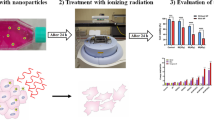
Abstract
Objectives
To investigate the effects of Au@Fe2O3 core–shell nanoparticle (NP), with and without conjugation to folic acid (FA) as a targeting ligand, on radiosensitization of both cancer and healthy cells.
Methods
Au@Fe2O3 NPs were first synthesized, then modified with FA, and finally characterized. Radiation dose enhancement studies were performed on KB cancer cells and L929 healthy cells. NPs at the concentration of 20 µg/ml were first incubated with both cell lines and then different doses of 6 MV X-ray radiation were examined. The end effects were evaluated via MTT assay and flow cytometry using AnnexinV/PI kit.
Results
It was indicated that viability of KB cells has a much lower rate than L929 cells when the cells were treated by {(FA-Au@Fe2O3) + (X-ray)} regimen. Cell viability was even decreased significantly when X-ray dose increased. Moreover, flow cytometry studies revealed that FA-targeted NPs induced higher level of apoptosis for KB cancer cells than L929 healthy cells.
Conclusion
Our findings provide a new perspective on high ability of the synthesized FA-targeted Au@Fe2O3 NPs which may be considered as an efficient radiosensitizer in the process of targeted radiation therapy of cancer.







Similar content being viewed by others
References
Lin J, Chen X, Huang P. Graphene-based nanomaterials for bioimaging. Adv Drug Deliv Rev. 2016;105:242–54.
Ryu JH, Lee S, Son S, Kim SH, Leary JF, Choi K, et al. Theranostic nanoparticles for future personalized medicine. J Control Release. 2014;190:477–84.
Wang C, Jiang Y, Li X, Hu L. Thioglucose-bound gold nanoparticles increase the radiosensitivity of a triple-negative breast cancer cell line (MDA-MB-231). Breast Cancer. 2015;22(4):413–20.
Liu J, Yang Y, Zhu W, Yi X, Dong Z, Xu X, et al. Nanoscale metal − organic frameworks for combined photodynamic & radiation therapy in cancer treatment. Biomaterials. 2016;97:1–9.
Deng Y, Li E, Cheng X, Zhu J, Lu S, Ge C, et al. Facile preparation of hybrid core–shell nanorods for photothermal and radiation combined therapy. Nanoscale. 2016;8(7):3895–9.
Kumar A, Zhang X, Liang X-J. Gold nanoparticles: emerging paradigm for targeted drug delivery system. Biotechnol Adv. 2013;31(5):593–606.
Rana S, Bajaj A, Mout R, Rotello VM. Monolayer coated gold nanoparticles for delivery applications. Adv Drug Deliv Rev. 2012;64(2):200–16.
Al Zaki A, Cormode D, Tsourkas A, Dorsey JF. Increasing the therapeutic efficacy of radiotherapy using nanoparticles. Increasing the therapeutic ratio of radiotherapy. Berlin: Springer; 2017. p. 241–65.
Her S, Jaffray DA, Allen C. Gold nanoparticles for applications in cancer radiotherapy: mechanisms and recent advancements. Advanced drug delivery reviews. 2017;109:84–101.
Shakeri-Zadeh A, Shiran M-B, Khoee S, Sharifi AM, Ghaznavi H, Khoei S. A new magnetic nanocapsule containing 5-fluorouracil: in vivo drug release, anti-tumor, and pro-apoptotic effects on CT26 cells allograft model. J Biomater Appl. 2014;29(4):548–56.
Lucarini M, Franchi P, Pedulli GF, Pengo P, Scrimin P, Pasquato L. EPR study of dialkyl nitroxides as probes to investigate the exchange of solutes between the ligand shell of monolayers of protected gold nanoparticles and aqueous solutions. J Am Chem Soc. 2004;126(30):9326–9.
Boisselier E, Astruc D. Gold nanoparticles in nanomedicine: preparations, imaging, diagnostics, therapies and toxicity. Chem Soc Rev. 2009;38(6):1759–82.
Beik J, Jafariyan M, Montazerabadi A, Ghadimi-Daresajini A, Tarighi P, Mahmoudabadi A, et al. The benefits of folic acid-modified gold nanoparticles in CT-based molecular imaging: radiation dose reduction and image contrast enhancement. Artif Cells Nanomed Biotechnol. 2017. https://doi.org/10.1080/21691401.2017.1408019.
Ghaznavi H, Hosseini-Nami S, Kamrava SK, Irajirad R, Maleki S, Shakeri-Zadeh A, et al. Folic acid conjugated PEG coated gold–iron oxide core–shell nanocomplex as a potential agent for targeted photothermal therapy of cancer. Artif Cells Nanomed Biotechnol. 2017. https://doi.org/10.1080/21691401.2017.1384384.
Zeinizade E, Tabei M, Shakeri-Zadeh A, Ghaznavi H, Attaran N, Komeili A, et al. Selective apoptosis induction in cancer cells using folate-conjugated gold nanoparticles and controlling the laser irradiation conditions. Artif Cells Nanomed Biotechnol. 2018. https://doi.org/10.1080/21691401.2018.1443116
Zhang X, Xing JZ, Chen J, Ko L, Amanie J, Gulavita S, et al. Enhanced radiation sensitivity in prostate cancer by gold-nanoparticles. Clin Invest Med. 2008;31(3):160–7.
Kong T, Zeng J, Wang X, Yang X, Yang J, McQuarrie S, et al. Enhancement of radiation cytotoxicity in Breast-cancer cells by localized attachment of gold nanoparticles. Small. 2008;4(9):1537–43.
Zhang X-D, Wu D, Shen X, Chen J, Sun Y-M, Liu P-X, et al. Size-dependent radiosensitization of PEG-coated gold nanoparticles for cancer radiation therapy. Biomaterials. 2012;33(27):6408–19.
Hainfeld JF, Smilowitz HM, O’Connor MJ, Dilmanian FA, Slatkin DN. Gold nanoparticle imaging and radiotherapy of brain tumors in mice. Nanomedicine. 2013;8(10):1601–9.
Tsiamas P, Liu B, Cifter F, Ngwa WF, Berbeco RI, Kappas C, et al. Impact of beam quality on megavoltage radiotherapy treatment techniques utilizing gold nanoparticles for dose enhancement. Phys Med Biol. 2013;58(3):451.
Eyvazzadeh N, Shakeri-Zadeh A, Fekrazad R, Amini E, Ghaznavi H, Kamrava SK. Gold-coated magnetic nanoparticle as a nanotheranostic agent for magnetic resonance imaging and photothermal therapy of cancer. Lasers Med Sci. 2017;32(7):1469–77.
Wang L, Park H-Y, Stephanie I, Lim I, Schadt MJ, Mott D, et al. Core@ shell nanomaterials: gold-coated magnetic oxide nanoparticles. J Mater Chem. 2008;18(23):2629–35.
Beik J, Khademi S, Attaran N, Sarkar S, Shakeri-Zadeh A, Ghaznavi H, et al. A nanotechnology-based strategy to increase the efficiency of cancer diagnosis and therapy: folate-conjugated gold nanoparticles. Curr Med Chem. 2017;24(39):4399–416.
Samadian H, Hosseini-Nami S, Kamrava SK, Ghaznavi H, Shakeri-Zadeh A. Folate-conjugated gold nanoparticle as a new nanoplatform for targeted cancer therapy. J Cancer Res Clin Oncol. 2016;142(11):2217–29.
Mirrahimi M, Hosseini V, Kamrava SK, Attaran N, Beik J, Kooranifar S, et al. Selective heat generation in cancer cells using a combination of 808 nm laser irradiation and the folate-conjugated Fe2O3@ Au nanocomplex. Artificial cells, nanomedicine, and biotechnology. 2017:1–13.
Shakeri-Zadeh A, Khoee S, Shiran M-B, Sharifi AM, Khoei S. Synergistic effects of magnetic drug targeting using a newly developed nanocapsule and tumor irradiation by ultrasound on CT26 tumors in BALB/c mice. J Mater Chem B. 2015;3(9):1879–87.
Sivakumar B, Aswathy RG, Sreejith R, Nagaoka Y, Iwai S, Suzuki M, et al. Bacterial exopolysaccharide based magnetic nanoparticles: a versatile nanotool for cancer cell imaging, targeted drug delivery and synergistic effect of drug and hyperthermia mediated cancer therapy. J Biomed Nanotechnol. 2014;10(6):885–99.
Banu H, Stanley B, Faheem S, Seenivasan R, Premkumar K, Vasanthakumar G. Thermal chemosensitization of breast cancer cells to cyclophosphamide treatment using folate receptor targeted gold nanoparticles. Plasmonics. 2014;9(6):1341–9.
Beik J, Abed Z, Ghoreishi FS, Hosseini-Nami S, Mehrzadi S, Shakeri-Zadeh A, et al. Nanotechnology in hyperthermia cancer therapy: from fundamental principles to advanced applications. J Control Release. 2016;235:205–21.
Chithrani BD, Ghazani AA, Chan WC. Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett. 2006;6(4):662–8.
Hu R, Zheng M, Wu J, Li C, Shen D, Yang D, et al. Core-Shell magnetic gold nanoparticles for magnetic field-enhanced radio-photothermal therapy in cervical cancer. Nanomaterials. 2017;7(5):111.
Peng X-H, Qian X, Mao H, Wang AY. Targeted magnetic iron oxide nanoparticles for tumor imaging and therapy. Int J Nanomed. 2008;3(3):311.
Chomoucka J, Drbohlavova J, Huska D, Adam V, Kizek R, Hubalek J. Magnetic nanoparticles and targeted drug delivering. Pharmacol Res. 2010;62(2):144–9.
Wolfe T, Chatterjee D, Lee J, Grant JD, Bhattarai S, Tailor R, et al. Targeted gold nanoparticles enhance sensitization of prostate tumors to megavoltage radiation therapy in vivo. Nanomed Nanotechnol Biol Med. 2015;11(5):1277–83.
Khoei S, Mahdavi SR, Fakhimikabir H, Shakeri-Zadeh A, Hashemian A. The role of iron oxide nanoparticles in the radiosensitization of human prostate carcinoma cell line DU145 at megavoltage radiation energies. Int J Radiat Biol. 2014;90(5):351–6.
Khoshgard K, Hashemi B, Arbabi A, Rasaee MJ, Soleimani M. Radiosensitization effect of folate-conjugated gold nanoparticles on HeLa cancer cells under orthovoltage superficial radiotherapy techniques. Phys Med Biol. 2014;59(9):2249.
Gao B, Shen L, He K-W, Xiao W-H. GNRs@ SiO2-FA in combination with radiotherapy induces the apoptosis of HepG2 cells by modulating the expression of apoptosis-related proteins. Int J Mol Med. 2015;36(5):1282–90.
Neshastehriz A, Tabei M, Maleki S, Eynali S, Shakeri-Zadeh A. Photothermal therapy using folate conjugated gold nanoparticles enhances the effects of 6MV X-ray on mouth epidermal carcinoma cells. J Photochem Photobiol, B. 2017;172:52–60.
Wyllie AH, Kerr JR, Currie A. Cell death: the significance of apoptosis. Int Rev Cytol. 1980;68:251–306.
Dewey WC, Ling CC, Meyn RE. Radiation-induced apoptosis: relevance to radiotherapy. Int J Radiat Oncol Biol Phys. 1995;33(4):781–96.
Cohen-Jonathan E, Bernhard EJ, McKenna WG. How does radiation kill cells? Curr Opin Chem Biol. 1999;3(1):77–83.
Chiou S-K, Rao L, White E. Bcl-2 blocks p53-dependent apoptosis. Mol Cell Biol. 1994;14(4):2556–63.
Igney FH, Krammer PH. Death and anti-death: tumour resistance to apoptosis. Nat Rev Cancer. 2002;2(4):277–88.
Li P, Y-w S, Li B-x X, W-c SZ-l, Zhou C, et al. Photo-thermal effect enhances the efficiency of radiotherapy using Arg-Gly-Asp peptides-conjugated gold nanorods that target αvβ3 in melanoma cancer cells. J Nanobiotechnol. 2015;13(1):52.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Research involving human participants
This research did not involve human participants and/or animals.
Informed consent
For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Mirrahimi, M., Hosseini, V., Shakeri-Zadeh, A. et al. Modulation of cancer cells’ radiation response in the presence of folate conjugated Au@Fe2O3 nanocomplex as a targeted radiosensitizer. Clin Transl Oncol 21, 479–488 (2019). https://doi.org/10.1007/s12094-018-1947-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-018-1947-8