Abstract
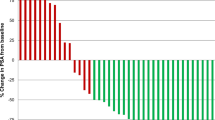
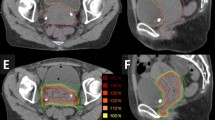
Ultra-hypofractionated radiotherapy (RT) is given over a shorter time with larger doses with respect to conventional fractionation in patients with localized prostate cancer (PCa). The use of hypofractionation is supported both from the radiobiological point of view (the low α/β-ratio in PCa and dose escalation) and from the rising number of clinical evidences. The aim of this study is to review our data regarding oncological outcomes, namely biochemical progression-free survival (b-PFS) and clinical progression-free survival (c-PFS), acute and long-term toxicities in patients treated with a ultra-hypofractionated RT. A series of 194 patients with clinically localized PCa treated primarily with ultra-hypofractionated RT using image-guided intensity modulated RT (IG-IMRT) at our Institute from 2012 to 2015 was included in this analysis. According to NCCN risk group classification, 65 (33.5%) patients were low risk, 101 (52.1%) intermediate risk, and 28 (14.4%) high risk. Androgen deprivation therapy (ADT) was given to 61 patients (31.4%). A 169 patients (87.1%) received 35 Gy in 5 fractions, while 25 patients (13%) received 32.5 Gy in 5 fractions (usually given in patients with comorbidity). The median duration of the treatment was 10 days (IQR 9–12). Biochemical relapse was defined as a rise of prostate specific antigen (PSA) > 2 ng/ml above nadir. b-PFS, c-PFS, and freedom from gastro-intestinal (GI) and genito-urinary (GU) toxicity curves were calculated by the Kaplan–Meier method. Log-rank test and multivariate Cox models were used to investigate the role RT dose and heterogeneity by NCCN risk groups adjusting for prognostic factors. Data on acute and late term toxicities were collected according to RTOG/EORTC grading system. With a median follow-up of 30 months, 17 patients experienced PSA failure (9%). The 3-year b-PFS was 87% for all patients and rates stratified for the NCCN risk were 94, 82, and 66% for low-, intermediate-, and high-risk groups, respectively. Log-rank tests indicate that biochemical progression was significantly greater for patients with initial PSA (iPSA) greater than 7 ng/ml (P = 0.04), high- and intermediate-risk groups (P = 0.002), low total dose (P = 0.02) and Gleason score (GS) equal or greater than 7 (P = 0.04). No statistically significant association was found with T stage nor ADT. In multivariate analyses, total dose (P = 0.03) and risk groups (P = 0.03) remained significantly associated with recurrence. Acute and late GI and GU toxicity were acceptable. The toxicity of ultra-hypofractionated IG-IMRT in a large clinical cohort of PCa patients was tolerable and confirmed that this treatment is safe and offers excellent tumor control. Moreover, the hypofractionated RT allows to deliver the whole RT over 10 days with a sensible impact in patients’ quality of life and potential overall health system and social benefits.




Similar content being viewed by others
References
Donovan JL, Hamdy FC, Lane JA, et al. ProtecT Study Group. Patient-Reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2016; 13(15):1425–37. https://doi.org/10.1056/NEJMoa1606221.
Arcangeli S, Arcangeli G. Moderate hypofractionation for prostate cancer. Oncotarget. 2017; 8(49):84612–3. https://doi.org/10.18632/oncotarget.21386.
Fowler JF. The radiobiology of prostate cancer including new aspects of fractionated radiotherapy. Acta Oncol. 2005;44:265–76.
Kuban DA, Tucker SL, Dong L, et al. Long-term results of the M. D. Anderson randomized dose-escalation trial for prostate cancer. Int J Radiat Oncol Biol Phys. 2008;70(1):67–74.
Zietman AL, DeSilvio ML, Slater JD, et al. Comparison of conventional-dose vs high-dose conformal radiation therapy in clinically localized adenocarcinoma of the prostate: a randomized controlled trial. JAMA. 2005;294:1233–9.
Benjamin LC, Tree AC, Dearnaley DP. The role of hypofractionated radiotherapy in prostate cancer. Curr Oncol Rep. 2017;19(4):30. https://doi.org/10.1007/s11912-017-0584-7.
Brenner DJ, Martinez AA, Edmundson GK, et al. Direct evidence that prostate tumors show high sensitivity to fractionation (low alpha/beta ratio), similar to late-responding normal tissue. Int J Radiat Oncol Biol Phys. 2002;52:6–13.
Martinez AA, Demanes J, Vargas C, et al. High-dose-rate prostate brachytherapy: an excellent accelerated-hypofractionated treatment for favorable prostate cancer. Am J Clin Oncol. 2010;33(5):481–8. https://doi.org/10.1097/COC.0b013e3181b9cd2f.
Demanes DJ, Martinez AA, Ghilezan M, et al. High-dose-rate monotherapy: safe and effective brachytherapy for patients with localized prostate cancer. Int J Radiat Oncol Biol Phys. 2011;81(5):1286–92. https://doi.org/10.1016/j.ijrobp.2010.10.015.
Meier R, Kaplan I, Beckman A, et al. Stereotactic body radiation therapy for intermediate-risk organ confined prostate cancer: interim toxicity and quality of life outcomes from a multiinstitutional study. Int J Radiat Oncol Biol Phys. 2012;84:S148. https://doi.org/10.1016/j.ijrobp.2012.07.382.
King CR, Brooks JD, Gill H, Presti JC. Long-term outcomes from a prospective trial of stereotactic body radiotherapy for low-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2012;82(2):877–82. https://doi.org/10.1016/j.ijrobp.2010.11.054.
De Bari B, Arcangeli S, Ciardo D, et al; on the behalf of the Italian Association of Radiation Oncology (AIRO). Extreme hypofractionation for early prostate cancer: biology meets technology. Cancer Treat Rev. 2016;50:48–60. https://doi.org/10.1016/j.ctrv.2016.08.005.
Cox JD, Stetz J, Pajak TF, et al. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European organization for research and treatment of cancer (EORTC). Int J Radiat Oncol Biol Phys. 1995;31(5):1341–6. https://doi.org/10.1016/0360-3016(95)00060-C PMID: 7713792.
Arcangeli S, Greco C. Hypofractionated radiotherapy for organ-confined prostate cancer: is less more? Nat Rev Urol. 2016;13(7):400–8. https://doi.org/10.1038/nrurol.2016.106.
Arcangeli G, Saracino B, Arcangeli S, et al. Moderate hypofractionation in high-risk, organ-confined prostate cancer: final results of a phase III randomized trial. J Clin Oncol. 2017;10(17):1891–7. https://doi.org/10.1200/JCO.2016.70.4189.
Fuller DB, Mardirossian G, Wong D, Diblasio F. Prospective evaluation of stereotactic body radiation therapy for low and intermediate-risk prostate cancer: emulating high-dose rate brachytherapy dose-distribution. Int J Radiat Oncol Biol Phys. 2012;84:S149. https://doi.org/10.1016/j.ijrobp.2012.07.384.
Katz AJ, Kang J. Quality of life and toxicity after SBRT for organ-confined prostate cancer, a 7-year study. Front Oncol. 2014;4:301. https://doi.org/10.3389/fonc.2014.00301.
Katz A. Stereotactic body radiotherapy for low-risk prostate cancer: a 10-year analysis. Cureus. 2017;9(9):e1668. https://doi.org/10.7759/cureus.1668.
Syed YA, Patel-Yadav AK, Rivers C, Singh AK. Stereotactic radiotherapy for prostate cancer: a review and future directions. World J Clin Oncol. 2017;8(5):389–97. https://doi.org/10.5306/wjco.v8.i5.389.
Katz AJ, Santoro M, Diblasio F, Ashley R. Stereotactic body radiotherapy for localized prostate cancer: disease control and quality of life at 6 years. Radiat Oncol. 2013;8(1):118. https://doi.org/10.1186/1748-717X-8-118.
Phase III. Study of HYPO-fractionated RadioTherapy of intermediate risk localised prostate cancer. 2009. http://www.controlled-trials.com/ISRCTN45905321.
Radiation Hypofractionation via extended versus accelerated therapy (HEAT) for prostate cancer. 2013. https://www.clinicaltrials.gov/show/NCT01794403.
NCCN. Clinical practice guidelines in oncology. http://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf. Accessed Dec 18 2017.
Grégoire V, Mackie TR. State of the art on dose prescription, reporting and recording in intensity-modulated radiation therapy (ICRU report No. 83). Cancer Radiother. 2011;15(6):555–9. https://doi.org/10.1016/j.canrad.2011.04.003.
Friedland JL, Freeman DE, Masterson-McGary ME, Spellberg DM. Stereotactic body radiotherapy: an emerging treatment approach for localized prostate cancer. Technol Cancer Res Treat. 2009;8:387–92.
King CR, Freeman D, Kaplan I, et al. Stereotactic body radiotherapy for localized prostate cancer: pooled analysis from a multi-institutional consortium of prospective phase II trials. Radiother Oncol. 2013;109(2):217–21. https://doi.org/10.1016/j.radonc.2013.08.030.
Martin JM, Frantzis J, Eade T, Chung P. Clinician’s guide to prostate IMRT plan assessment and optimization. J Med Imaging Radiat Oncol. 2010;54(6):569–75. https://doi.org/10.1111/j.1754-9485.2010.02217.x.
Hentschel B, Oehler W, Strauss D, et al. Definition of the CTV prostate in CT and MRI by using CT-MRI image fusion in IMRT planning for prostate cancer. Strahlenther Onkol. 2011;187(3):183–90. https://doi.org/10.1007/s00066-010-2179-1.
Timon G, Ciardo D, Bazani A, et al. Rationale and protocol of AIRC IG-13218, short-term radiotherapy for early prostate cancer with concomitant boost to the dominant lesion. Tumori. 2016;102(5):536–40. https://doi.org/10.5301/tj.5000547.
Riva G, Timon G, Ciardo D, et al. High precision radiotherapy for early prostate cancer with concomitant boost to the dominant lesion. Radiother Oncol. 2017;123:S717-S718. https://doi.org/10.1016/S0167-8140(17)31773-5.
Ciardo D, Jereczek-Fossa BA, Petralia G, et al. Multimodal image registration for the identification of dominant intraprostatic lesion in high-precision radiotherapy treatments. Br J Radiol. 2017;90(1079):20170021. https://doi.org/10.1259/bjr.20170021.
Henderson DR, Tree AC, van As NJ. Stereotactic body radiotherapy for prostate cancer. Clin Oncol. 2015;27(5):270–9. https://doi.org/10.1016/j.clon.2015.01.011.
Mantz C. A phase II trial of stereotactic ablative body radiotherapy for low-risk prostate cancer using a non-robotic linear accelerator and real-time target tracking: report of toxicity, quality of life, and disease control outcomes with 5-year minimum follow-up. Front Oncol. 2014;4:279. https://doi.org/10.3389/fonc.2014.00279.
Aluwini S, Beltramo G, Van Rooij P, et al. Stereotactic body radiotherapy with four fraction for low and intermediate risk prostate cancer: acute and late toxicity. Radioter Oncol. 2013;8:84. https://doi.org/10.1186/1748-717X-8-84.
Meier R, Kaplan I, Beckman A, et al. Patient-reported quality of life outcomes in intermediate-risk prostate cancer patients treated with stereotactic body radiation therapy. Int J Radiat Oncol Biol Phys. 2013;87:S25. https://doi.org/10.1016/j.ijrobp.2013.06.069.
Ricco A, Hanlon A, Lanciano R. Propensity score matched comparison of intensity modulated radiation therapy vs stereotactic body radiation therapy for localized prostate cancer: a survival analysis from the National cancer database. Front Oncol. 2017;7:185. https://doi.org/10.3389/fonc.2017.00185.
ASTRO. Stereotactic body radiation therapy model policy. In: Stereotactic Body Radiation Therapy Model Policy; 2013.
Prostate advances in comparative evidence (PACE). 2012. https://clinicaltrials.gov/show/NCT01584258.
Prostate accurately targeted radiotherapy investigation of overall treatment time (PATRIOT). 2011. http://clinicaltrials.gov/show/NCT01423474.
Randomized phase A II trial of hypofractionated radiotherapy for favorable risk prostate cancer-RTOG CCOP Study (RTOG 0938). https://www.rtog.org/ClinicalTrials/ProtocolTable/StudyDetails.aspx?study=0938.
Acknowledgements
This work was partially supported by the research grants from the Associazione Italiana per la Ricerca sul Cancro (AIRC): IG-13218 “Short-term high precision RT for early prostate cancer with concomitant boost to the dominant lesion,” registered at ClinicalTrials.gov NCT01913717, approved by IEO S768/113 and IG-14300 “Carbon ions boost followed by pelvic photon radiotherapy for high risk prostate cancer,” The Sponsor did not play any role in the study design, collection, analysis and interpretation of data, nor in the writing of the manuscript, nor in the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interest.
Ethical approval
In this research, no animals were involved. All patients signed a written informed consent for stereotactic body radiation therapy (SBRT) and written informed consent for the use of the anonymized data for research or educational purpose. The study was performed within the Institutional Ethics Committee notification regarding clinical and dosimetric aspects of hypofractionated image-guided RT (IGRT) for PCa (CE notification n. 79).
Rights and permissions
About this article
Cite this article
Marvaso, G., Riva, G., Ciardo, D. et al. “Give me five” ultra-hypofractionated radiotherapy for localized prostate cancer: non-invasive ablative approach. Med Oncol 35, 96 (2018). https://doi.org/10.1007/s12032-018-1155-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-018-1155-y