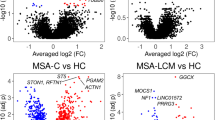
Abstract
Brain regions such as the cerebellum (CB) have been neglected for a long time in the study of Alzheimer’s disease (AD) pathogenesis. In reference to a new emerging hypothesis according to which there is an altered cerebellar synaptic processing in AD, we verified the possible role played by new biomarkers in the CB of AD patients compared with not-demented healthy control subjects (NDHS). Using a bioinformatics approach, we have collected several microarray datasets and obtained 626 cerebella sample biopsies belonging to subjects who did not die from causes related to neurological diseases and 199 cerebella belonging to AD. The analysis of logical relations between the transcriptome dataset highlighted guanine nucleotide–binding protein (G protein) gamma 13 (GNG13) as a potential new biomarker for Purkinje cells (PCs). We have correlated GNG13 expression levels with already widely existing bibliography of PC marker genes, such as Purkinje cell protein 2 (PCP2), Purkinje cell protein 4 (PCP4), and cerebellin 3 (CBLN3). We showed that expression levels of GNG13 and PCP2, PCP4, and CBLN3 were significantly correlated with each other in NDHS and in AD and significantly reduced in AD patients compared with NDHS subjects. In addition, we highlighted a negative correlation between the expression levels of PC biomarkers and age. From the outcome of our investigation, it is possible to conclude that the identification of GNG13 as a potentially biomarker in PCs represents also a state of health of CB, in association with the expression of PCP2, PCP4, and CBLN3.






Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- Aβ:
-
Amyloid beta
- CNS:
-
Central nervous system
- PFC:
-
Prefrontal cortex
- TL:
-
The temporal lobes
- PL:
-
Parietal lobe
- PCP2:
-
Purkinje cell protein 2
- PCP4:
-
Purkinje cell protein 4
- CBLN3:
-
Cerebellin 3
- GPCRs:
-
G protein–coupled receptors
- NDHS:
-
Not-demented healthy control subjects
- HPA:
-
Human Protein Atlas
- G protein:
-
Guanine nucleotide binding protein
- GNG13:
-
Gamma 13
- SDEG:
-
Significant differentially expressed genes
- MeV:
-
MultiExperiment Viewer
- HIP:
-
Hippocampus
- CB:
-
Cerebellum
- PCs:
-
Purkinje cells
- PCL:
-
Purkinje cell layer
- LCMD:
-
Laser capture microdissection
- EGFP:
-
Enhanced green fluorescent protein
- APP:
-
Amyloid precursor protein
References
Andreasen NC, O’Leary DS, Paradiso S, Cizadlo T, Arndt S, Watkins GL, Boles Ponto LL, Hichwa RD (1999) The cerebellum plays a role in conscious episodic memory retrieval. Hum Brain Mapp 8:226–234
Apps R, Garwicz M (2005) Anatomical and physiological foundations of cerebellar information processing. Nat Rev Neurosci 6:297–311. https://doi.org/10.1038/nrn1646
Asrican B, Augustine GJ, Berglund K, Chen S, Chow N, Deisseroth K, Feng G, Gloss B, Hira R, Hoffmann C, Kasai H, Katarya M, Kim J, Kudolo J, Lee LM, Lo SQ, Mancuso J, Matsuzaki M, Nakajima R, Qiu L, Tan G, Tang Y, Ting JT, Tsuda S, Wen L, Zhang X, Zhao S (2013) Next-generation transgenic mice for optogenetic analysis of neural circuits. Front Neural Circuits 7:160. https://doi.org/10.3389/fncir.2013.00160
Bao D, Pang Z, Morgan MA, Parris J, Rong Y, Li L, Morgan JI (2006) Cbln1 is essential for interaction-dependent secretion of Cbln3. Mol Cell Biol 26:9327–9337. https://doi.org/10.1128/MCB.01161-06
Baumann O et al (2015) Consensus paper: the role of the cerebellum in perceptual processes. Cerebellum 14:197–220. https://doi.org/10.1007/s12311-014-0627-7
Becker EB, Stoodley CJ (2013) Autism spectrum disorder and the cerebellum. Int Rev Neurobiol 113:1–34. https://doi.org/10.1016/B978-0-12-418700-9.00001-0
Berrebi AS, Oberdick J, Sangameswaran L, Christakos S, Morgan JI, Mugnaini E (1991) Cerebellar Purkinje cell markers are expressed in retinal bipolar neurons. J Comp Neurol 308:630–649. https://doi.org/10.1002/cne.903080409
Blakemore SJ, Wolpert DM, Frith CD (1999) The cerebellum contributes to somatosensory cortical activity during self-produced tactile stimulation. NeuroImage 10:448–459. https://doi.org/10.1006/nimg.1999.0478
Blennow K, de Leon MJ, Zetterberg H (2006) Alzheimer’s disease. Lancet 368:387–403. https://doi.org/10.1016/S0140-6736(06)69113-7
Braak H, Braak E, Bohl J, Lang W (1989) Alzheimer’s disease: amyloid plaques in the cerebellum. J Neurol Sci 93:277–287. https://doi.org/10.1016/0022-510x(89)90197-4
Bucks RS, Radford SA (2004) Emotion processing in Alzheimer’s disease. Aging Ment Health 8:222–232. https://doi.org/10.1080/13607860410001669750
Byron CD, Vanvalkinburgh D, Northcutt K, Young V (2013) Plasticity in the cerebellum and primary somatosensory cortex relating to habitual and continuous slender branch climbing in laboratory mice (Mus musculus). Anat Rec 296:822–833. https://doi.org/10.1002/ar.22685
Cabin DE, Gardiner K, Reeves RH (1996) Molecular genetic characterization and comparative mapping of the human PCP4 gene. Somat Cell Mol Genet 22:167–175. https://doi.org/10.1007/bf02369907
Caltabiano R et al (2019) Identification of novel markers of prostate cancer progression, potentially modulated by vitamin D. Appl Sci - Basel 9:ARTN 4923. https://doi.org/10.3390/app9224923
Care MA, Barrans S, Worrillow L, Jack A, Westhead DR, Tooze RM (2013) A microarray platform-independent classification tool for cell of origin class allows comparative analysis of gene expression in diffuse large B-cell lymphoma. PLoS One 8:e55895. https://doi.org/10.1371/journal.pone.0055895
Castrogiovanni P et al (2018) Fasting and fast food diet play an opposite role in mice brain aging. Mol Neurobiol 55:6881–6893. https://doi.org/10.1007/s12035-018-0891-5
Catrogiovanni P, Musumeci G, Giunta S, Imbesi R, Di Rosa M (2020) The expression levels of CHI3L1 and IL15Ralpha correlate with TGM2 in duodenum biopsies of patients with celiac disease. Inflamm Res 69:925–935. https://doi.org/10.1007/s00011-020-01371-9
Chacon PJ, del Marco A, Arevalo A, Dominguez-Gimenez P, Garcia-Segura LM, Rodriguez-Tebar A (2015) Cerebellin 4, a synaptic protein, enhances inhibitory activity and resistance of neurons to amyloid-beta toxicity. Neurobiol Aging 36:1057–1071. https://doi.org/10.1016/j.neurobiolaging.2014.11.006
Cheadle C, Cho-Chung YS, Becker KG, Vawter MP (2003a) Application of z-score transformation to Affymetrix data. Appl Bioinforma 2:209–217
Cheadle C, Vawter MP, Freed WJ, Becker KG (2003b) Analysis of microarray data using Z score transformation. J Mol Diagn 5:73–81
Chen C et al. (2018) The transcription factor POU3F2 regulates a gene coexpression network in brain tissue from patients with psychiatric disorders. Sci Transl Med 10. https://doi.org/10.1126/scitranslmed.aat8178
Chen QR, Song YK, Wei JS, Bilke S, Asgharzadeh S, Seeger RC, Khan J (2008) An integrated cross-platform prognosis study on neuroblastoma patients. Genomics 92:195–203. https://doi.org/10.1016/j.ygeno.2008.05.014
Dalman MR, Deeter A, Nimishakavi G, Duan ZH (2012) Fold change and p-value cutoffs significantly alter microarray interpretations. BMC Bioinformatics 13(Suppl 2):S11. https://doi.org/10.1186/1471-2105-13-S2-S11
Di Rosa M, Sanfilippo C, Libra M, Musumeci G, Malaguarnera L (2015) Different pediatric brain tumors are associated with different gene expression profiling. Acta Histochem 117:477–485. https://doi.org/10.1016/j.acthis.2015.02.010
Di Rosa M, Tibullo D, Vecchio M, Nunnari G, Saccone S, Di Raimondo F, Malaguarnera L (2014) Determination of chitinases family during osteoclastogenesis. Bone 61:55–63. https://doi.org/10.1016/j.bone.2014.01.005
Doyle JP et al (2008) Application of a translational profiling approach for the comparative analysis of CNS cell types. Cell 135:749–762. https://doi.org/10.1016/j.cell.2008.10.029
Ernst TM, Brol AE, Gratz M, Ritter C, Bingel U, Schlamann M, Maderwald S, Quick HH, Merz CJ, Timmann D (2019) The cerebellum is involved in processing of predictions and prediction errors in a fear conditioning paradigm. Elife 8. https://doi.org/10.7554/eLife.46831
Fagone P et al (2019) Identification of CD4+ T cell biomarkers for predicting the response of patients with relapsing remitting multiple sclerosis to natalizumab treatment. Mol Med Rep 20:678–684. https://doi.org/10.3892/mmr.2019.10283
Fagone P et al (2016) Induction of OAS gene family in HIV monocyte infected patients with high and low viral load. Antivir Res 131:66–73. https://doi.org/10.1016/j.antiviral.2016.04.009
Feng C et al (2018) Expression of Bcl-2 is a favorable prognostic biomarker in lung squamous cell carcinoma. Oncol Lett 15:6925–6930. https://doi.org/10.3892/ol.2018.8198
Ferdon S, Murphy C (2003) The cerebellum and olfaction in the aging brain: a functional magnetic resonance imaging study. Neuroimage 20:12–21. https://doi.org/10.1016/s1053-8119(03)00276-3
Frankel R et al (2019) Autocatalytic amplification of Alzheimer-associated Abeta42 peptide aggregation in human cerebrospinal fluid. Commun Biol 2:365. https://doi.org/10.1038/s42003-019-0612-2
Fusar-Poli P et al (2009) Functional atlas of emotional faces processing: a voxel-based meta-analysis of 105 functional magnetic resonance imaging studies. J Psychiatry Neurosci JPN 34:418–432
Garcia LI, Garcia-Banuelos P, Aranda-Abreu GE, Herrera-Meza G, Coria-Avila GA, Manzo J (2015) Activation of the cerebellum by olfactory stimulation in sexually naive male rats. Neurologia 30:264–269. https://doi.org/10.1016/j.nrl.2014.02.002
Gibbs JR, van der Brug MP, Hernandez DG, Traynor BJ, Nalls MA, Lai SL, Arepalli S, Dillman A, Rafferty IP, Troncoso J, Johnson R, Zielke HR, Ferrucci L, Longo DL, Cookson MR, Singleton AB (2010) Abundant quantitative trait loci exist for DNA methylation and gene expression in human brain. PLoS Genet 6:e1000952. https://doi.org/10.1371/journal.pgen.1000952
Heintz N (2004) Gene expression nervous system atlas (GENSAT). Nat Neurosci 7:483. https://doi.org/10.1038/nn0504-483
Herman AM, Critchley H, Duka T (2018) Decreased olfactory discrimination is associated with impulsivity in healthy volunteers. Sci Rep 8:15584. https://doi.org/10.1038/s41598-018-34056-9
Hernandez DG et al (2012) Integration of GWAS SNPs and tissue specific expression profiling reveal discrete eQTLs for human traits in blood and brain. Neurobiol Dis 47:20–28. https://doi.org/10.1016/j.nbd.2012.03.020
Hirai H et al (2005) Cbln1 is essential for synaptic integrity and plasticity in the cerebellum. Nat Neurosci 8:1534–1541. https://doi.org/10.1038/nn1576
Hornsey VS, Waterston YG, Prowse CV (1988) Artificial factor VIII deficient plasma: preparation using monoclonal antibodies and its use in one stage coagulation assays. J Clin Pathol 41:562–567. https://doi.org/10.1136/jcp.41.5.562
Huang L et al (1999) Ggamma13 colocalizes with gustducin in taste receptor cells and mediates IP3 responses to bitter denatonium. Nat Neurosci 2:1055–1062. https://doi.org/10.1038/15981
Jacobs HIL, Hopkins DA, Mayrhofer HC, Bruner E, van Leeuwen FW, Raaijmakers W, Schmahmann JD (2018) The cerebellum in Alzheimer’s disease: evaluating its role in cognitive decline. Brain 141:37–47. https://doi.org/10.1093/brain/awx194
Kang C, Huo Y, Xin L, Tian B, Yu B (2019) Feature selection and tumor classification for microarray data using relaxed lasso and generalized multi-class support vector machine. J Theor Biol 463:77–91. https://doi.org/10.1016/j.jtbi.2018.12.010
Kang HJ et al (2011) Spatio-temporal transcriptome of the human brain. Nature 478:483–489. https://doi.org/10.1038/nature10523
Kaya I, Brinet D, Michno W, Baskurt M, Zetterberg H, Blenow K, Hanrieder J (2017) Novel trimodal MALDI imaging mass spectrometry (IMS3) at 10 mum reveals spatial lipid and peptide correlates implicated in Aβ plaque pathology in Alzheimer’s disease. ACS Chem Neurosci 8:2778–2790. https://doi.org/10.1021/acschemneuro.7b00314
Kerr DS, Von Dannecker LE, Davalos M, Michaloski JS, Malnic B (2008) Ric-8B interacts with G alpha olf and G gamma 13 and co-localizes with G alpha olf, G beta 1 and G gamma 13 in the cilia of olfactory sensory neurons. Mol Cell Neurosci 38:341–348. https://doi.org/10.1016/j.mcn.2008.03.006
Kumar A et al (2013) Age-associated changes in gene expression in human brain and isolated neurons. Neurobiol Aging 34:1199–1209. https://doi.org/10.1016/j.neurobiolaging.2012.10.021
Le Cao KA, Rohart F, McHugh L, Korn O, Wells CA (2014) YuGene: a simple approach to scale gene expression data derived from different platforms for integrated analyses. Genomics 103:239–251. https://doi.org/10.1016/j.ygeno.2014.03.001
Li JZ et al (2013) Circadian patterns of gene expression in the human brain and disruption in major depressive disorder. Proc Natl Acad Sci U S A 110:9950–9955. https://doi.org/10.1073/pnas.1305814110
Liu Q, Li S, Lu C, Yu CR, Huang L (2018) G protein gamma subunit Ggamma13 is essential for olfactory function and aggressive behavior in mice. Neuroreport 29:1333–1339. https://doi.org/10.1097/WNR.0000000000001122
Liu Z et al (2012) Identification of new binding partners of the chemosensory signaling protein Ggamma13 expressed in taste and olfactory sensory cells. Front Cell Neurosci 6:26. https://doi.org/10.3389/fncel.2012.00026
Magdaleno S, Jensen P, Brumwell CL, Seal A, Lehman K, Asbury A, Cheung T, Cornelius T, Batten DM, Eden C, Norland SM, Rice DS, Dosooye N, Shakya S, Mehta P, Curran T (2006) BGEM: an in situ hybridization database of gene expression in the embryonic and adult mouse nervous system. PLoS Biol 4:e86. https://doi.org/10.1371/journal.pbio.0040086
Mainland JD, Johnson BN, Khan R, Ivry RB, Sobel N (2005) Olfactory impairments in patients with unilateral cerebellar lesions are selective to inputs from the contralesional nostril. J Neurosci 25:6362–6371. https://doi.org/10.1523/JNEUROSCI.0920-05.2005
Malaguarnera L, Imbesi R, Di Rosa M, Scuto A, Castrogiovanni P, Messina A, Sanfilippo S (2005) Action of prolactin, IFN-gamma, TNF-alpha and LPS on heme oxygenase-1 expression and VEGF release in human monocytes/macrophages. Int Immunopharmacol 5:1458–1469. https://doi.org/10.1016/j.intimp.2005.04.002
Malaguarnera L, Nunnari G, Di Rosa M (2016) Nuclear import sequence identification in hOAS3 protein. Inflamm Res 65:895–904. https://doi.org/10.1007/s00011-016-0972-8
Mammana S et al (2018) Preclinical evaluation of the PI3K/Akt/mTOR pathway in animal models of multiple sclerosis. Oncotarget 9:8263–8277. https://doi.org/10.18632/oncotarget.23862
Mangano K, Cavalli E, Mammana S, Basile MS, Caltabiano R, Pesce A, Puleo S, Atanasov AG, Magro G, Nicoletti F, Fagone P (2018) Involvement of the Nrf2/HO-1/CO axis and therapeutic intervention with the CO-releasing molecule CORM-A1, in a murine model of autoimmune hepatitis. J Cell Physiol 233:4156–4165. https://doi.org/10.1002/jcp.26223
Manto M et al (2012) Consensus paper: roles of the cerebellum in motor control--the diversity of ideas on cerebellar involvement in movement. Cerebellum 11:457–487. https://doi.org/10.1007/s12311-011-0331-9
Maschke M et al (2002) Fear conditioned changes of heart rate in patients with medial cerebellar lesions. J Neurol Neurosurg Psychiatry 72:116–118
Mehmood R, El-Ashram S, Bie R, Dawood H, Kos A (2017) Clustering by fast search and merge of local density peaks for gene expression microarray data. Sci Rep 7:45602. https://doi.org/10.1038/srep45602
Musumeci G et al (2018) Expression of the OAS gene family is highly modulated in subjects affected by juvenile dermatomyositis, resembling an immune response to a dsRNA virus infection. Int J Mol Sci 19. https://doi.org/10.3390/ijms19092786
Nicoletti A et al (2011) Parkinson’s disease is associated with increased serum levels of macrophage migration inhibitory factor. Cytokine 55:165–167. https://doi.org/10.1016/j.cyto.2011.03.027
Nordquist DT, Kozak CA, Orr HT (1988) cDNA cloning and characterization of three genes uniquely expressed in cerebellum by Purkinje neurons. J Neurosci 8:4780–4789
Nunnari G et al (2012) The role of micronutrients in the diet of HIV-1-infected individuals. Front Biosci 4:2442–2456. https://doi.org/10.2741/e556
Nunnari G et al (2020) Network perturbation analysis in human bronchial epithelial cells following SARS-CoV2 infection. Exp Cell Res 395:112204. https://doi.org/10.1016/j.yexcr.2020.112204
Pang Z, Zuo J, Morgan JI (2000) Cbln3, a novel member of the precerebellin family that binds specifically to Cbln1. J Neurosci 20:6333–6339
Patel H, Hodges AK, Curtis C, Lee SH, Troakes C, Dobson RJB, Newhouse SJ (2019) Transcriptomic analysis of probable asymptomatic and symptomatic Alzheimer brains. Brain Behav Immun 80:644–656. https://doi.org/10.1016/j.bbi.2019.05.009
Phillips JR, Hewedi DH, Eissa AM, Moustafa AA (2015) The cerebellum and psychiatric disorders. Front Public Health 3:66. https://doi.org/10.3389/fpubh.2015.00066
Pinzone MR et al (2013) LPS and HIV gp120 modulate monocyte/macrophage CYP27B1 and CYP24A1 expression leading to vitamin D consumption and hypovitaminosis D in HIV-infected individuals. Eur Rev Med Pharmacol Sci 17:1938–1950
Puller C, Ondreka K, Haverkamp S (2011) Bipolar cells of the ground squirrel retina. J Comp Neurol 519:759–774. https://doi.org/10.1002/cne.22546
Rao CV, Farooqui M, Zhang Y, Asch AS, Yamada HY (2018) Spontaneous development of Alzheimer’s disease-associated brain pathology in a Shugoshin-1 mouse cohesinopathy model. Aging Cell 17:e12797. https://doi.org/10.1111/acel.12797
Reddy TB et al (2009) TB database: an integrated platform for tuberculosis research. Nucleic Acids Res 37:D499–D508. https://doi.org/10.1093/nar/gkn652
Renelt M, von Bohlen und Halbach V, von Bohlen und Halbach O (2014) Distribution of PCP4 protein in the forebrain of adult mice. Acta Histochem 116:1056–1061. https://doi.org/10.1016/j.acthis.2014.04.012
Rong Y, Wang T, Morgan JI (2004) Identification of candidate Purkinje cell-specific markers by gene expression profiling in wild-type and pcd(3J) mice. Brain Res Mol Brain Res 132:128–145. https://doi.org/10.1016/j.molbrainres.2004.10.015
Rong Y et al (2012) Comparison of Cbln1 and Cbln2 functions using transgenic and knockout mice. J Neurochem 120:528–540. https://doi.org/10.1111/j.1471-4159.2011.07604.x
Sanfilippo C, Castrogiovanni P, Imbesi R, Di Rosa M (2020) CHI3L2 expression levels are correlated with AIF1, PECAM1, and CALB1 in the brains of Alzheimer’s disease patients J Mol Neurosci. https://doi.org/10.1007/s12031-020-01667-9
Sanfilippo C et al (2019a) Sex difference in CHI3L1 expression levels in human brain aging and in Alzheimer’s disease. Brain Res 1720:146305. https://doi.org/10.1016/j.brainres.2019.146305
Sanfilippo C et al (2019b) Middle-aged healthy women and Alzheimer’s disease patients present an overlapping of brain cell transcriptional profile. Neuroscience 406:333–344. https://doi.org/10.1016/j.neuroscience.2019.03.008
Sanfilippo C et al (2017a) CHI3L1 and CHI3L2 overexpression in motor cortex and spinal cord of sALS patients. Mol Cell Neurosci 85:162–169. https://doi.org/10.1016/j.mcn.2017.10.001
Sanfilippo C, Malaguarnera L, Di Rosa M (2016) Chitinase expression in Alzheimer’s disease and non-demented brains regions. J Neurol Sci 369:242–249. https://doi.org/10.1016/j.jns.2016.08.029
Sanfilippo C, Nunnari G, Calcagno A, Malaguarnera L, Blennow K, Zetterberg H, Di Rosa M (2017b) The chitinases expression is related to Simian Immunodeficiency Virus Encephalitis (SIVE) and in HIV encephalitis (HIVE). Virus Res 227:220–230. https://doi.org/10.1016/j.virusres.2016.10.012
Sanfilippo C et al (2018) OAS gene family expression is associated with HIV-related neurocognitive disorders. Mol Neurobiol 55:1905–1914. https://doi.org/10.1007/s12035-017-0460-3
Scarpino M et al (2013) Kidney disease in HIV-infected patients. Eur Rev Med Pharmacol Sci 17:2660–2667
Schafer CB, Hoebeek FE (2018) Convergence of primary sensory cortex and cerebellar nuclei pathways in the whisker system. Neuroscience 368:229–239. https://doi.org/10.1016/j.neuroscience.2017.07.036
Schmahmann JD, Caplan D (2006) Cognition, emotion and the cerebellum. Brain J Neurol 129:290–292. https://doi.org/10.1093/brain/awh729
Schmahmann JD, Pandya DN (1997) The cerebrocerebellar system. Int Rev Neurobiol 41:31–60
Scoles DR et al (2017) Antisense oligonucleotide therapy for spinocerebellar ataxia type 2. Nature 544:362–366. https://doi.org/10.1038/nature22044
Seigneur E, Sudhof TC (2017) Cerebellins are differentially expressed in selective subsets of neurons throughout the brain. J Comp Neurol 525:3286–3311. https://doi.org/10.1002/cne.24278
Setti SE, Hunsberger HC, Reed MN (2017) Alterations in hippocampal activity and Alzheimer’s disease. Transl Issues Psychol Sci 3:348–356. https://doi.org/10.1037/tps0000124
Skold K et al (2006) Decreased striatal levels of PEP-19 following MPTP lesion in the mouse. J Proteome Res 5:262–269. https://doi.org/10.1021/pr050281f
Slemmon JR, Danho W, Hempstead JL, Morgan JI (1985) Cerebellin: a quantifiable marker for Purkinje cell maturation. Proc Natl Acad Sci U S A 82:7145–7148. https://doi.org/10.1073/pnas.82.20.7145
Slemmon JR, Feng B, Erhardt JA (2000) Small proteins that modulate calmodulin-dependent signal transduction: effects of PEP-19, neuromodulin, and neurogranin on enzyme activation and cellular homeostasis. Mol Neurobiol 22:99–113. https://doi.org/10.1385/MN:22:1-3:099
Smyth GK (2004) Linear models and empirical Bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol 3:3. https://doi.org/10.2202/1544-6115.1027
Soudry Y, Lemogne C, Malinvaud D, Consoli SM, Bonfils P (2011) Olfactory system and emotion: common substrates. Eur Ann Otorhinolaryngol Head Neck Dis 128:18–23. https://doi.org/10.1016/j.anorl.2010.09.007
Stein JF, Glickstein M (1992) Role of the cerebellum in visual guidance of movement. Physiol Rev 72:967–1017. https://doi.org/10.1152/physrev.1992.72.4.967
Strata P (2015) The emotional cerebellum. Cerebellum 14:570–577. https://doi.org/10.1007/s12311-015-0649-9
Thul PJ, Åkesson L, Wiking M, Mahdessian D, Geladaki A, Ait Blal H, Alm T, Asplund A, Björk L, Breckels LM, Bäckström A, Danielsson F, Fagerberg L, Fall J, Gatto L, Gnann C, Hober S, Hjelmare M, Johansson F, Lee S, Lindskog C, Mulder J, Mulvey CM, Nilsson P, Oksvold P, Rockberg J, Schutten R, Schwenk JM, Sivertsson Å, Sjöstedt E, Skogs M, Stadler C, Sullivan DP, Tegel H, Winsnes C, Zhang C, Zwahlen M, Mardinoglu A, Pontén F, von Feilitzen K, Lilley KS, Uhlén M, Lundberg E (2017) A subcellular map of the human proteome. Science 356. https://doi.org/10.1126/science.aal3321
Tiao GEPBGC (1992) Bayesian inference in statistical analysis. https://doi.org/10.1002/9781118033197
Trabzuni D et al (2013) Widespread sex differences in gene expression and splicing in the adult human brain. Nat Commun 4:2771. https://doi.org/10.1038/ncomms3771
Uhlen M, Fagerberg L, Hallstrom BM, Lindskog C, Oksvold P, Mardinoglu A, Sivertsson A, Kampf C, Sjostedt E, Asplund A, Olsson I, Edlund K, Lundberg E, Navani S, Szigyarto CAK, Odeberg J, Djureinovic D, Takanen JO, Hober S, Alm T, Edqvist PH, Berling H, Tegel H, Mulder J, Rockberg J, Nilsson P, Schwenk JM, Hamsten M, von Feilitzen K, Forsberg M, Persson L, Johansson F, Zwahlen M, von Heijne G, Nielsen J, Ponten F (2015) Proteomics. Tissue-based map of the human proteome. Science 347:1260419. https://doi.org/10.1126/science.1260419
Uhlen M, Zhang C, Lee S, Sjöstedt E, Fagerberg L, Bidkhori G, Benfeitas R, Arif M, Liu Z, Edfors F, Sanli K, von Feilitzen K, Oksvold P, Lundberg E, Hober S, Nilsson P, Mattsson J, Schwenk JM, Brunnström H, Glimelius B, Sjöblom T, Edqvist PH, Djureinovic D, Micke P, Lindskog C, Mardinoglu A, Ponten F (2017) A pathology atlas of the human cancer transcriptome. Science 357. https://doi.org/10.1126/science.aan2507
Utal AK, Stopka AL, Roy M, Coleman PD (1998) PEP-19 immunohistochemistry defines the basal ganglia and associated structures in the adult human brain, and is dramatically reduced in Huntington’s disease. Neuroscience 86:1055–1063. https://doi.org/10.1016/s0306-4522(98)00130-4
Vasavada MM, Martinez B, Wang J, Eslinger PJ, Gill DJ, Sun X, Karunanayaka P, Yang QX (2017) Central olfactory dysfunction in Alzheimer’s disease and mild cognitive impairment: a functional MRI study. J Alzheimers Dis 59:359–368. https://doi.org/10.3233/JAD-170310
Wang J, Coombes KR, Highsmith WE, Keating MJ, Abruzzo LV (2004) Differences in gene expression between B-cell chronic lymphocytic leukemia and normal B cells: a meta-analysis of three microarray studies. Bioinformatics 20:3166–3178. https://doi.org/10.1093/bioinformatics/bth381
Willard FS, McCudden CR, Siderovski DP (2006) G-protein alpha subunit interaction and guanine nucleotide dissociation inhibitor activity of the dual GoLoco motif protein PCP-2 (Purkinje cell protein-2). Cell Signal 18:1226–1234. https://doi.org/10.1016/j.cellsig.2005.10.003
Xia X, Jiang Q, McDermott J, Han JJ (2018) Aging and Alzheimer’s disease: comparison and associations from molecular to system level. Aging Cell 17:e12802. https://doi.org/10.1111/acel.12802
Xu J, Patassini S, Rustogi N, Riba-Garcia I, Hale BD, Phillips AM, Waldvogel H, Haines R, Bradbury P, Stevens A, Faull RLM, Dowsey AW, Cooper GJS, Unwin RD (2019) Regional protein expression in human Alzheimer’s brain correlates with disease severity. Commun Biol 2:43. https://doi.org/10.1038/s42003-018-0254-9
Yasrebi H, Sperisen P, Praz V, Bucher P (2009) Can survival prediction be improved by merging gene expression data sets? PLoS One 4:e7431. https://doi.org/10.1371/journal.pone.0007431
Yeganeh-Doost P, Gruber O, Falkai P, Schmitt A (2011) The role of the cerebellum in schizophrenia: from cognition to molecular pathways. Clinics 66(Suppl 1):71–77
Zhang B et al (2013) Integrated systems approach identifies genetic nodes and networks in late-onset Alzheimer’s disease. Cell 153:707–720. https://doi.org/10.1016/j.cell.2013.03.030
Zhang D, Aravind L (2012) Novel transglutaminase-like peptidase and C2 domains elucidate the structure, biogenesis and evolution of the ciliary compartment. Cell Cycle 11:3861–3875. https://doi.org/10.4161/cc.22068
Acknowledgments
We would like to show our gratitude to the authors of microarray datasets (Table 1) made available online, for consultation and re-analysis. We also want to thank the Sigur Rós band for realizing the song “hoppipolla,” an inspiration during the manuscript writing.
Funding
This work was supported by the University Research Project Grant (Triennial Research Plan 2020–2022), Department of Biomedical and Biotechnological Sciences (BIOMETEC), University of Catania, Italy.
Author information
Authors and Affiliations
Contributions
Data curation, project administration, and software, M.D.; formal analysis, methodology, and writing of original draft, M.D. and C.S.; investigation, C.S.; resources, G.M. and V.M.; supervision, M.D. and G.M.; validation, P.C.; visualization, V.M. and M.K.; writing, reviewing, and editing, G.M. and R.I.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Figure S1
GNG13, PCP2, PCP4 and CBLN3 expression levels in cerebellar cortex and in cerebellum. Basal expression levels in healthy control subjects showed that GNG13, PCP2, PCP4 and CBLN3 expression levels were significantly higher in the cerebellar cortex than in the cerebellum. Data are expressed as z-score intensity expression levels and presented as violin dot plots. p values <0.05 were considered to be statistically significant (**p < 0.005; ****p < 0.00005). (PNG 732 kb)
Table s1
Anova results. (PDF 404 kb)
Rights and permissions
About this article
Cite this article
Sanfilippo, C., Musumeci, G., Kazakova, M. et al. GNG13 Is a Potential Marker of the State of Health of Alzheimer’s Disease Patients’ Cerebellum. J Mol Neurosci 71, 1046–1060 (2021). https://doi.org/10.1007/s12031-020-01726-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-020-01726-1