Abstract
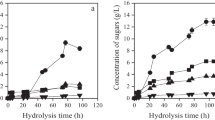
A novel simplified configuration is proposed for the conversion of biomass to ethanol using whole medium enzymatic cocktails (WM) and enzymatic extracts (EE) from different filamentous fungi (Trichoderma reesei, Aspergillus niger, and Aspergillus oryzae) cultivated under solid-state fermentation (SSF) for the hydrolysis of steam-exploded sugarcane bagasse (SESB). The hydrolyzed material derived from the saccharification of SESB using the combinations A. niger WM + T. reesei EE, A. oryzae WM + A. niger EE, and A. niger EE + T. reesei WM resulted in the best biomass conversion yields (66, 65, and 64 % of the theoretical reducing sugar yields, respectively). The best ethanol production (84 % of the theoretical yield) was obtained using the material hydrolyzed by a combination of A. oryzae WM + A. niger EE. The enzymatic conversion of SESB using on-site produced enzymes from the whole SSF cultivation medium, followed by an ethanol production step, is a potential configuration for the biomass to ethanol conversion process. This novel simplified configuration would enable the use of a single reactor system, avoiding the need for additional separation steps.



Similar content being viewed by others
References
Pottmaier D, Melo CR, Sartor MN, Kuester S, Amadio TM, Fernandes CAH, Marinha D, Alarcon OE (2013) The Brazilian energy matrix: from a materials science and engineering perspective. Renew Sust Energ Rev 19:678–691
Amorim HV, Lopes ML, Oliveira JVD, Buckeridge MS, Goldman GH (2011) Scientific challenges of bioethanol production in Brazil. Appl Microbiol Biotechnol 91:1267–1275
Horn SJ, Vaaje-Kolstad G, Westereng B, Eijsink VGH (2012) Novel enzymes for the degradation of cellulose. Biotechnol Biofuels 5:45
Giese EC, Pierozzi M, Dussan KJ, Chandel AK, da Silva SS (2013) Enzymatic saccharification of acid-alkali pretreated sugarcane bagasse using commercial enzyme preparations. J Chem Technol Biotechnol 88:1266–1272
Delabona PD, Cota J, Hoffmam ZB, Paixao DAA, Farinas CS, Cairo J, Lima DJ, Squina FM, Ruller R, Pradella JGD (2013) Understanding the cellulolytic system of Trichoderma harzianum P49P11 and enhancing saccharification of pretreated sugarcane bagasse by supplementation with pectinase and alpha-l-arabinofuranosidase. Bioresour Technol 131:500–507
Rodriguez-Zuniga UF, Couri S, Neto VB, Crestana S, Farinas CS (2013) Integrated strategies to enhance cellulolytic enzyme production using an instrumented bioreactor for solid-state fermentation of sugarcane bagasse. Bioenerg Res 6:142–152
Sukumaran RK, Singhania RR, Mathew GM, Pandey A (2009) Cellulase production using biomass feed stock and its application in lignocellulose saccharification for bio-ethanol production. Renew Energy 34:421–424
Klein-Marcuschamer D, Oleskowicz-Popiel P, Simmons BA, Blanch HW (2012) The challenge of enzyme cost in the production of lignocellulosic biofuels. Biotechnol Bioeng 109:1083–1087
Gusakov AV (2011) Alternatives to Trichoderma reesei in biofuel production. Trends Biotechnol 29:419–425
King BC, Waxman KD, Nenni NV, Walker LP, Bergstrom GC, Gibson DM (2011) Arsenal of plant cell wall degrading enzymes reflects host preference among plant pathogenic fungi. Biotechnol Biofuels 4:14
Berlin A, Maximenko V, Gilkes N, Saddler J (2007) Optimization of enzyme complexes for lignocellulose hydrolysis. Biotechnol Bioeng 97:287–296
Ximenes EA, Dien BS, Ladisch MR, Mosier N, Cotta MA, Li XL (2007) Enzyme production by industrially relevant fungi cultured on coproduct from corn dry grind ethanol plants. Appl Biochem Biotechnol 137:171–183
Kovacs K, Szakacs G, Zacchi G (2009) Enzymatic hydrolysis and simultaneous saccharification and fermentation of steam-pretreated spruce using crude Trichoderma reesei and Trichoderma atroviride enzymes. Process Biochem 44:1323–1329
Lu J, Weerasiri RR, Liu Y, Wang W, Ji SW, Lee I (2013) Enzyme production by the mixed fungal culture with nano-shear pretreated biomass and lignocellulose hydrolysis. Biotechnol Bioeng 110:2123–2130
Pirota RSPB, Baleeiro FC, Farinas CS (2013) Saccharification of biomass using whole solid-state fermentation medium to avoid additional separation steps. Biotechnol Prog 29:1430–1440
Schell DJ, Hinman ND, Wyman CE, Werdene PJ (1990) Whole broth cellulase production for use in simultaneous saccharification and fermentation. Appl Biochem Biotechnol 24–5:287–297
Park EY, Naruse K, Kato T (2012) One-pot bioethanol production from cellulose by co-culture of Acremonium cellulolyticus and Saccharomyces cerevisiae. Biotechnol Biofuels 5:64
Raimbault R (1998) General and microbiological aspects of solid substrate fermentation. Eletron J Biotechnol 1:3–45
Singhania RR, Sukumaran RK, Patel AK, Larroche C, Pandey A (2010) Advancement and comparative profiles in the production technologies using solid-state and submerged fermentation for microbial cellulases. Enzym Microb Technol 46:541–549
Delabona P, Pirota R, Codima C, Tremacoldi C, Rodrigues A, Farinas C (2012) Using Amazon forest fungi and agricultural residues as a strategy to produce cellulolytic enzymes. Biomass Bioenergy 37:243–250
Delabona P, Farinas C, da Silva M, Azzoni S, Pradella J (2012) Use of a new Trichoderma harzianum strain isolated from the Amazon rainforest with pretreated sugar cane bagasse for on-site cellulase production. Bioresour Technol 107:517–521
Mandels M, Sternberg D (1976) Recent advances in cellulase technology. J Ferment Technol 54:267–286
Pirota R, Tonelotto M, Delabona PD, Fonseca RF, Paixao DAA, Baleeiro FCF, Neto VB, Farinas CS (2013) Enhancing xylanases production by a new Amazon Forest strain of Aspergillus oryzae using solid-state fermentation under controlled operation conditions. Ind Crop Prod 45:465–471
Bailey MJ, Poutanen K (1989) Production of xylanolytic enzymes by strains of Aspergillus. Appl Microbiol Biotechnol 30:5–10
Miller G (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Pirota R, Miotto LS, Delabona PS, Farinas CS (2013) Improving the extraction conditions of endoglucanase produced by Aspergillus niger under solid-state fermentation. Braz J Chem Eng 30:117–123
Ximenes E, Kim Y, Mosier N, Dien B, Ladisch M (2010) Inhibition of cellulases by phenols. Enzym Microb Technol 46:170–176
Soccol CR, Vandenberghe LPD, Medeiros ABP, Karp SG, Buckeridge M, Ramos LP, Pitarelo AP, Ferreira-Leitao V, Gottschalk LMF, Ferrara MA, Bon EPD, de Moraes LMP, Araujo JD, Torres FAG (2010) Bioethanol from lignocelluloses: status and perspectives in Brazil. Bioresour Technol 101:4820–4825
Lynd L, Weimer P, van Zyl W, Pretorius I (2002) Microbial cellulose utilization: fundamentals and biotechnology. Microbiol Mol Biol Rev 66:506–577
Kim Y, Ximenes E, Mosier NS, Ladisch MR (2011) Soluble inhibitors/deactivators of cellulase enzymes from lignocellulosic biomass. Enzym Microb Technol 48:408–415
Ximenes E, Kim Y, Mosier N, Dien B, Ladisch M (2011) Deactivation of cellulases by phenols. Enzym Microb Technol 48:54–60
Hu H, van den Brink J, Gruben B, Wosten H, Gu J, de Vries R (2011) Improved enzyme production by co-cultivation of Aspergillus niger and Aspergillus oryzae and with other fungi. Int Biodeterior Biodegrad 65:248–252
Kobayashi T, Abe K, Asai K, Gomi K, Juvvadi P, Kato M, Kitamoto K, Takeuchi M, Machida M (2007) Genomics of Aspergillus oryzae. Biosci Biotechnol Biochem 71:646–670
Acknowledgments
The authors would like to thank Embrapa, CNPq, Capes, and FAPESP (all from Brazil) for their financial support. We also acknowledge Dr. Sonia Couri as the researcher responsible for the isolation and mutation studies of the A. niger 3T5B8 strain and are grateful to Dr. Sandra C. Pereira for the internal review of this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pirota, R.D.P.B., Delabona, P.S. & Farinas, C.S. Simplification of the Biomass to Ethanol Conversion Process by Using the Whole Medium of Filamentous Fungi Cultivated Under Solid-State Fermentation. Bioenerg. Res. 7, 744–752 (2014). https://doi.org/10.1007/s12155-013-9406-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-013-9406-4