Abstract
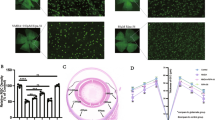
Retinal excitotoxicity is one of the major causes of retinal ganglion cell (RGC) death in glaucoma. Pituitary adenylate cyclase-activating polypeptide (PACAP) is a pleiotropic peptide with potent neuroprotective activity; however, whether it exerts such an effect in the retina and the mechanism by which RGCs are protected is still not well understood. In this study, we examined the effect of exogenous and endogenous PACAP on RGC death induced by N-methyl-d-aspartate acid (NMDA). The vitreous body of anesthetized adult male mice (C57/BL6J) was injected with NMDA (40 nmol in a 2 μL saline solution). The number of RGCs decreased from days 1 to 7 after NMDA injection, and the number of dUTP end-labeling (TUNEL)-positive cells, an indicator of cell death, peaked at day 3. However, when PACAP38 (10−8, 10−10, 10−12, 10−14, or 10−16M) was co-administered with NMDA, the 10−10M dose resulted in significantly increased RGC survival at day 7, and a decrease in the number of TUNEL-positive RGCs at day 3. We next investigated the neuroprotective effect of endogenous PACAP using PACAP heterozygote(+/−) mice. Under normal circumstances, there was no significant difference in the number of RGCs in the PACAP(+/−) mice compared with their wild-type counterparts. However, the number of RGCs significantly decreased in the PACAP(+/−) mice 7 days after NMDA injection, relative to their wild-type counterparts. The number of TUNEL-positive RGCs peaked at day 1 in the PACAP(+/−) mice. These effects in the PACAP(+/−) mice were reversed by intravitreous injection of 10−10M PACAP38. This suggests that exogenous PACAP is able to counteract NMDA-induced toxicity, and that endogenous PACAP exerts a neuroprotective effect in the retina.






Similar content being viewed by others
References
Arimura A (1998) Perspectives on pituitary adenylate cyclase activating polypeptide (PACAP) in the neuroendocrine, endocrine, and nervous systems. Jpn J Physiol 48:301–331
Arimura A, Shioda S (1995) Pituitary adenylate cyclase activating polypeptide (PACAP) and its receptors: neuroendocrine and endocrine interaction. Front Neuroendocrinol 16:53–88
Atlasz T, Szabadfi K, Reglodi D et al (2009) Effects of pituitary adenylate cyclase activating polypeptide and its fragments on retinal degeneration induced by neonatal monosodium glutamate treatment. Ann NY Acad Sci 1163:348–352
Atlasz T, Szabadfi K, Kiss P et al (2010) Evaluation of the protective effects of PACAP with cell-specific marlers in ischemia-induced retinal degeneration. Brain Res Bull 81:497–504
Brooks DE, Garcia GA, Dreyer EB, Zurakowski D, Franco-Bourland RE (1997) Vitreous body glutamate concentration in dogs with glaucoma. Am J Vet Res 58:864–867
Dkhissi O, Chanut E, Wasowicz M, Savoldelli M, Nguyen-Legros J, Minvielle F, Versaux-Botteri C (1999) Retinal TUNEL-positive cells and high glutamate levels in vitreous humor of mutant quail with a glaucoma-like disorder. Invest Ophthalmol Vis Sci 40:990–995
Hare WA, Wheeler L (2009) Experimental glutamatergic excitotoxicity in rabbit retinal ganglion cells: block by memantine. Invest Ophthalmol Vis Sci 50:2940–2948
Hashimoto H, Shintani N, Tanaka K et al (2001) Altered psychomotor behaviors in mice lacking pituitary adenylate cyclase-activating polypeptide (PACAP). Proc Natl Acad Sci USA 98:13355–13360
Kido N, Tanihara H, Honjo M, Inatani M, Tatsuno T, Nakayama C, Honda Y (2000) Neuroprotective effects of brain-derived neurotrophic factor in eyes with NMDA-induced neuronal death. Brain Res 884:59–67
Lam TT, Abler AS, Kwong JM, Tso MO (1999) N-methyl-d-aspartate (NMDA)-induced apoptosis in rat retina. Invest Ophthalmol Vis Sci 40:2391–2397
Li M, David C, Kikuta T, Somogyvari-Vigh A, Arimura A (2005) Signaling cascades involved in neuroprotection by subpicomolar pituitary adenylate cyclase-activating polypeptide 38. J Mol Neurosci 27:91–105
Liu GJ, Madsen BW (1997) PACAP38 modulates activity of NMDA receptors in cultured chick cortical neurons. J Neurophysiol 78:2231–2234
Nakamachi T, Endo S, Ohtaki H, Yin L, Kenji D, Kudo Y, Funahashi H, Matsuda K, Shioda S (2005) Orexin-1 receptor expression after global ischemia in mice. Regul Pept 126:49–54
Nakamachi T, Li M, Shioda S, Arimura A (2006) Signaling involved in pituitary adenylate cyclase-activating polypeptide-stimulated ADNP expression. Peptides 27:1859–1864
Ohtaki H, Nakamachi T, Dohi K, Aizawa Y, Takaki A, Hodoyama K, Yofu S, Hashimoto H, Shintani N, Baba A, Kopf M, Iwakura Y, Matsuda K, Arimura A, Shioda S (2006) Pituitary adenylate cyclase-activating polypeptide (PACAP) decreases ischemic neuronal cell death in association with IL-6. Proc Natl Acad Sci USA 103:7488–7493
Quigley HA (1995) Ganglion cell death in glaucoma: pathology recapitulates ontogeny. Aust N Z J Ophthalmol 23:85–91
Racz B, Gallyas F Jr, Kiss P et al (2006) The neuroprotective effects of PACAP in monosodium glutamate-induced retinal lesion involve inhibition of proapoptotic signaling pathways. Regul Pept 137:20–26
Racz B, Gallyas F Jr, Kiss P et al (2007) Effects of pituitary adenylate cyclase activating polypeptide (PACAP) on the PKA-BAD-14-3-3 signaling pathway in glutamate-induced retinal injury in neonatal rat. Neurotox Res 12(2):95–104
Seki T, Shioda S, Ogino D, Nakai Y, Arimura A, Koide R (1997) Distribution and ultrastructural localization of a receptor for pituitary adenylate cyclase activating polypeptide and its mRNA in the rat retina. Neurosci Lett 238:127–130
Seki T, Shioda S, Nakai Y, Arimura A, Koide R (1998) Distribution and ultrastructural localization of pituitary adenylate cyclase-activating polypeptide (PACAP) and its receptor in the rat retina. Ann NY Acad Sci 865:408–411
Seki T, Izumi S, Shioda S, Zhou CJ, Arimura A, Koide R (2000a) Gene expression for PACAP receptor mRNA in the rat retina by in situ hybridization and in situ RT-PCR. Ann NY Acad Sci 921:366–369
Seki T, Shioda S, Izumi S, Arimura A, Koide R (2000b) Electron microscopic observation of pituitary adenylate cyclase-activating polypeptide (PACAP)-containing neurons in the rat retina. Peptides 21:109–113
Seki T, Nakatani M, Taki C, Shinohara Y, Ozawa M, Nishimura S, Ito H, Shioda S (2006) Neuroprotective effect of PACAP against kainic acid-induced neurotoxicity in rat retina. Ann NY Acad Sci 1070:531–534
Seki T, Itoh H, Nakamachi T, Shioda S (2008) Suppression of ganglion cell death by PACAP following optic nerve transection in the rat. J Mol Neurosci 36:57–60
Seki T, Itoh H, Nakamachi T, Endo K, Waday, Nakamura K, Shioda S (2010) Suppression of rat retinal ganglion cell death by PACAP following transient ischemia induced by high intraocular pressure. J Mol Neurosci (in press)
Sherwood NM, Krueckl SL, McRory JE (2000) The origin and function of the pituitary adenylate cyclase-activating polypeptide (PACAP)/glucagon superfamily. Endocr Rev 21:619–670
Shintani N, Suetake S, Hashimoto H, Koga K, Kasai A, Kawaguchi C, Morita Y, Hirose M, Sakai Y, Tomimoto S, Matsuda T, Baba A (2005) Neuroprotective action of endogenous PACAP in cultured rat cortical neurons. Regul Pept 126:123–128
Shioda S, Ohtaki H, Nakamachi T, Dohi K, Watanabe J, Nakajo S, Arata S, Kitamura S, Okuda H, Takenoya F, Kitamura Y (2006) Pleiotropic functions of PACAP in the CNS: neuroprotection and neurodevelopment. Ann NY Acad Sci 1070:550–560
Sucher NJ, Aizenman E, Lipton SA (1991) N-methyl-D-aspartate antagonists prevent kainate neurotoxicity in rat retinal ganglion cells in vitro. J Neurosci 11:966–971
Tamas A, Gabriel R, Racz B, Denes V, Kiss P, Lubics A, Lengvari I, Reglodi D (2004) Effects of pituitary adenylate cyclase activating polypeptide in retinal degeneration induced by monosodium-glutamate. Neurosci Lett 372:110–113
Teuchner B, Dimmer A, Humpel C et al (2010) VIP, PACAP-38, BDNF and ADNP in NMDA-induced excitotoxicity in the rat retina. Acta Ophthalmol (in press)
Uchida D, Arimura A, Somogyvari-Vigh A, Shioda S, Banks WA (1996) Prevention of ischemia-induced death of hippocampal neurons by pituitary adenylate cyclase activating polypeptide. Brain Res 736:280–286
Vaudry D, Gonzalez BJ, Basille M, Pamantung TF, Fournier A, Vaudry H (2000) PACAP acts as a neurotrophic factor during histogenesis of the rat cerebellar cortex. Ann NY Acad Sci 921:293–299
Vaudry D, Hamelink C, Damadzic R, Eskay RL, Gonzalez B, Eiden LE (2005) Endogenous PACAP acts as a stress response peptide to protect cerebellar neurons from ethanol or oxidative insult. Peptides 26:2518–2524
Varga B, Szabadfi K, Kiss P et al (2010) PACAP Improves Functional Outcome in Excitotoxic Retinal Lesion: An Electroretinographic Study. J Mol Neurosci. (in press)
Yaka R, He DY, Phamluong K, Ron D (2003) Pituitary adenylate cyclase-activating polypeptide (PACAP (1–38)) enhances N-methyl-d-aspartate receptor function and brain-derived neurotrophic factor expression via RACK1. J Biol Chem 278:9630–9638
Acknowledgment
This work was supported by the Ministry of Education, Science, Sports and Culture of Japan and in part by a High-Technology Research Center Project from the Ministry of Education, Science, Sports and Culture of Japan (T.S., T.N., S.S.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Endo, K., Nakamachi, T., Seki, T. et al. Neuroprotective Effect of PACAP Against NMDA-Induced Retinal Damage in the Mouse. J Mol Neurosci 43, 22–29 (2011). https://doi.org/10.1007/s12031-010-9434-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-010-9434-x