Abstract
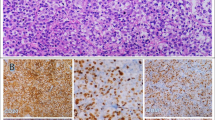
The objective was to investigate the expression and prognostic value of CD59 expression in patients with diffuse large B-cell lymphoma (DLBCL) who underwent rituximab-cyclophosphamide, adriamycin, vincristine, and prednisone (R-CHOP). The immunohistochemical expressions of CD59 in 186 well-characterized DLBCL patients were evaluated using tissue microarrays and then were related to known tumor- and patient-related variables and to survival. The results show that CD59 expressions were not statistically different between the germinal center B-cell-like-type and the activated B-cell-like-type. We also analyzed the relationships of CD59 expression with overall survival (OS) and progression-free survival (PFS) in DLBCL patients who were uniformly treated with R-CHOP. The high expression of CD59 was correlated with poor OS and PFS compared with the low-expression CD59. Our findings indicate that the CD59 level at onset is an independent predictor of the prognosis of DLBCL patients treated with R-CHOP.



Similar content being viewed by others
References
Swerdlow SHCE, et al. WHO classification of tumours of haematopoietic and lymphoid tissues. 4th ed. Lyon, France: IARC Press; 2008.
Habermann TM, et al. Rituximab-CHOP versus CHOP alone or with maintenance rituximab in older patients with diffuse large B-cell lymphoma. J Clin Oncol. 2006;24:3121–7.
Friendberg Jonathan W. New strategies in diffuse large B-cell lymphoma: translating finding from gene expression analyses into clinical practice. Clin Cancer Res. 2011;17:6112–6.
Okamura T. A predictive model for aggressive non-Hodgkin’s lymphoma. The international Non-Hodgkin’s lymphoma prognostic factors project. N Engl J Med. 1993;329:987–94.
Hou Y, Wang HQ, Ba Y. High expression of cell division cycle 7 protein correlates with poor prognosis in patients with diffuse large B-cell lymphoma. Med Oncol. 2012;29:3498–503.
Provencio M, et al. Caspase 3a: new prognostic marker for diffuse large B-cell lymphoma in the rituximab era. Leuk Lymphoma. 2010;51:2021–30.
Wang H, et al. A recombinant adenovirus type 35 fiber knob protein sensitizes lymphoma cells to rituximab therapy. Blood. 2010;115:592–600.
Golay J, et al. Biologic response of B lymphoma cells to anti-CD20 monoclonal antibody rituximab in vitro: CD55 and CD59 regulate complement-mediated cell lysis. Blood. 2000;95:3900–8.
Kononen J, Bubendorf L, Kallioniemi A, Barlund M, Schraml P, Leighton S, Torhorst J, Mihatsch MJ, Sauter G, Kallioniemi OP. Tissue microarrays for high throughput molecular profiling of tumour specimens. Nat Med. 1998;4:844–7.
Cheson BD, et al. Revised response criteria for malignant lymphoma. J Clin Oncol. 2007;25:579–86.
Torhorst J, et al. Tissue microarrays for the rapid linking of molecular changes to clinical endpoints. Am J Pathol. 2001;159:2249–56.
Camp RL, Charette LA, Rimm DL. Validation of tissue microarray technology in breast carcinoma. Lab Invest. 2000;80:1943–9.
Coiffier B, et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med. 2002;346:235–42.
Pfreundschuh M, et al. CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large-B-cell lymphoma: a randomised controlled trial by the MabThera International Trial (MInT) Group. Lancet Oncol. 2006;7:379–91.
Sehn LH, et al. Introduction of combined CHOP plus rituximab therapy dramatically improved outcome of diffuse large B-cell lymphoma in British Columbia. J Clin Oncol. 2005;23:5027–33.
Shustik J, et al. Correlations between BCL6 rearrangement and outcome in patients with diffuse large B-cell lymphoma treated with CHOP or R-CHOP. Haematologica. 2010;95:96–101.
Maloney DG, et al. IDEC-C2B8 (rituximab) anti-CD20monoclonal antibody therapy in patients with relapsed low-grade non-Hodgkin’s lymphoma. Blood. 1997;90:2188–95.
Villamor N, Montserrat E, Colomer D. Mechanism of action and resistance to monoclonal antibody therapy. Semin Oncol. 2003;30:424–33.
You T, Hu WG, Ge XW, Shen JN, Qin XB. Application of a novel inhibitor of human CD59 for the enhancement of complement-dependent cytolysis on cancer cells. Cell Mol Immunol. 2011;8:157–63.
Hu WG, Ge XW, You T, Xu T, Zhang JY. Human CD59 inhibitor sensitizes rituximab-resistant lymphoma cells to complement-mediated cytolysis. Cancer Res. 2011;71:2298–307.
Ge XW, Wu L, Hu WG, Fernandes S. rILYd4, a human CD59 inhibitor, enhances complement-dependent cytotoxicity of ofatumumab against rituximab resistant B-cell lymphoma cells and chronic lymphocytic leukemia. Clin Cancer Res. 2011;17:6702–11.
Golay J, Lazzari M, Facchinetti V. CD20 levels determine the in vitro susceptibility to rituximab and complement of B-cell chronic lymphocytic leukemia: further regulation by CD55 and CD59. Blood. 2001;98:3383–9.
Dzietczenia J, et al. Expression of complement regulatory proteins: CD46, CD55, and CD59 and response to rituximab in patients with CD20 + non-Hodgkin’s lymphoma. Med Oncol. 2010;27:743–6.
Weng WK. Levy: Expression of complement inhibitors CD46, CD55, and CD59 on tumor cells does not predict clinical outcome after rituximab treatment in follicular non-Hodgkin lymphoma. Blood. 2001;98:1352–7.
Conflict of interest
The author(s) confirm that this article content has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, G., Cho, W.C., Gu, L. et al. Increased CD59 protein expression is associated with the outcome of patients with diffuse large B-cell lymphoma treated with R-CHOP. Med Oncol 31, 56 (2014). https://doi.org/10.1007/s12032-014-0056-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-014-0056-y