of 177lutetium-labeled J591, a monoclonal antibody to prostate-specific membrane antigen, in patients with androgen-independent prostate cancer. J Clin Oncol 23:4591–4601. https://doi.org/10.1200/jco
Bařinka C, Rojas C, Slusher B, Pomper M (2012) Glutamate carboxypeptidase II in diagnosis and treatment of neurologic disorders and prostate cancer. Curr Med Chem 19:856–870. https://doi.org/10.2174/092986712799034888
Behe M, Alt K, Deininger F et al (2011) In vivo testing of 177Lu-labelled anti-PSMA antibody as a new radioimmunotherapeutic agent against prostate cancer. In Vivo (Brooklyn) 25:55–59
Behnam Azad B, Banerjee SR, Pullambhatla M et al (2015) Evaluation of a PSMA-targeted BNF nanoparticle construct. Nanoscale 7:4432–4442. https://doi.org/10.1039/c4nr06069e
Behr TM, Griesinger F, Riggert J et al (2002) High-dose myeloablative radioimmunotherapy of mantle cell non-Hodgkin lymphoma with the iodine-131-labeled chimeric anti-CD20 antibody C2B8 and autologous stem cell support. Results of a pilot study. Cancer 94:1363–1372. https://doi.org/10.1002/cncr.10307
Bethge WA, Wilbur DS, Sandmaier BM (2006) Radioimmunotherapy as non-myeloablative conditioning for allogeneic marrow transplantation. Leuk Lymphoma 47:1205–1214. https://doi.org/10.1080/00423110500485822
Bourgeois DJ, Kraus S, Maaloof BN, Sartor O (2011) Radiation for bone metastases. Curr Opin Support Palliat Care 5:227–232. https://doi.org/10.1097/spc.0b013e3283499caa
Buchmann I, Meyer RG, Mier W, Haberkorn U (2009) Myeloablative radioimmunotherapy in conditioning prior to haematological stem cell transplantation: closing the gap between benefit and toxicity? Eur J Nucl Med Mol Imaging 36:484–498. https://doi.org/10.1007/s00259-008-0996-6
Cederkrantz E, Andersson H, Bernhardt P et al (2015) Absorbed doses and risk estimates of 211At-MX35 F(ab′) < inf > 2</inf > in intraperitoneal therapy of ovarian cancer patients. Int J Radiat Oncol Biol Phys 93:569–576. https://doi.org/10.1016/j.ijrobp.2015.07.005
Chang C, Sharkey RM, Rossi EA et al (2002) Molecular advances in pretargeting radioimunotherapy with bispecific antibodies. Mol Cancer Ther 1:553–563
Coleman R (2016) Treatment of metastatic bone disease and the emerging role of radium-223. Semin Nucl Med 46:99–104. https://doi.org/10.1053/j.semnuclmed.2015.10.012
Coleman R, Aksnes AK, Naume B et al (2014) A phase IIa, nonrandomized study of radium-223 dichloride in advanced breast cancer patients with bone-dominant disease. Breast Cancer Res Treat 145:411–418. https://doi.org/10.1007/s10549-014-2939-1
Contessa JN, Roberson PL (2006) Targeted radiation for melanoma: alpha therapy prepares for a beta test. Cancer Biol Ther 5:118–119. https://doi.org/10.4161/cbt.5.1.2401
Cordier D, Forrer F, Bruchertseifer F et al (2010) Targeted alpha-radionuclide therapy of functionally critically located gliomas with 213Bi-DOTA-[Thi8, Met(O2)11]- substance P: a pilot trial. Eur J Nucl Med Mol Imaging 37:1335–1344. https://doi.org/10.1007/s00259-010-1385-5
Dadwal M, Kang CS, Song HA et al (2011) Synthesis and evaluation of a bifunctional chelate for development of Bi(III)-labeled radioimmunoconjugates. Bioorg Med Chem Lett 21:7513–7515. https://doi.org/10.1016/j.bmcl.2011.06.107.synthesis
Data explorer|ECIS. https://ecis.jrc.ec.europa.eu/explorer.php?$0-0$1-AE28$4-1,2$3-All$6-0,14$5-2008,2008$7-8$CEstByCancer$X0_8-3$CEstRelativeCanc$X1_8-3$X1_9-AE28. Accessed 6 Dec 2019
de Kruijff RM, Raavé R, Kip A et al (2019) The in vivo fate of 225Ac daughter nuclides using polymersomes as a model carrier. Sci Rep 9:11671. https://doi.org/10.1038/s41598-019-48298-8
Dekempeneer Y, Keyaerts M, Krasniqi A et al (2016) Targeted alpha therapy using short-lived alpha-particles and the promise of nanobodies as targeting vehicle. Expert Opin Biol Ther 16:1035–1047. https://doi.org/10.1080/14712598.2016.1185412
Dekempeneer Y, Bäck T, Aneheim E et al (2019) Labeling of anti-HER2 nanobodies with astatine-211: optimization and the effect of different coupling reagents on their in vivo behavior. Mol Pharm 16:3524–3533. https://doi.org/10.1021/acs.molpharmaceut.9b00354
D’Huyvetter M, Xavier C, Caveliers V et al (2014) Radiolabeled nanobodies as theranostic tools in targeted radionuclide therapy of cancer. Expert Opin Drug Deliv 131:1–16. https://doi.org/10.1517/17425247.2014.941803
Elgqvist J, Andersson H, Bäck T et al (2006) Fractionated radioimmunotherapy of intraperitoneally growing ovarian cancer in nude mice with 211At-MX35 F(ab′)2: therapeutic efficacy and myelotoxicity. Nucl Med Biol 33:1065–1072. https://doi.org/10.1016/j.nucmedbio.2006.07.009
Elgqvist J, Frost S, Pouget J-P, Albertsson P (2014) The potential and hurdles of targeted alpha therapy—clinical trials and beyond. Front Oncol 3:324. https://doi.org/10.3389/fonc.2013.00324
Enzalutamide with or without radium Ra 223 dichloride in patients with metastatic, castration-resistant prostate cancer. https://clinicaltrials.gov/ct2/show/NCT03344211?term=alpha-particle&draw=2&rank=13. Accessed 18 Dec 2019
European Commission (2014) Council Directive 2013/59/Euratom of 5 December 2013. Off J Eur Union. https://doi.org/10.3000/19770677.l_2013.124.eng
Evans JC, Malhotra M, Cryan JF, O’Driscoll CM (2016) The therapeutic and diagnostic potential of the prostate specific membrane antigen/glutamate carboxypeptidase II (PSMA/GCPII) in cancer and neurological disease. Br J Pharmacol. https://doi.org/10.1111/bph.13576
Exploratory study of radium-223 and VEGF-targeted therapy in patients with metastatic renal cell carcinoma and bone mets. https://clinicaltrials.gov/show/NCT02406521
First-in-human study of BAY2287411 injection, a thorium-227 labeled antibody-chelator conjugate, in patients with tumors known to express mesothelin. https://clinicaltrials.gov/ct2/show/NCT03507452. Accessed 9 Jan 2020
Friesen C, Glatting G, Koop B et al (2007) Breaking chemoresistance and radioresistance with [213Bi]anti- CD45 antibodies in leukemia cells. Cancer Res 67:1950–1958. https://doi.org/10.1158/0008-5472.can-06-3569
Friesen C, Roscher M, Hormann I et al (2013) Anti-CD33-antibodies labelled with the alpha-emitter Bismuth-213 kill CD33-positive acute myeloid leukaemia cells specifically by activation of caspases and break radio- and chemoresistance by inhibition of the anti-apoptotic proteins X-linked inhibitor o. Eur J Cancer 49:2542–2554. https://doi.org/10.1016/j.ejca.2013.04.008
Gao X-F, Zhou T, Chen G-H et al (2014) Radioiodine therapy for castration-resistant prostate cancer following prostate-specific membrane antigen promoter-mediated transfer of the human sodium iodide symporter. Asian J Androl 16:120–123. https://doi.org/10.4103/1008-682x.122354
Gudkov SV, Shilyagina NY, Vodeneev VA, Zvyagin AV (2016) Targeted radionuclide therapy of human tumors. Int J Mol Sci 17:33. https://doi.org/10.3390/ijms17010033
Hadaschik B, Boegemann M (2017) Why targeting PSMA is a valuable addition in the management of castration-resistant prostate cancer: the Urologists’ point of view. J Nucl Med. https://doi.org/10.2967/jnumed.117.194753
Hamers-Casterman C, Atarhouch T, Muyldermans S et al (1993) Naturally occurring antibodies devoid of light chains. Nature 363:446–448. https://doi.org/10.1038/363446a0
Hammer S, Hagemann UB, Zitzmann-Kolbe S et al (2019) Preclinical efficacy of a PSMA-targeted thorium-227 conjugate (PSMA-TTC), a targeted alpha therapy for prostate cancer. Clin Cancer Res Clin 2268:2019. https://doi.org/10.1158/1078-0432.ccr-19-2268
Heeger S, Moldenhauer G, Egerer G et al (2003) Alpha-radioimmunotherapy of B-lineage non-Hodgkin’s lymphoma using 213Bi-labelled anti-CD19-and anti-CD20-CHX-A″-DTPA conjugates. Abstr Pap Am Chem Soc 225:U261
Heidenreich A, Gillessen S, Heinrich D et al (2019) Radium-223 in asymptomatic patients with castration-resistant prostate cancer and bone metastases treated in an international early access program. BMC Cancer 19:12. https://doi.org/10.1186/s12885-018-5203-y
Hicks RJ, Kwekkeboom DJ, Krenning E et al (2017) ENETS consensus guidelines for the standards of care in neuroendocrine neoplasia: peptide receptor radionuclide therapy with radiolabeled somatostatin analogues. Neuroendocrinology 105:295–309. https://doi.org/10.1159/000475526
Home-ClinicalTrials.gov. https://clinicaltrials.gov/. Accessed 8 Jan 2020
Horoszkiewicz J, Kawinski E, Murphy G (1987) Monoclonal antibodies to a new antigenic market in epithelial cells and serum of prostatic cancer patients. Anticancer Res 7:927–936
Humm JL, Sartor O, Parker C et al (2015) Radium-223 in the treatment of osteoblastic metastases: a critical clinical review. Int J Radiat Oncol Biol Phys 91:898–906. https://doi.org/10.1016/j.ijrobp.2014.12.061
Israeli RS, Powell CT, Fair WR, Heston WD (1993) Molecular cloning of a complementary DNA encoding a prostate-specific membrane antigen. Cancer Res 53:227–230
Jurcic JG, Larson SM, Sgouros G et al (2002) Targeted alpha particle immunotherapy for myeloid leukemia. Blood 100:1233–1239
Kassis A (2008) Therapeutic radionuclides: biophysical and radiobiologic principles. Semin Nucl Med 38:358–366. https://doi.org/10.1053/j.semnuclmed.2008.05.002.therapeutic
Kellerer AM, Chmelevsky D (1975) Concepts of microdosimetry. I. Quantities. Radiat Environ Biophys 12:61–69. https://doi.org/10.1007/bf02339810
Khreish F, Ebert N, Ries M et al (2019) 225Ac-PSMA-617/177Lu-PSMA-617 tandem therapy of metastatic castration-resistant prostate cancer: pilot experience. Eur J Nucl Med Mol Imaging. https://doi.org/10.1007/s00259-019-04612-0
Kiess AP, Minn I, Chen Y et al (2015) Auger radiopharmaceutical therapy targeting prostate-specific membrane antigen. J Nucl Med 56:1401–1407. https://doi.org/10.2967/jnumed.115.155929
Kneifel S, Cordier D, Good S et al (2006) Local targeting of malignant gliomas by the diffusible peptidic. Clin Cancer Res 12:3843–3850. https://doi.org/10.1158/1078-0432.CCR-05-2820
Kratochwil C, Giesel FL, Bruchertseifer F et al (2014) 213Bi-DOTATOC receptor-targeted alpha-radionuclide therapy induces remission in neuroendocrine tumours refractory to beta radiation: a first-in-human experience. Eur J Nucl Med Mol Imaging 41:2106–2119. https://doi.org/10.1007/s00259-014-2857-9
Kratochwil C, Giesel FL, Eder M et al (2015) [177Lu]Lutetium-labelled PSMA ligand-induced remission in a patient with metastatic prostate cancer. Eur J Nucl Med Mol Imaging 42:987–988. https://doi.org/10.1007/s00259-014-2978-1
Kratochwil C, Bruchertseifer F, Giesel FL et al (2016) 225Ac-PSMA-617 for PSMA-targeted -radiation therapy of metastatic castration-resistant prostate cancer. J Nucl Med 57:1941–1944. https://doi.org/10.2967/jnumed.116.178673
Kratochwil C, Bruchertseifer F, Rathke H et al (2017) Targeted Alpha Therapy of mCRPC with 225 actinium-PSMA-617: dosimetry estimate and empirical dose finding. J Nucl Med. https://doi.org/10.2967/jnumed.117.191395
Kratochwil C, Bruchertseifer F, Rathke H et al (2018) Targeted α-therapy of metastatic castration-resistant prostate cancer with 225 Ac-PSMA-617: swimmer-plot analysis suggests efficacy regarding duration of tumor control. J Nucl Med 59:795–802. https://doi.org/10.2967/jnumed.117.203539
Kratochwil C, Giesel FL, Heussel CP et al (2019) Patients resistant against PSMA-targeting alpha-radiation therapy often harbor mutations in DNA-repair associated genes. J Nucl Med. https://doi.org/10.2967/jnumed.119.234559
Królicki L, Bruchertseifer F, Kunikowska J et al (2019) Safety and efficacy of targeted alpha therapy with 213Bi-DOTA-substance P in recurrent glioblastoma. Eur J Nucl Med Mol Imaging 46:614–622. https://doi.org/10.1007/s00259-018-4225-7
Lange PH, Vessella RL (1999) Mechanisms, hypotheses and questions regarding prostate cancer micrometastases to bone. Cancer Metastasis Rev 17:331–336. https://doi.org/10.1023/a:1006106209527
Lesch HP, Kaikkonen MU, Pikkarainen JT, Ylä-Herttuala S (2010) Avidin–biotin technology in targeted therapy. Expert Opin Drug Deliv 7:551–564. https://doi.org/10.1517/17425241003677749
Lintuzumab-Ac225 in combination with CLAG-M chemotherapy in patients with relapsed/refractory acute myeloid leukemia. https://clinicaltrials.gov/ct2/show/NCT03441048?term=actinium&draw=2&rank=9. Accessed 14 Jan 2020
Macey DJ, Meredith RF (1999) A strategy to reduce red marrow dose for intraperitoneal radioimmunotherapy. Clin Cancer Res 5:1–2
McKay RR, Gray KP, Polacek L et al (2017) An exploratory study of radium-223 and vascular endothelial growth factor targeted therapy (VEGF TT). J Clin Oncol 35:suppl:Abstract 466
Medicines Agency E (2018) EMA restricts use of prostate cancer medicine Xofigo
Meredith R, Torgue J, Shen S et al (2014a) Dose escalation and dosimetry of first-in-human radioimmunotherapy with 212Pb-TCMC-trastuzumab. J Nucl Med 55:1636–1642. https://doi.org/10.2967/jnumed.114.143842
Meredith RF, Torgue J, Azure MT et al (2014b) Pharmacokinetics and imaging of 212 Pb-TCMC-trastuzumab after intraperitoneal administration in ovarian cancer patients. Cancer Biother Radiopharm 29:12–17. https://doi.org/10.1089/cbr.2013.1531
Miederer M, Henriksen G, Alke A et al (2008) Preclinical evaluation of the alpha-particle generator nuclide 225Ac for somatostatin receptor radiotherapy of neuroendocrine tumors. Clin Cancer Res 14:3555–3561. https://doi.org/10.1158/1078-0432.ccr-07-4647
Morgenstern A, Krolicki L, Kunikowska J et al (2014) Targeted alpha therapy of glioblastoma multiforme: first clinical experience with 213Bi-substance P. J Nucl Med 55:390
Mottet N, Bellmunt J, Briers E et al (2020) EAU – ESTRO – ESUR – SIOG Guidelines on Prostate Cancer, Edn. In: Presented at the EAU annual congress amsterdam 2020. EAU Guidelines Office, Arnhem, The Netherlands. https://uroweb.org/guideline/prostate-cancer/
Nademanee A, Forman S, Molina A et al (2005) A phase 1/2 trial of high-dose yttrium-90-ibritumomab tiuxetan in combination with high-dose etoposide and cyclophosphamide followed by autologous stem cell transplantation in patients with poor-risk or relapsed non-Hodgkin lymphoma. Blood 106:2896–2902. https://doi.org/10.1182/blood-2005-03-1310
Nilsson S, Larsen RH, Fossa SD et al (2005) First clinical experience with alpha-emitting radium-223 in the treatment of skeletal metastases. Clin Cancer Res 11:4451–4459. https://doi.org/10.1158/1078-0432.ccr-04-2244
Nilsson S, Franzen L, Parker C et al (2013) Two-year survival follow-up of the randomized, double-blind, placebo-controlled phase II study of radium-223 chloride in patients with castration-resistant prostate cancer and bone metastases. Clin Genitourin Cancer 11:20–26. https://doi.org/10.1016/j.clgc.2012.07.002
Norenberg JP (2006) 213Bi-[DOTA0, Tyr3]octreotide peptide receptor radionuclide therapy of pancreatic tumors in a preclinical animal model. Clin Cancer Res 12:897–903. https://doi.org/10.1158/1078-0432.ccr-05-1264
O’Keefe DS, Su SL, Bacich DJ et al (1998) Mapping, genomic organization and promoter analysis of the human prostate-specific membrane antigen gene. Biochim Biophys Acta 1443:113–127
Oliveira S, Schiffelers RM, van der Veeken J et al (2010) Downregulation of EGFR by a novel multivalent nanobody-liposome platform. J Control Release 145:165–175. https://doi.org/10.1016/j.jconrel.2010.03.020
Open label phase two trial of radium Ra 223 dichloride with concurrent administration of abiraterone acetate plus prednisone in symptomatic castration-resistant (hormone-refractory) prostate cancer subjects with bone metastasis (eRADicAte). https://clinicaltrials.gov/ct2/show/record/NCT02097303?term=alpha-particle&draw=2&rank=16. Accessed 18 Dec 2019
Pagel JM, Kenoyer AL, Bäck T et al (2011) Anti-CD45 pretargeted radioimmunotherapy using bismuth-213: high rates of complete remission and long-term survival in a mouse myeloid leukemia xenograft model. Blood 118:703–711. https://doi.org/10.1182/blood-2011-04-347039
Palm S, Bäck T, Claesson I et al (2007) Therapeutic efficacy of astatine-211-labeled trastuzumab on radioresistant SKOV-3 tumors in nude mice. Int J Radiat Oncol Biol Phys 69:572–579. https://doi.org/10.1016/j.ijrobp.2007.06.023
Park SI, Shenoi J, Pagel JM et al (2010) Conventional and pretargeted radioimmunotherapy with bismuth-213 to target and treat CD20-expressing non-Hodgkin lymphoma: a preclinical model for consolidation therapy to eradicate minimal residual disease. Blood 116:4231–4239. https://doi.org/10.1182/blood-2010-05-282327.the
Parker C, Nilsson S, Heinrich D et al (2013a) Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med 369:213–223. https://doi.org/10.1056/nejmoa1213755
Parker CC, Pascoe S, Chodacki A et al (2013b) A randomized, double-blind, dose-finding, multicenter, phase 2 study of radium chloride (Ra 223) in patients with bone metastases and castration-resistant prostate cancer. Eur Urol 63:189–197. https://doi.org/10.1016/j.eururo.2012.09.008
Petrich T, Korkmaz Z, Krull D et al (2010) In vitro experimental (211)At-anti-CD33 antibody therapy of leukaemia cells overcomes cellular resistance seen in vivo against gemtuzumab ozogamicin. Eur J Nucl Med Mol Imaging 37:851–861. https://doi.org/10.1007/s00259-009-1356-x
Phase 1 study of AlphaMedixTM in adult subjects with SSTR (+) NET. https://clinicaltrials.gov/ct2/show/NCT03466216?term=alpha-particle&draw=2&rank=12. Accessed 18 Dec 2019
Popovtzer A, Rosenfeld E, Mizrachi A et al (2019) Initial safety and tumor control results from a “first-in-human” multicenter prospective trial evaluating a novel alpha-emitting radionuclide for the treatment of locally advanced recurrent squamous cell carcinomas of the skin and head and neck. Int J Radiat Oncol. https://doi.org/10.1016/j.ijrobp.2019.10.048
Pozzi OR, Zalutsky MR (2017) Radiopharmaceutical chemistry of targeted radiotherapeutics, part 4: strategies for 211 At labeling at high activities and radiation doses of At α -particles. Nucl Med Biol 46:43–49. https://doi.org/10.1016/j.nucmedbio.2016.11.009
Price EW, Orvig C (2014) Matching chelators to radiometals for radiopharmaceuticals. Chem Soc Rev 43:260–290. https://doi.org/10.1039/c3cs60304k
Rahbar K, Ahmadzadehfar H, Kratochwil C et al (2017) German multicenter study investigating 177 Lu-PSMA-617 radioligand therapy in advanced prostate cancer patients. J Nucl Med 58:85–90. https://doi.org/10.2967/jnumed.116.183194
Raja C, Graham P, Abbas Rizvi SM et al (2007) Interim analysis of toxicity and response in phase 1 trial of systemic targeted alpha therapy for metastatic melanoma. Cancer Biol Ther 6:846–852. https://doi.org/10.4161/cbt.6.6.4089
Ramdahl T, Bonge-hansen HT, Ryan OB et al (2016) An efficient chelator for complexation of thorium-227. Bioorgan Med Chem Lett 26:4318–4321. https://doi.org/10.1016/j.bmcl.2016.07.034
Richman CM, Denardo SJ, O’Donnell RT et al (2005) High-dose radioimmunotherapy combined with fixed, low-dose paclitaxel in metastatic prostate and breast cancer by using a MUC-1 monoclonal antibody, m170, linked to indium-111/yttrium-90 via a cathepsin cleavable linker with cyclosporine to prevent human. Clin Cancer Res 11:5920–5927. https://doi.org/10.1158/1078-0432.ccr-05-0211
Rosenblat TL, McDevitt MR, Mulford DA et al (2010) Sequential cytarabine and alpha-particle immunotherapy with bismuth-213-lintuzumab (HuM195) for acute myeloid leukemia. Clin Cancer Res 16:5303–5311. https://doi.org/10.1158/1078-0432.ccr-10-0382
Saad F, Gillessen S, Heinrich D et al (2019) Disease characteristics and completion of treatment in patients with metastatic castration-resistant prostate cancer treated with radium-223 in an international early access program. Clin Genitourin Cancer 17:348.e5–355.e5. https://doi.org/10.1016/j.clgc.2019.05.012
Safety study of 212Pb-TCMC-trastuzumab radio immunotherapy. https://clinicaltrials.gov/ct2/show/NCT01384253. Accessed 23 Dec 2019
Safety and tolerability of BAY1862864 injection in subjects with relapsed or refractory CD22-positive non-Hodgkin’s lymphoma. https://clinicaltrials.gov/show/NCT02581878. Accessed 23 Dec 2019
Sartor O, Coleman R, Nilsson S et al (2014) Effect of radium-223 dichloride on symptomatic skeletal events in patients with castration-resistant prostate cancer and bone metastases: results from a phase 3, double-blind, randomised trial. Lancet Oncol 15:738–746. https://doi.org/10.1016/s1470-2045(14)70183-4
Search of: radium-223|recruiting, active, not recruiting studies|interventional studies|prostate cancer—list results—ClinicalTrials.gov. https://clinicaltrials.gov/ct2/results?term=radium-223&cond=Prostate+Cancer&recrs=a&recrs=d&age_v=&gndr=&type=Intr&rslt=&Search=Apply. Accessed 7 Jan 2020
Sgouros G (2008) Alpha-particles for targeted therapy. Adv Drug Deliv Rev 60:1402–1406. https://doi.org/10.1016/j.addr.2008.04.007
Sgouros G, Roeske JC, McDevitt MR et al (2010) MIRD Pamphlet no. 22 (abridged): radiobiology and dosimetry of -particle emitters for targeted radionuclide therapy. J Nucl Med 51:311–328. https://doi.org/10.2967/jnumed.108.058651
Sollini M, Boni R, Traino ACC et al (2015) New approaches for imaging and therapy of solid cancer. Q J Nucl Med Mol Imaging 59:168–183
Study evaluating the addition of pembrolizumab to radium-223 in mCRPC. https://clinicaltrials.gov/ct2/show/NCT03093428?term=alpha-particle&draw=2&rank=17. Accessed 18 Dec 2019
Study of radium-223 dichloride versus placebo and treatment with exemestane/everolimus in subjects with bone predominant HER2 (human epidermal growth factor receptor 2) negative hormone receptor positive metastatic breast cancer. https://clinicaltrials.gov/ct2/show/NCT02258451. Accessed 23 Dec 2019
Study to test the safety and how radium-223 dichloride an alpha particle-emitting radioactive agent works in combination with pembrolizumab an immune checkpoint inhibitor in patients with stage IV non-small cell lung cancer with bone metastases. https://clinicaltrials.gov/ct2/show/NCT03996473?term=alpha-particle&draw=2&rank=9. Accessed 18 Dec 2019
Tagawa ST, Milowsky MI, Morris M et al (2013) Phase II study of lutetium-177-labeled anti-prostate-specific membrane antigen monoclonal antibody J591 for metastatic castration-resistant prostate cancer. Clin Cancer Res 19:5182–5191. https://doi.org/10.1158/1078-0432.ccr-13-0231
Targeted Alpha Therapy Working Group, Parker C, Lewington V et al (2018) Targeted alpha therapy, an emerging class of cancer agents: a review. JAMA Oncol 4:1765–1772. https://doi.org/10.1001/jamaoncol.2018.4044
Total body irradiation and astatine-211-labeled BC8-B10 monoclonal antibody for the treatment of nonmalignant diseases. https://clinicaltrials.gov/ct2/show/NCT04083183?term=astatine&draw=2&rank=1. Accessed 14 Jan 2020
Trial of Ra-223 dichloride in combination with hormonal therapy and denosumab in the treatment of patients with hormone-positive bone-dominant metastatic breast cancer. https://clinicaltrials.gov/show/NCT02366130. Accessed 23 Dec 2019
Vallabhajosula S, Smith-Jones PM, Navarro V et al (2004) Radioimmunotherapy of prostate cancer in human xenografts using monoclonal antibodies specific to prostate specific membrane antigen (PSMA): studies in nude mice. Prostate 58:145–155. https://doi.org/10.1002/pros.10281
Vallabhajosula S, Goldsmith SJ, Kostakoglu L et al (2005) Radioimmunotherapy of prostate cancer using 90Y- and 177Lu-labeled J591 monoclonal antibodies: effect of multiple treatments on myelotoxicity. Clin Cancer Res 11:7195s–7200s. https://doi.org/10.1158/1078-0432.ccr-1004-0023
Vaneycken I, Govaert J, Vincke C et al (2010) In vitro analysis and in vivo tumor targeting of a humanized, grafted nanobody in mice using pinhole SPECT/micro-CT. J Nucl Med 51:1099–1106. https://doi.org/10.2967/jnumed.109.069823
Vaneycken I, Devoogdt N, Van Gassen N et al (2011) Preclinical screening of anti-HER2 nanobodies for molecular imaging of breast cancer. FASEB J 25:2433–2446. https://doi.org/10.1096/fj.10-180331
Venetoclax, azacitidine, and lintuzumab-Ac225 in AML patients. https://clinicaltrials.gov/ct2/show/NCT03932318?term=actinium&draw=2&rank=5. Accessed 14 Jan 2020
Venetoclax and lintuzumab-Ac225 in AML patients. https://clinicaltrials.gov/ct2/show/NCT03867682?term=actinium&draw=2&rank=6. Accessed 14 Jan 2020
Verheijen RH, Massuger LF, Benigno BB et al (2006) Phase III trial of intraperitoneal therapy with yttrium-90-labeled HMFG1 murine monoclonal antibody in patients with epithelial ovarian cancer after a surgically defined complete remission. J Clin Oncol 24:571–578. https://doi.org/10.1200/jco.2005.02.5973
Wilbur DS (2011) Chemical and radiochemical considerations in radiolabeling with α-emitting radionuclides. Curr Radiopharm 4:214–247. https://doi.org/10.2174/1874471011104030214
Yao V, Parwani A, Maier C et al (2008) Moderate expression of prostate-specific membrane antigen, a tissue differentiation antigen and folate hydrolase, facilitates prostate carcinogenesis. Cancer Res 68:9070–9077. https://doi.org/10.1158/0008-5472.can-08-2328
Zaknun JJ, Bodei L, Mueller-Brand J et al (2013) The joint IAEA, EANM, and SNMMI practical guidance on peptide receptor radionuclide therapy (PRRNT) in neuroendocrine tumours. Eur J Nucl Med Mol Imaging 40:800–816. https://doi.org/10.1007/s00259-012-2330-6
Zalutsky MR, Reardon DA, Akabani G et al (2008) Clinical experience with -particle emitting 211At: treatment of recurrent brain tumor patients with 211At-labeled chimeric antitenascin monoclonal antibody 81C6. J Nucl Med 49:30–38. https://doi.org/10.2967/jnumed.107.046938
Zechmann CM, Afshar-Oromieh A, Armor T et al (2014) Radiation dosimetry and first therapy results with a 124I/131I-labeled small molecule (MIP-1095) targeting PSMA for prostate cancer therapy. Eur J Nucl Med Mol Imaging 41:1280–1292. https://doi.org/10.1007/s00259-014-2713-y
Zhang M, Zhang Z, Garmestani K et al (2003) Pretarget radiotherapy with an anti-CD25 antibody-streptavidin fusion protein was effective in therapy of leukemia/lymphoma xenografts. Proc Natl Acad Sci USA 100:1891–1895. https://doi.org/10.1073/pnas.0437788100
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
MS, KM, and MK declare no conflict of interest. AC received speaker honoraria from General Electric and Blue Earth Diagnostics, acted as a scientific advisor for Blue Earth Diagnostics and Advanced Accelerator Applications, and benefited from an unconditional Grant from Sanofi to Institution. All honoraria and Grants are outside the scope of the submitted work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The author M. Sollini received the international prize “Francesco de Luca” for Cancer Research in 2019, attributed by the Accademia Nazionale dei Lincei, Rome.
Rights and permissions
About this article
Cite this article
Sollini, M., Marzo, K., Chiti, A. et al. The five “W”s and “How” of Targeted Alpha Therapy: Why? Who? What? Where? When? and How?. Rend. Fis. Acc. Lincei 31, 231–247 (2020). https://doi.org/10.1007/s12210-020-00900-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12210-020-00900-2