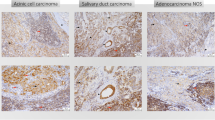
Abstract
Background
Recently, many studies have investigated the role of tumor immune microenvironment (TIME) in carcinogenesis, highlighting its relation to both tumor regression and progression. In particular, the “inflammatory system”, made of innate and adaptive immune cells, interacts with cancer cells and their surrounding stroma. In this setting, the aim of this review is to summarize the current literature regarding the TIME of major salivary gland carcinomas (MSGCs), with particular attention on the characteristics and prognostic role of tumor infiltrating lymphocytes (TILs), the mechanisms that lead to TILs exhaustion and the important additional immune infiltrating factors that help SGC progression or remission.
Methods
A comprehensive literature search was performed concerning published articles on the role of TIME in MSGCs.
Results
In this work we summarize the advancing knowledge on TIME in SGCs by demonstrating the key prognostic and/or predictive value of specific immune features.
Conclusion
From the analysis of the current ‘status of the art’ it clearly emerges a need for precise, unambiguous phenotyping of immune cell populations, as well as a more thorough understanding of the frequencies and interactions of multiple immune cell types inside the TIME and their spatial localization (intratumoral vs. stromal).


Similar content being viewed by others
Data Availability
Data is available upon reasonable request from the corresponding author.
Code Availability
None.
References
De Visser KE, Eichten A, Coussens LM (2006) Paradoxical roles of the immune system during cancer development. Nat Rev Cancer 6(1):24–37. https://doi.org/10.1038/nrc1782
Mantovani A, Allavena P, Sica A, Balkwill F (2008) Cancer-related inflammation. Nature 454(7203):436–444. https://doi.org/10.1038/nature07205
St Paul M, Ohashi PS (2020) The roles of CD8+ T cell subsets in antitumor immunity. Trends Cell Biol 30(9):695–704. https://doi.org/10.1016/j.tcb.2020.06.003
Pardoll DM, Topalian SL (1998) The role of CD4+ T cell responses in antitumor immunity. Curr Opin Immunol 10(5):588–594. https://doi.org/10.1016/s0952-7915(98)80228-8
Kurts C, Carbone FR, Barnden M et al (1997) CD4+ T cell help impairs CD8+ T cell deletion induced by cross-presentation of self-antigens and favors autoimmunity. J Exp Med 186(12):2057–2062. https://doi.org/10.1084/jem.186.12.2057
Sakaguchi S (2004) Naturally arising CD4+ regulatory t cells for immunologic self-tolerance and negative control of immune responses. Annu Rev Immunol 22:531–562. https://doi.org/10.1146/annurev.immunol.21.120601.141122
Shibuya TY, Nugyen N, McLaren CE et al (2002) Clinical significance of poor CD3 response in head and neck cancer. Clin Cancer Res 8(3):745–751
Russell S, Angell T, Lechner M et al (2013) Immune cell infiltration patterns and survival in head and neck squamous cell carcinoma. Head Neck Oncol 5(3):24
Näsman A, Romanitan M, Nordfors C et al (2012) Tumor infiltrating CD8+ and Foxp3+ lymphocytes correlate to clinical outcome and human papillomavirus (HPV) status in tonsillar cancer. PLoS ONE 7(6):e38711. https://doi.org/10.1371/journal.pone.0038711
Ward MJ, Thirdborough SM, Mellows T et al (2014) Tumour-infiltrating lymphocytes predict for outcome in HPV-positive oropharyngeal cancer. Br J Cancer 110(2):489–500. https://doi.org/10.1038/bjc.2013.639
Rodrigo JP, Sánchez-Canteli M, López F et al (2021) Tumor-infiltrating lymphocytes in the tumor microenvironment of laryngeal squamous cell carcinoma: systematic review and meta-analysis. Biomedicines 9(5):486. https://doi.org/10.3390/biomedicines9050486
Sun W, Li WJ, Fu QL et al (2015) Functionally distinct subsets of CD4+ regulatory T cells in patients with laryngeal squamous cell carcinoma are indicative of immune deregulation and disease progression. Oncol Rep 33(1):354–362. https://doi.org/10.3892/or.2014.3553
Skálová A, Hyrcza MD, Leivo I (2022) Update from the 5th edition of the World Health Organization Classification of head and neck tumors: salivary glands. Head Neck Pathol 16(1):40–53. https://doi.org/10.1007/s12105-022-01420-1
Sarradin V, Siegfried A, Uro-Coste E, Delord JP (2018) Classification de l’OMS 2017 des tumeurs de la tête et du cou : principales nouveautés et mise à jour des méthodes diagnostiques [WHO classification of head and neck tumours 2017: main novelties and update of diagnostic methods]. Bull Cancer 105(6):596–602. https://doi.org/10.1016/j.bulcan.2018.04.004
Young A, Okuyemi OT (2022) Malignant salivary gland tumors. StatPearls Publishing, Treasure Island
Carlson ER, McCoy JM (2017) Margins for benign salivary gland neoplasms of the head and neck. Oral Maxillofac Surg Clin N Am 29(3):325–340. https://doi.org/10.1016/j.coms.2017.03.009
Andreasen S, Bjørndal K, Agander TK, Wessel I, Homøe P (2016) Tumors of the sublingual gland: a national clinicopathologic study of 29 cases. Eur Arch Otorhinolaryngol 273(11):3847–3856. https://doi.org/10.1007/s00405-016-4000-y
Seethala RR (2011) Histologic grading and prognostic biomarkers in salivary gland carcinomas. Adv Anat Pathol 18(1):29–45. https://doi.org/10.1097/PAP.0b013e318202645a
Lima RA, Tavares MR, Dias FL et al (2005) Clinical prognostic factors in malignant parotid gland tumors. Otolaryngol Head Neck Surg 133(5):702–708. https://doi.org/10.1016/j.otohns.2005.08.001
Blankenstein T, Coulie PG, Gilboa E, Jaffee EM (2012) The determinants of tumour immunogenicity. Nat Rev Cancer 12(4):307–313. https://doi.org/10.1038/nrc3246
Sato F, Ono T, Kawahara A et al (2021) Prognostic value of tumor proportion score in salivary gland carcinoma. Laryngoscope 131(5):E1481–E1488. https://doi.org/10.1002/lary.29120
Linxweiler M, Kuo F, Katabi N et al (2020) The immune microenvironment and neoantigen landscape of aggressive salivary gland carcinomas differ by subtype. Clin Cancer Res 26(12):2859–2870. https://doi.org/10.1158/1078-0432.CCR-19-3758
Kesar N, Winkelmann R, Oppermann J et al (2020) Prognostic impact of CD8-positive tumour-infiltrating lymphocytes and PD-L1 expression in salivary gland cancer. Oral Oncol 111:104931. https://doi.org/10.1016/j.oraloncology.2020.104931
Witte HM, Gebauer N, Lappöhn D et al (2020) Prognostic impact of PD-L1 expression in malignant salivary gland tumors as assessed by established scoring criteria: Tumor Proportion Score (TPS), Combined Positivity Score (CPS), and Immune Cell (IC) Infiltrate. Cancers (Basel) 12(4):873. https://doi.org/10.3390/cancers12040873
Alame M, Cornillot E, Cacheux V et al (2020) The molecular landscape and microenvironment of salivary duct carcinoma reveal new therapeutic opportunities. Theranostics 10(10):4383–4394. https://doi.org/10.7150/thno.42986
Chatzopoulos K, Collins AR, Sotiriou S et al (2020) Increased ERBB2 gene copy numbers reveal a subset of salivary duct carcinomas with high densities of tumor infiltrating lymphocytes and PD-L1 expression. Head Neck Pathol 14(4):951–965. https://doi.org/10.1007/s12105-020-01163-x
Chan TA, Yarchoan M, Jaffee E et al (2019) Development of tumor mutation burden as an immunotherapy biomarker: utility for the oncology clinic. Ann Oncol 30(1):44–56. https://doi.org/10.1093/annonc/mdy495
Sridharan V, Gjini E, Liao X et al (2016) Immune profiling of adenoid cystic carcinoma: PD-L2 expression and associations with tumor-infiltrating lymphocytes. Cancer Immunol Res 4(8):679–687. https://doi.org/10.1158/2326-6066.CIR-16-0031
Chen W, Fung AS, McIntyre JB et al (2021) Assessment of tumour infiltrating lymphocytes and Pd-l1 expression in adenoid cystic carcinoma of the salivary gland. Clin Invest Med 44(1):38–41. https://doi.org/10.25011/cim.v44i1.35218
Mosconi C, de Arruda JAA, de Farias ACR et al (2019) Immune microenvironment and evasion mechanisms in adenoid cystic carcinomas of salivary glands. Oral Oncol 88:95–101. https://doi.org/10.1016/j.oraloncology.2018.11.028
Mosconi C, Oliveira GAQ, de Farias ACR et al (2020) Association of the tumor-infiltrating lymphocytes with recurrence in adenoid cystic carcinoma of salivary glands. Oral Surg Oral Med Oral Pathol Oral Radiol. https://doi.org/10.1016/j.oooo.2019.06.598
Nakano T, Takizawa K, Uezato A, Taguchi K, Toh S, Masuda M (2019) Prognostic value of programed death ligand-1 and ligand-2 co-expression in salivary gland carcinomas. Oral Oncol 90:30–37. https://doi.org/10.1016/j.oraloncology.2019.01.015
Barnes L, Eveson JW, Reichart P, Sidransky D (eds) (2005) World Health Organization classification of tumours. Pathology and genetics of head and neck tumours. IARC Press, Lyon
Hiss S, Eckstein M, Segschneider P et al (2021) Tumour-infiltrating lymphocytes (TILs) and PD-L1 expression correlate with lymph node metastasis, high-grade transformation and shorter metastasis-free survival in patients with acinic cell carcinoma (AciCC) of the salivary glands. Cancers (Basel) 13(5):965. https://doi.org/10.3390/cancers13050965
Vander Poorten V, Triantafyllou A, Thompson LD et al (2016) Salivary acinic cell carcinoma: reappraisal and update. Eur Arch Otorhinolaryngol 273(11):3511–3531. https://doi.org/10.1007/s00405-015-3855-7
Schwarz S, Zenk J, Müller M et al (2012) The many faces of acinic cell carcinomas of the salivary glands: a study of 40 cases relating histological and immunohistological subtypes to clinical parameters and prognosis. Histopathology 61(3):395–408. https://doi.org/10.1111/j.1365-2559.2012.04233.x
Dublin JC, Oliver JR, Tam MM et al (2022) Nodal metastases in pediatric and adult acinic cell carcinoma of the major salivary glands. Otolaryngol Head Neck Surg 167(6):941–951. https://doi.org/10.1177/01945998221083094
Dutsch-Wicherek M, Lazar A, Tomaszewska R (2010) The involvement of RCAS1 in creating a suppressive tumor microenvironment in patients with salivary gland adenocarcinoma. Cancer Microenviron 4(1):13–21. https://doi.org/10.1007/s12307-010-0051-6
Spiro RH, Huvos AG, Strong EW (1982) Adenocarcinoma of salivary origin. Clinicopathologic study of 204 patients. Am J Surg 144(4):423–431. https://doi.org/10.1016/0002-9610(82)90416-0
Mosconi C, Arantes DAC, Gonçalves AS et al (2017) Immunohistochemical investigations on the expression of programmed cell death ligand 1, human leukocyte antigens G and E, and granzyme B in intraoral mucoepidermoid carcinoma. Arch Oral Biol 83:55–62. https://doi.org/10.1016/j.archoralbio.2017.07.004
Zhang N, Bevan MJ (2011) CD8(+) T cells: foot soldiers of the immune system. Immunity 35(2):161–168. https://doi.org/10.1016/j.immuni.2011.07.010
Greten FR, Grivennikov SI (2019) Inflammation and cancer: triggers, mechanisms, and consequences. Immunity 51(1):27–41. https://doi.org/10.1016/j.immuni.2019.06.025
Speight PM, Barrett AW (2002) Salivary gland tumours. Oral Dis 8(5):229–240. https://doi.org/10.1034/j.1601-0825.2002.02870.x
Chang H, Kim JS, Choi YJ et al (2017) Overexpression of PD-L2 is associated with shorter relapse-free survival in patients with malignant salivary gland tumors. Onco Targets Ther 10:2983–2992. https://doi.org/10.2147/OTT.S134589
Lemound J, Schenk M, Keller G et al (2016) Cytogenetic and immunohistochemical biomarker profiling of therapy-relevant factors in salivary gland carcinomas. J Oral Pathol Med 45(9):655–663. https://doi.org/10.1111/jop.12429
Haghshenas MR, Khademi B, Faghih Z, Ghaderi A, Erfani N (2015) Immune regulatory cells and IL17-producing lymphocytes in patients with benign and malignant salivary gland tumors. Immunol Lett 164(2):109–116. https://doi.org/10.1016/j.imlet.2015.02.008
Cosmi L, Maggi L, Santarlasci V, Liotta F, Annunziato F (2014) T helper cells plasticity in inflammation. Cytometry A 85(1):36–42. https://doi.org/10.1002/cyto.a.22348
Mojic M, Takeda K, Hayakawa Y (2017) The dark side of IFN-γ: its role in promoting cancer immunoevasion. Int J Mol Sci 19(1):89. https://doi.org/10.3390/ijms19010089
Haghshenas MR, Khademi B, Ashraf MJ, Ghaderi A, Erfani N (2016) Helper and cytotoxic T-cell subsets (Th1, Th2, Tc1, and Tc2) in benign and malignant salivary gland tumors. Oral Dis 22(6):566–572. https://doi.org/10.1111/odi.12496
Schuster M, Plaza-Sirvent C, Visekruna A, Huehn J, Schmitz I (2019) Generation of Foxp3+CD25- regulatory T-cell precursors requires c-Rel and IκBNS. Front Immunol 10:1583. https://doi.org/10.3389/fimmu.2019.01583
Schvartsman G, Bell D, Rubin ML et al (2021) The tumor immune contexture of salivary duct carcinoma. Head Neck 43(4):1213–1219. https://doi.org/10.1002/hed.26587
Vivier E, Tomasello E, Baratin M, Walzer T, Ugolini S (2008) Functions of natural killer cells. Nat Immunol 9(5):503–510. https://doi.org/10.1038/ni1582
Hellquist HB, Karlsson MG, Viale G et al (1996) Bcl-2 immunoreactivity in salivary gland neoplasms is unrelated to the expression of mRNA for natural killer cell stimulatory cytokines interleukin (IL)-2 and IL-12. Virchows Arch 429(2–3):149–158. https://doi.org/10.1007/BF00192437
Dieu-Nosjean MC, Giraldo NA, Kaplon H, Germain C, Fridman WH, Sautès-Fridman C (2016) Tertiary lymphoid structures, drivers of the anti-tumor responses in human cancers. Immunol Rev 271(1):260–275. https://doi.org/10.1111/imr.12405
Gavrielatou N, Vathiotis I, Economopoulou P, Psyrri A (2021) The role of B cells in head and neck cancer. Cancers (Basel) 13(21):5383. https://doi.org/10.3390/cancers13215383
Nelson BH (2010) CD20+ B cells: the other tumor-infiltrating lymphocytes. J Immunol 185(9):4977–4982. https://doi.org/10.4049/jimmunol.1001323
Zhou X, Su YX, Lao XM, Liang YJ, Liao GQ (2016) CD19(+)IL-10(+) regulatory B cells affect survival of tongue squamous cell carcinoma patients and induce resting CD4(+) T cells to CD4(+)Foxp3(+) regulatory T cells. Oral Oncol 53:27–35. https://doi.org/10.1016/j.oraloncology.2015.11.003
Blank CU, Haining WN, Held W et al (2019) Defining “T cell exhaustion.” Nat Rev Immunol 19(11):665–674. https://doi.org/10.1038/s41577-019-0221-9
Wherry EJ (2011) T cell exhaustion. Nat Immunol 12(6):492–499. https://doi.org/10.1038/ni.2035
Freeman GJ, Long AJ, Iwai Y et al (2000) Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med 192(7):1027–1034. https://doi.org/10.1084/jem.192.7.1027
Fang Q, Wu Y, Du W, Zhang X, Chen D (2021) Incidence and prognostic significance of PD-L1 expression in high-grade salivary gland carcinoma [published correction appears in Front Oncol. 2021 Dec 30;11:836903]. Front Oncol 11:701181. https://doi.org/10.3389/fonc.2021.701181
Vital D, Ikenberg K, Moch H, Rössle M, Huber GF (2019) The expression of PD-L1 in salivary gland carcinomas. Sci Rep 9(1):12724. https://doi.org/10.1038/s41598-019-49215-9
Mukaigawa T, Hayashi R, Hashimoto K, Ugumori T, Hato N, Fujii S (2016) Programmed death ligand-1 expression is associated with poor disease free survival in salivary gland carcinomas. J Surg Oncol 114(1):36–43. https://doi.org/10.1002/jso.24266
Kuchar M, Strizova Z, Capkova L et al (2021) The periphery of salivary gland carcinoma tumors reveals a PD-L1/PD-1 biomarker niche for the evaluation of disease severity and tumor-immune system interplay. Biomedicines 9(2):97. https://doi.org/10.3390/biomedicines9020097
Guazzo E, Cooper C, Wilkinson L et al (2021) Therapeutic implications of immune-profiling and EGFR expression in salivary gland carcinoma. Head Neck 43(3):768–777. https://doi.org/10.1002/hed.26529
Arolt C, Meyer M, Ruesseler V et al (2020) Lymphocyte activation gene 3 (LAG3) protein expression on tumor-infiltrating lymphocytes in aggressive and TP53-mutated salivary gland carcinomas. Cancer Immunol Immunother 69(7):1363–1373. https://doi.org/10.1007/s00262-020-02551-6
Nguyen LT, Ohashi PS (2015) Clinical blockade of PD1 and LAG3–potential mechanisms of action. Nat Rev Immunol 15(1):45–56. https://doi.org/10.1038/nri3790
Camisaschi C, Casati C, Rini F et al (2010) LAG-3 expression defines a subset of CD4(+)CD25(high)Foxp3(+) regulatory T cells that are expanded at tumor sites. J Immunol 184(11):6545–6551. https://doi.org/10.4049/jimmunol.0903879
Even C, Delord JP, Price KA et al (2022) Evaluation of pembrolizumab monotherapy in patients with previously treated advanced salivary gland carcinoma in the phase 2 KEYNOTE-158 study. Eur J Cancer 171:259–268. https://doi.org/10.1016/j.ejca.2022.05.007
Mahmood U, Bang A, Chen YH et al (2021) A randomized phase 2 study of pembrolizumab with or without radiation in patients with recurrent or metastatic adenoid cystic carcinoma. Int J Radiat Oncol Biol Phys 109(1):134–144. https://doi.org/10.1016/j.ijrobp.2020.08.018
Rodriguez CP, Wu QV, Voutsinas J et al (2020) A phase II trial of pembrolizumab and vorinostat in recurrent metastatic head and neck squamous cell carcinomas and salivary gland cancer. Clin Cancer Res 26(4):837–845. https://doi.org/10.1158/1078-0432.CCR-19-2214
Ross JS, Gay LM, Wang K et al (2017) Comprehensive genomic profiles of metastatic and relapsed salivary gland carcinomas are associated with tumor type and reveal new routes to targeted therapies. Ann Oncol 28(10):2539–2546. https://doi.org/10.1093/annonc/mdx399
Fayette J, Even C, Digue L et al (2019) NISCAHN: a phase II, multi-center nonrandomized trial aiming at evaluating nivolumab (N) in two cohorts of patients (pts) with recurrent/metastatic (R/M) salivary gland carcinoma of the head and neck (SGCHN), on behalf of the Unicancer Head & Neck Group. J Clin Oncol 37:6083. https://doi.org/10.1200/JCO.2019.37.15_suppl.6083
Pittet MJ, Michielin O, Migliorini D (2022) Clinical relevance of tumour-associated macrophages [published correction appears in Nat Rev Clin Oncol. 2022 Apr 6]. Nat Rev Clin Oncol 19(6):402–421. https://doi.org/10.1038/s41571-022-00620-6
Mantovani A, Marchesi F, Malesci A, Laghi L, Allavena P (2017) Tumour-associated macrophages as treatment targets in oncology. Nat Rev Clin Oncol 14(7):399–416. https://doi.org/10.1038/nrclinonc.2016.217
Mantovani A, Sozzani S, Locati M, Allavena P, Sica A (2002) Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol 23(11):549–555. https://doi.org/10.1016/s1471-4906(02)02302-5
Qian BZ, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141(1):39–51. https://doi.org/10.1016/j.cell.2010.03.014
Bingle L, Brown NJ, Lewis CE (2002) The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol 196(3):254–265. https://doi.org/10.1002/path.1027
Donadon M, Torzilli G, Cortese N et al (2020) Macrophage morphology correlates with single-cell diversity and prognosis in colorectal liver metastasis. J Exp Med 217(11):e20191847. https://doi.org/10.1084/jem.20191847
Barros MH, Hauck F, Dreyer JH, Kempkes B, Niedobitek G (2013) Macrophage polarisation: an immunohistochemical approach for identifying M1 and M2 macrophages. PLoS ONE 8(11):e80908. https://doi.org/10.1371/journal.pone.0080908
Shieh YS, Hung YJ, Hsieh CB, Chen JS, Chou KC, Liu SY (2009) Tumor-associated macrophage correlated with angiogenesis and progression of mucoepidermoid carcinoma of salivary glands. Ann Surg Oncol 16(3):751–760. https://doi.org/10.1245/s10434-008-0259-6
Yang Z, Li H, Wang W et al (2019) CCL2/CCR2 Axis promotes the progression of salivary adenoid cystic carcinoma via recruiting and reprogramming the tumor-associated macrophages. Front Oncol 9:231. https://doi.org/10.3389/fonc.2019.00231
Li C, Chen Q, Tian Z et al (2019) Expression of MIF, Beclin1, and LC3 in human salivary gland adenoid cystic carcinoma and its prognostic value. Medicine (Baltimore) 98(20):e15402. https://doi.org/10.1097/MD.0000000000015402
Liu H, Chen G, Zhang W et al (2013) Overexpression of macrophage migration inhibitory factor in adenoid cystic carcinoma: correlation with enhanced metastatic potential. J Cancer Res Clin Oncol 139(2):287–295. https://doi.org/10.1007/s00432-012-1330-z
Acknowledgements
None
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to Participate
For this type of study informed consent is not required.
Consent for Publication
For this type of study informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Veneroni, M.V., Festa, B.M., Costantino, A. et al. Prognostic Impact of Tumor Immune Microenvironment and Its Predictive Role in Salivary Gland Cancer. Head and Neck Pathol 17, 515–527 (2023). https://doi.org/10.1007/s12105-023-01528-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-023-01528-y