Abstract
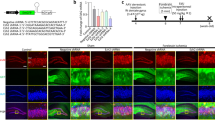
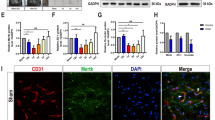
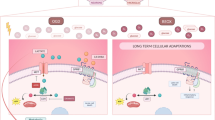
Nuclear factor of activated T cells (NFAT) is a multifunctional cytokine family. NFAT5 was recently reported to be involved in many neuronal functions, but its specific function remains unclear. In this study, our aim is to investigate whether NFAT5 overexpression can protect astrocytes against oxygen–glucose–serum deprivation/restoration (OGSD/R) damage. In vivo, rats were subjected to ischemia–reperfusion injury, resulting in increased water content, infarct volume, and expression of NFAT5 protein in rat spinal cord. After primary culture for spinal cord astrocytes, the in vitro OGSD/R model was established. The results of the CCK8 assay and flow cytometry showed that, in the OGSD/R group, astrocyte cell viability was downregulated, but astrocyte apoptosis increased. Caspase 3 activity increased as well. Levels of NFAT5, as detected by real-time quantitative PCR and western blot, decreased under OGSD/R, as did SIRT1. Commercial kits for activity assays were used to show that OGSD/R inhibited SIRT1 activation but accelerated SOD activation after OGSD/R. Next, pcDNA-NFAT5 or NFAT5 siRNA was transfected into astrocytes. Overexpression of NFAT5 not only promoted the survival of the astrocytes and SIRT1 activation under OGSD/R but also inhibited cell apoptosis and SOD activation. Moreover, overexpression of NFAT5 apparently diminished histone acetylation and promoted the nuclear transport of Nrf2. Our results show that NFAT5 protects spinal astrocytes in a manner that depends on activation of the SIRT1/Nrf2 pathway. These findings present a novel potential molecular mechanism for NFAT5 therapy in the context of spinal cord injury.
Access this article
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.





Similar content being viewed by others
Abbreviations
- NFAT:
-
Nuclear factor of activated T cells
- OGSD/R:
-
Oxygen–glucose–serum deprivation/restoration
- SCI:
-
Spinal cord injury
- I/R:
-
Ischemia–reperfusion
- SIRT1:
-
Sirtuin 1
- HSP70:
-
Heat shock protein 70
- HO-1:
-
Hemeoxygenase1
- Nrf2:
-
Nuclear factor-E2 related factor 2
- Keap1:
-
Kelch-like ECH-associated protein 1
- TTC:
-
2,3,5-triphenyltetrazolium chloride
- siRNA:
-
Small interfering RNA
- GFAP:
-
Glial fibrillary acid protein
References
Arockiaraj J, Palanisamy R, Bhatt P, Kumaresan V, Gnanam AJ, Pasupuleti M, Kasi M (2014) A novel murrel Channa striatus mitochondrial manganese superoxide dismutase: gene silencing, SOD activity, superoxide anion production and expression. Fish Physiol Biochem 40:1937–1955
Cambria RP et al (1997) Clinical experience with epidural cooling for spinal cord protection during thoracic and thoracoabdominal aneurysm repair. J Vasc Surg 25:241–243
Cheung CY, Ko BC (2013) NFAT5 in cellular adaptation to hypertonic stress—regulations and functional significance. J Mol Signal 8:1–9
Chi Z, Ma X, Cui G, Li M, Li F (2013) Cinnamtannin B-1 regulates cell proliferation of spinal cord astrocytes and protects the cell from oxygen-glucose-serum deprivation/reoxygenation-induced apoptosis. Int J Mol Sci 14:15827–15837
Choi DW (1988) Glutamate neurotoxicity and diseases of the nervous system. Neuron 1:623–634
Choi IS, Rickert E, Olefsky J, Webster N (2012) Reproductive effects of SirT1 expression in astrocytes. FASEB J 26:762.715
Chong ZZ, Shang YC, Wang S, Maiese K (2012) SIRT1: new avenues of discovery for disorders of oxidative stress. Expert Opin Ther Targets 16:167–178
Ding Y-W et al (2016) SIRT1 exerts protective effects against paraquat-induced injury in mouse type II alveolar epithelial cells by deacetylating NRF2 in vitro. Int J Mol Med 37:1049–1058
Fan L, Wang K, Shi Z, Die J, Wang C, Dang X (2011) Tetramethylpyrazine protects spinal cord and reduces inflammation in a rat model of spinal cord ischemia-reperfusion injury. J Vasc Surg 54:192–200
Huang K et al (2013) Sirt1 resists advanced glycation end products-induced expressions of fibronectin and TGF- β 1 by activating the Nrf2/ARE pathway in glomerular mesangial cells. Free Radic Biol Med 65:528–540
Jayanthi S, Deng X, Ladenheim B, Mccoy MT, Cai NS, Cadet JL (2005) Calcineurin/NFAT-induced up-regulation of the Fas ligand/Fas death pathway is involved in methamphetamine-induced neuronal apoptosis. Proc Natl Acad Sci U S A 102:868–873
Jia YY et al (2014) The involvement of NFAT transcriptional activity suppression in SIRT1-mediated inhibition of COX-2 expression induced by PMA/Ionomycin. PLoS One 9:57–57
Jin Y, Miao W, Lin X, Pan X, Ye Y, Xu M, Fu Z (2014) Acute exposure to 3-methylcholanthrene induces hepatic oxidative stress via activation of the Nrf2/ARE signaling pathway in mice. Environ Toxicol 29:1399–1408
Kim MS, Shutov LP, Gnanasekaran A, Lin Z, Rysted JE, Ulrich JD, Usachev YM (2014) Nerve growth factor (NGF) regulates activity of nuclear factor of activated T-cells (NFAT) in neurons via the phosphatidylinositol 3-kinase (PI3K)-Akt-glycogen synthase kinase 3β (GSK3β) pathway. J Biol Chem 289:31349–31360
Kwon HS, Ott M (2008) The ups and downs of SIRT1. Trends Biochem Sci 33:517–525
Li XQ, Bo F, Tan WF, Wang ZL, Sun XJ, Zhang ZL, Hong M (2016) miR-320a affects spinal cord edema through negatively regulating aquaporin-1 of blood–spinal cord barrier during bimodal stage after ischemia reperfusion injury in rats. BMC Neurosci 17:1–14
Mak M, Lo A, Lam A, Chung S, Chung S (2006) NFAT5 deficiency increases the severity of neuronal cell death in ischemic. Annual Meeting of the Society for Neuroscience
Mak KM, Lo AC, Lam AK, Yeung PK, Ko BC, Chung SS, Chung SK (2012) Nuclear factor of activated T cells 5 deficiency increases the severity of neuronal cell death in ischemic injury. Neurosignals 20:237–251
Quadri S, Siragy HM (2014) Regulation of (pro) renin receptor expression in mIMCD via the GSK-3β-NFAT5-SIRT-1 signaling pathway. Am J Physiol Ren Physiol 307:F593–F600
Rodrigo R, Libuy M, Feli F, Hasson D (2013) Oxidative stress-related biomarkers in essential hypertension and ischemia-reperfusion myocardial damage. Dis Markers 35:773–790
Sahu BD, Mahesh KJ, Sistla R (2015) Baicalein, a bioflavonoid, prevents cisplatin-induced acute kidney injury by up-regulating antioxidant defenses and down-regulating the MAPKs and NF-kB pathways. PLoS One 10:e0134139
Schwartz N, Schohl A, Ruthazer ES (2009) Neural activity regulates synaptic properties and dendritic structure in vivo through calcineurin/NFAT signaling. Neuron 62:655–669
Shao H et al (2014) Spinal SIRT1 activation attenuates neuropathic pain in mice. PLoS One 9:e100938
Shioda N, Han F, Moriguchi S, Fukunaga K (2007) Constitutively active calcineurin mediates delayed neuronal death through Fas-ligand expression via activation of NFAT and FKHR transcriptional activities in mouse brain ischemia. J Neurochem 102:1506–1517
Sotoudeh A, Jahanshahi A, Zareiy S, Darvishi M, Roodbari N, Bazzazan A (2015) The influence of low-level laser irradiation on spinal cord injuries following ischemia- reperfusion in rats. Acta Cir Bras 30:611–616
Straley KS, Foo CW, Heilshorn SC (2010) Biomaterial design strategies for the treatment of spinal cord injuries. J Neurotrauma 27:1–19
Thuret S, Moon LD, Gage FH (2006) Therapeutic interventions after spinal cord injury. Nat Rev Neurosci 7:628–643
Timucin AC, Bodur C, Basaga H (2015) SIRT1 contributes to aldose reductase expression through modulating NFAT5 under osmotic stress: In vitro and in silico insights. Cell Signal 27:2160–2172
Vashishta A, Habas A, Pruunsild P, Zheng JJ, Timmusk T, Hetman M (2009) Nuclear factor of activated T-cells isoform c4 (NFATc4/NFAT3) as a mediator of antiapoptotic transcription in NMDA receptor-stimulated cortical neurons. J Neurosci 29:15331–15340
Webster BR, Lu Z, Sack MN, Scott I (2011) The role of sirtuins in modulating redox stressors. Free Radic Biol Med 52:281–290
Wong CH, Crack PJ (2008) Modulation of neuro-inflammation and vascular response by oxidative stress following cerebral ischemia-reperfusion injury. Curr Med Chem 15:1–14
Woo S, Sang L, Kwon MH (2002a) TonEBP transcriptional activator in the cellular response to increased osmolality. Pflugers Arch - Eur J Physiol 444:579–585
Woo SK, Do Lee S, Na KY, Park WK, Kwon HM (2002b) TonEBP/NFAT5 stimulates transcription of HSP70 in response to hypertonicity. Mol Cell Biol 22:5753–5760
Xia X et al. (2016) Impact of heat shock protein A 12B overexpression on spinal astrocyte survival against oxygen-glucose-serum deprivation/restoration in primary cultured astrocytes. J Mol Neurosci 59:511–520
Yan C, Liang W, Zhao J, Zhang H, Ying T, Liang H, Qiang M (2016) Serum response factor protects retinal ganglion cells against high-glucose damage. J Mol Neurosci 59:232–240
Yi MH et al (2012) NFAT5-dependent expression of AQP4 in astrocytes. Cell Mol Neurobiol 33:223–232
Zhang A et al (2010) EIF2α and caspase-12 activation are involved in oxygen–glucose–serum deprivation/restoration-induced apoptosis of spinal cord astrocytes. South Afr For J 478:32–36
Zhang J, Shapiro M (2012) Activity-dependent transcriptional regulation of M-type (Kv7) K + channels by AKAP79/150-mediated NFAT actions. Neuron 76:1133–1146
Zhang Z-Y, Wu Y-Q, Luo H, Liu D-X (2015) The effect of nuclear factor of activated T-cells (NFAT) in kidney I/R mediated by C5a/C5aR. Int J Clin Exp Med 8:15535
Zhou YF, Li L, Feng F, Yuan H, Gao DK, Fu LA, Fei Z (2013) Osthole attenuates spinal cord ischemia–reperfusion injury through mitochondrial biogenesis–independent inhibition of mitochondrial dysfunction in rats. J Surg Res 185:805–814
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interests
The authors have declared no conflict of interest.
Additional information
Xun Xia, Bo Qu and Yun-Ming Li contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xia, X., Qu, B., Li, YM. et al. NFAT5 protects astrocytes against oxygen–glucose–serum deprivation/restoration damage via the SIRT1/Nrf2 pathway. J Mol Neurosci 61, 96–104 (2017). https://doi.org/10.1007/s12031-016-0849-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-016-0849-x