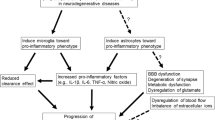
Abstract
Microglia are the main effectors in the inflammatory process of the central nervous system. As the first line of defense, microglia play an important role in the inflammatory reaction. When there is pathogen invasion or cell debris, microglia will be activated rapidly and remove it, while releasing the inflammatory cytokines to mediate inflammatory reaction. Activated microglia were found surrounding lesions of various neurodegenerative diseases, such as Alzheimer’s disease, Parkinson’s disease, muscular amyotrophic lateral sclerosis, and multiple sclerosis. Microglia, the effectors of neuronal degeneration and necrosis, are involved in the removal of necrotic neurons. But over activated microglia may accelerate the process of some neurodegenerative diseases. Activated microglia can release cytotoxic factor and cytokines. Some of them may cause further damage to neuron, and some of them can regulate inflammatory cells to gather to the lesion. Microglia-mediated inflammation was considered to be the possible mechanism for the occurrence or deterioration of neurodegenerative diseases. Therefore, inhibiting the activity of microglia appropriately may be an effective way for the treatment of neurodegenerative diseases.
Similar content being viewed by others
References
Streit WJ (2002) Microglia as neuroprotective, immunocompetent cells of the CNS. Glia 40(2):133–139
Cao L, Su Z, Zhou Q, Lv B, Liu X, Jiao L, Li Z, Zhu Y et al (2006) Glial cell line-derived neurotrophic factor promotes olfactory ensheathing cells migration. Glia 54(6):536–544
Park S, Lee KS, Lee YJ, Shin HA, Cho HY, Wang KC, Yong SK, Lee HT et al (2004) Generation of dopaminergic neurons in vitro from human embryonic stem cells treated with neurotrophic factors. Neurosci Lett 359(1–2):99–103
Stadelmann C, Kerschensteiner M, Misgeld T, Brück W, Hohlfeld R, Lassmann H (2002) BDNF and gp145trkB in multiple sclerosis brain lesions: neuroprotective interactions between immune and neuronal cells? Brain J Neurol 125(1):75–85
Wilms H, Rosenstiel P, Sievers J, Deuschl G, Zecca L, Lucius R (2003) Activation of microglia by human neuromelanin is NF-κB dependent and involves p38 mitogen-activated protein kinase: implications for Parkinson’s disease. FASEB J 17(3):500–502
Ginhoux F, Greter M, Leboeuf M, Nandi S, See P, Gokhan S, Mehler M, Conway S et al (2010) Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330(6005):841–845
Greter M, Merad M (2013) Regulation of microglia development and homeostasis. Glia 61(1):121–127
Davies LC, Jenkins SJ, Allen JE, Taylor PR (2013) Tissue-resident macrophages. Nat Immunol 14(10):986–995
Mizutani M, Pino PA, Saederup N, Charo IF, Ransohoff RM, Cardona AE (2012) The fractalkine receptor but not CCR2 is present on microglia from embryonic development throughout adulthood 1. J Immunol 188(1):29–36
Magni P, Ruscica M, Dozio E, Rizzi E, Beretta G, Maffei FR (2012) Parthenolide inhibits the LPS-induced secretion of IL-6 and TNF-α and NF-κB nuclear translocation in BV-2 microglia. Phytother Res 26(9):1405–1409
Hao YT, Chen CH, Yang XS, Zhang GB (2007) Changes in activation and secretion of microglia after electromagnetic field radiation. J Clin Rehabil Tissue Eng Res 11:9816–9819
Yang XS, Hao YT, He GL, Chen CH, Wang Y, Zhang GB, Yu ZP (2010) Relationship between activation of microglia and Jaks phosphorylation induced by microwave irradiation. Chin J Ind Hyg Occup Dis 28(6):401–404
Glass C (2010) Mechanisms underlying inflammation in neurodegeneration. Cell 140(6):918–934
Fuhrmann M, Bittner T, Jung CK, Burgold S, Page RM, Mitteregger G, Haass C, Laferla FM et al (2010) Microglial Cx3cr1 knockout prevents neuron loss in a mouse model of Alzheimer’s disease. Nat Neurosci 13(4):411–413
Hirsch EC, Hunot S (2009) Neuroinflammation in Parkinson’s disease: a target for neuroprotection? Lancet Neurol 8(4):382–397
Zhang Y, Fan Y, Wang M, Wang D, Li X (2013) Atorvastatin attenuates the production of IL-1β, IL-6, and TNF-α in the hippocampus of an amyloid β1-42-induced rat model of Alzheimer’s disease. Clin Interv Aging 8:103–110
Kocur M, Schneider R, Pulm AK, Bauer J, Kropp S, Gliem M, Ingwersen J, Goebels N et al (2015) IFNbeta secreted by microglia mediates clearance of myelin debris in CNS autoimmunity. Acta Neuropathol Commun 3(1):1–16
Gebicke-Haerter PJ, Spleiss O, Ren LQ, Li H, Dichmann S, Norgauer J, Boddeke HW (2001) Microglial chemokines and chemokine receptors. Prog Brain Res 132:525–532
Jander S, Schroeter M, Fischer J, Stoll G (2000) Differential regulation of microglial keratan sulfate immunoreactivity by proinflammatory cytokines and colony-stimulating factors. Glia 30(4):401–410
Mckhann G, Drachman D, Folstein M, Katzman R, Price D, Em S (1984) Clinical diagnosis of Alzheimer’s disease: report of the NINCDS–ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s disease. Neurology 34(7):939–944
Grutzendler J, Morris J (2006) Cholinesterase inhibitors for Alzheimer’s disease. Drugs 61(1):41–45
Sims NR, Smith CCT, Davison AN, Bowen DM, Flack RHA, Snowden JS, Neary D (1980) Glucose metabolism and acetylcholine synthesis in relation to neuronal activity in Alzheimer’s disease. Lancet 315(8164):333–336
Xu B, Wu X (2004) Advances in molecular biology and clinical study of amyloid precursor protein for Alzheimer’s disease. Acta Acad Med Sin 26(2):201–209
Lue LF, Walker DG, Rogers J (2001) Modeling microglial activation in Alzheimer’s disease with human postmortem microglial cultures. Neurobiol Aging 22(6):945–956
Benoit M, Hernandez M, Dinh M, Benavente F, Vasquez O, Tenner A (2013) C1q-induced LRP1B and GPR6 proteins expressed early in Alzheimer disease mouse models, are essential for the C1q-mediated protection against amyloid-β neurotoxicity. J Biol Chem 288(1):654–665
Fuhrmann M, Bittner T, Jung C, Burgold S, Mitteregger G, Kretzschmar H, Laferla FM, Herms J (2009) Microglia mediated and fractalkine receptor dependent neuron loss in a mouse model of Alzheimer’s disease. Alzheimers Dement 5(4):82
Pong WW, Higer SB, Gianino SM, Emnett RJ, Gutmann DH (2013) Reduced microglial CX3CR1 expression delays neurofibromatosis-1 glioma formation. Ann Neurol 73(2):303–308
He X, Suo A, Xu Y, Zhang J, Li W (2009) Effects of Aβ_(1-42)-induced microglial inflammatory supernatant on rat neuronal apoptosis and expression of Caspase-3 and PARP. J Zhengzhou Univ 44:1191–1193
Wilkinson K, Khoury JE (2012) Microglial scavenger receptors and their roles in the pathogenesis of Alzheimer’s disease. Int J Alzheimers Dis 2012:489456
Marchi N, Granata T, Ghosh C, Janigro D (2012) Blood-brain barrier dysfunction and epilepsy: pathophysiologic role and therapeutic approaches. Epilepsia 53(11):1877–1886
Chiang Y, Shih R, Chiu P, Hung K (2009) Hyaluronic acid inhibits the glial scar formation after brain damage with tissue loss in rats. Surg Neurol 72(2):50–54
Hamill CE, Goldshmidt A, Nicole O, Mckeon RJ, Brat DJ, Sf T (2005) Special lecture: glial reactivity after damage: implications for scar formation and neuronal recovery. Clin Neurosurg 52:29–44
Lee J, Auyeung WW, Mattson MP (2003) Interactive effects of excitotoxic injury and dietary restriction on microgliosis and neurogenesis in the hippocampus of adult mice. Neruomol Med 4(3):179–195
Choi J, Nordli DR Jr, Alden TD, DiPatri A Jr, Laux L, Kelley K, Rosenow J, Schuele SU et al (2009) Cellular injury and neuroinflammation in children with chronic intractable epilepsy. J Neuroinflammation 6(13):38
Liang J, Takeuchi H, Jin S, Noda M, Li H, Doi Y, Kawanokuchi J, Sonobe Y et al (2010) Glutamate induces neurotrophic factor production from microglia via protein kinase C pathway. Brain Res 1322(4):8–23
Dambach H, Hinkerohe D, Prochnow N, Stienen MN, Moinfar Z, Haase CG, Hufnagel A, Faustmann PM (2014) Glia and epilepsy: experimental investigation of antiepileptic drugs in an astroglia/microglia co-culture model of inflammation. Epilepsia 55(1):184–192
Tsirka MSE (2011) A functional role for microglia in epilepsy. Intech
Compston A, Coles A (2002) Multiple sclerosis. Lancet 359(9313):1221–1231
Sharrack B, Hughes RA (1996) Clinical scales for multiple sclerosis. J Neurol Sci 135(1):1–9
Elliot MF, Michael KR, Cedric SR (2006) Multiple sclerosis-the plaque and its pathogenesis. N Engl J Med 354(9):943–955
Gold R, Linington C, Lassmann H (2006) Understanding pathogenesis and therapy of multiple sclerosis via animal models: 70 years of merits and culprits in experimental autoimmune encephalomyelitis research. Brain 129(4):1953–1971
Oksenberg JR (2008) The genetics of multiple sclerosis: SNPs to pathways to pathogenesis. Nat Rev Genet 9(7):516–526
Bsibsi M, Peferoen L, Holtman I, Nacken P, Gerritsen W, Witte M, van Horssen J, Eggen B et al (2014) Demyelination during multiple sclerosis is associated with combined activation of microglia/macrophages by IFN-Γ and alpha B-crystallin. Acta Neuropathol 128(2):215–229
Genain C, Cannella B, Hauser S, Raine C (1999) Identification of autoantibodies associated with myelin damage in multiple sclerosis. Nat Med 5(2):170–175
Huizinga R, van der Star BJ, Kipp M, Jong R, Gerritsen W, Clarner T, Puentes F, Dijkstra CD et al (2012) Phagocytosis of neuronal debris by microglia is associated with neuronal damage in multiple sclerosis. Glia 60(3):422–431
Storer PD, Xu J, Chavis J, Pd D (2005) Peroxisome proliferator-activated receptor-gamma agonists inhibit the activation of microglia and astrocytes: implications for multiple sclerosis. J Neuroimmunol 161:113–122
Napoli I, Neumann H (2010) Protective effects of microglia in multiple sclerosis. Exp Neurol 225(1):24–28
Gilad O, Shefer-Averbuch N, Garty BZ (2015) Primary varicella infection presenting with headache and elevated intracranial pressure. J Child Neurol 30(6):793–795
Wang JP, Bowen GN, Zhou S, Cerny A, Zacharia A, Knipe DM, Finberg RW, Kurt-Jones EA (2012) Role of specific innate immune responses in herpes simplex virus infection of the central nervous system. J Virol 86(4):2273–2281
Olson JK, Girvin AM, Miller SD (2001) Direct activation of innate and antigen-presenting functions of microglia following infection with Theiler’s virus. J Virol 75(20):9780–9789
Williams K, Ulvestad E, Antel J (1994) Immune regulatory and effector properties of human adult microglia studied in vitro and in situ. Adv Neuroimmunol 4(3):273–281
Hsieh HL, Lin CC, Shih RH, Hsiao LD, Yang CM (2012) NADPH oxidase-mediated redox signal contributes to lipoteichoic acid-induced MMP-9 upregulation in brain astrocytes. J Neuroinflammation 9(8):78–79
Miller BA, Crum JM, Tovar CA, Ferguson AR, Bresnahan JC, Beattie MS (2007) Developmental stage of oligodendrocytes determines their response to activated microglia in vitro. J Neuroinflammation 4(12):109–114
Ramesh G, Benge S, Pahar B, Philipp MT (2012) A possible role for inflammation in mediating apoptosis of oligodendrocytes as induced by the Lyme disease spirochete Borrelia burgdorferi. J Neuroinflammation 9(1):97–101
Holley MM (2011) Th1 and Th17 cells regulate innate immune responses and bacterial clearance during central nervous system infection. J Immunol 188(3):1360–1370
Mckimmie CS, Roy D, Forster T, Fazakerley JK (2006) Innate immune response gene expression profiles of N9 microglia are pathogen-type specific. J Neuroimmunol 175(1–2):128–141
Ishibashi D, Atarashi R, Nishida N (2012) Protective role of MyD88-independent innate immune responses against prion infection. Prion 6(5):443–446
Lynch WP, Czub S, Mcatee FJ, Hayes SF, Portis JL (1991) Murine retrovirus-induced spongiform encephalopathy: productive infection of microglia and cerebellar neurons in accelerated CNS disease. Neuron 7(3):365–379
Payami H, Zareparsi S, James D, Nutt J (2002) Familial aggregation of Parkinson disease: a comparative study of early-onset and late-onset disease. Arch Neurol 59(5):848–850
Yeomans JS (1995) Role of tegmental cholinergic neurons in dopaminergic activation, antimuscarinic psychosis and schizophrenia. Neuropsychopharmacology 12(1):3–16
Hartmann A, Troadec J, Hunot S, Kikly K, Faucheux B, Mouatt-Prigent A, Ruberg M, Agid Y et al (2001) Caspase-8 is an effector in apoptotic death of dopaminergic neurons in Parkinson’s disease, but pathway inhibition results in neuronal necrosis. J Neurosci 21(7):2247–2255
Hirsch E, Hunot S, Faucheux B, Agid Y, Mizuno Y, Mochizuki H, Tatton W, Tatton N et al (1999) Dopaminergic neurons degenerate by apoptosis in Parkinson’s disease. Mov Disord 14(2):383–385
Politis M, Wu K, Molloy S, G Bain P, Chaudhuri KR, Piccini P (2010) Parkinson’s disease symptoms: the patient’s perspective. Mov Disord 25(11):1646–1651
Abramsky O, Litvin Y (1978) Autoimmune response to dopamine-receptor as a possible mechanism in the pathogenesis of Parkinson’s disease and schizophrenia. Perspect Biol Med 22(1):104–114
Marinova-Mutafchieva L, Sadeghian M, Broom L, Davis JB, Medhurst AD, Dexter DT (2009) Relationship between microglial activation and dopaminergic neuronal loss in the substantia nigra: a time course study in a 6-hydroxydopamine model of Parkinson’s disease. J Neurochem 110(3):966–975
Drechsel DA, Patel M (2008) Role of reactive oxygen species in the neurotoxicity of environmental agents implicated in Parkinson’s disease. Free Radic Biol Med 44(11):1873–1886
Tieu K, Ischiropoulos H, Przedborski S (2003) Nitric oxide and reactive oxygen species in Parkinson’s disease. IUBMB Life 55(6):329–335
Schaller B, Graf R (2004) Cerebral ischemia and reperfusion: the pathophysiologic concept as a basis for clinical therapy. J Cereb Blood Flow Metab 24(4):351–371
Coles J (2004) Incidence and mechanisms of cerebral ischemia in early clinical head injury. J Cereb Blood Flow Metab 24(2):202–211
Carolei A, Marini C, Prencipe M (1993) A prospective study of cerebral ischemia in the young analysis of pathogenic determinants. Stroke 24(3):362–367
Powers WJ, Clarke WR, Jr GR, Videen TO, Jr AH, Derdeyn CP (2011) Extracranial-intracranial bypass surgery for stroke prevention in hemodynamic cerebral ischemia: the Carotid Occlusion Surgery Study randomized trial. JAMA 306(5):1983–1992
Arranz AM, Gottlieb M, Pérez-Cerdá F, Matute C (2010) Increased expression of glutamate transporters in subcortical white matter after transient focal cerebral ischemia. Neurobiol Dis 37(1):156–165
Kong LL, Hu JF, Zhang W, Yuan YH, Ma KL, Han N, Chen NH (2011) Expression of chemokine-like factor 1 after focal cerebral ischemia in the rat. Neurosci Lett 505(1):14–18
Schilling M, Strecker JK, Ringelstein EB, Schäbitz WR, Kiefer R (2009) The role of CC chemokine receptor 2 on microglia activation and blood-borne cell recruitment after transient focal cerebral ischemia in mice. Brain Res 1289:79–84
Hazelton JL, Balan I, Elmer GI, Kristian T, Rosenthal RE, Krause G, Sanderson TH, Fiskum G (2010) Hyperoxic reperfusion after global cerebral ischemia promotes inflammation and long-term hippocampal neuronal death. J Neurotrauma 27(4):753–762
Yenari MA, Xu L, Tang XN, Qiao Y, Giffard RG (2006) Microglia potentiate damage to blood-brain barrier constituents: improvement by minocycline in vivo and in vitro. Stroke 37(4):1087–1093
Acknowledgments
This work was supported by the Liaoning Provincial Education Department Foundation of China (L2015030).
Author information
Authors and Affiliations
Corresponding author
Additional information
Ling Xu and Dan He contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xu, L., He, D. & Bai, Y. Microglia-Mediated Inflammation and Neurodegenerative Disease. Mol Neurobiol 53, 6709–6715 (2016). https://doi.org/10.1007/s12035-015-9593-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9593-4