Abstract
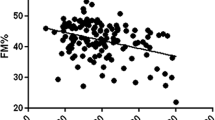
To investigate the role of pro- and anti-inflammatory adipokines in the bone metabolism of non-alcoholic fatty liver disease (NAFLD) obese adolescents as well as the effects of long-term interdisciplinary therapy on metabolic-related risk factors. Forty post-puberty obese adolescents were randomly assigned into two groups: (1) NAFLD group and (2) non-NAFLD group (diagnosis by ultrasonography) and submitted to a weight loss therapy. Body composition was analyzed by air displacement plethysmography, bone mineral density (BMD) and content by dual-energy X-ray absorptiometry, blood samples were collected to measure lipid profile, hepatic enzymes, and adipokines. Leptin and adiponectin concentrations were measured by ELISA. A decrease in total body mass, BMI, body fat, visceral and subcutaneous fat, insulin concentration, HOMA-IR, total cholesterol and an increase in lean body mass were observed in both groups after therapy. It was found positive correlation between the Δ BMD and the Δ fat mass (%) (r = 0.31, P = 0.01) and negative correlations between Δ BMC with Δ HOMA-IR (r = −0.34, P = 0.02) and Δ HOMA-IR with Δ leptin (r = −0.34, P = 0.02). In addition, increased levels of adiponectin and reduction in leptin concentrations were observed in NAFLD group. In the simple regression analysis, the HOMA-IR was an independent predictor changes in BMC in total obese adolescents and in the non-NAFLD group. One year of interdisciplinary weight loss therapy for obese adolescents with or without NAFLD, could regulate bone mineral metabolism as result of an increased BMC and improved inflammatory state.


Similar content being viewed by others
References
N.K. Pollock, P.J. Bernard, B. Gutin, C.L. Davis, H. Zhu, Y. Dong, Adolescent obesity, bone mass and cardiometabolic risk factors. J. Pediatr. 158, 727–734 (2011)
L.E. Polgreen, Keeping a broad approach to managing pediatric bone disease. J. Pediatr. Gastroenterol. Nutr. 51, 128–129 (2010)
P. Dimitri, N. Bishop, J.S. Walsh, R. Eastell, Obesity is a risk factor for fracture in children but is protective against fracture in adults: a paradox. Bone 50(2), 457–466 (2011)
M.B. Leonard, J. Shults, B.A. Wilson, A.M. Tershakovec, B.S. Zemel, Obesity during childhood and adolescence augments bone mass and bone dimensions. Am. J. Clin. Nutr. 80, 514–523 (2002)
E. Rocher, C. Chappard, C. Jaffre, C.L. Benhamou, D. Courteix, Bone mineral density in prepubertal obese and control children: relation to body weight, lean mass, and fat mass. J. Bone Miner. Metab. 26, 73–78 (2008)
D.K. Hwang, H.J. Choi, The relationship between low bone mass and metabolic syndrome in Korean women. Osteoporos. Int. 21, 425–431 (2010)
W.L. do Prado, A. de Piano, M. Lazaretti-Castro, M.T. de Mello, S.G. Stella, S. Tufik et al., Relationship between bone mineral density, leptin and insulin concentration in Brazilian obese adolescents. J. Bone Miner. Metab. 27, 613–619 (2009)
A. de Piano, L. Tock, J. Carnier, D. Foschini, P.L. Sanches, F.A. Corrêa et al., The role of nutritional profile in the orexigenic neuropeptide secretion in nonalcoholic fatty liver disease obese adolescents. Eur. J. Gastroenterol. Hepatol. 22, 557–563 (2010)
A. de Piano, W.L. Prado, D.A. Caranti, K.O. Siqueira, S.G. Stella, M. Lofrano et al., Metabolic and nutritional profile of obese adolescents with nonalcoholic fatty liver disease. J. Pediatr. Gastroenterol. Nutr. 44, 446–452 (2007)
A. de Piano, W.L. Prado, D.A. Caranti, K.O. Siqueira, S.G. Stella, M. Lofrano et al., Relationship between nonalcoholic fatty liver disease prevalence and visceral fat in obese adolescents. Dig. Liver Dis. 40, 132–139 (2008)
R. Wang, Q. Lu, J. Feng, F. Yin, C. Qin, B. Liu, Coexistence of non-alcoholic fatty liver disease with elevated alanine aminotransferase is associated with insulin resistance in young Han males. Endocrine 41(1), 70–75 (2011)
E.A. Roberts, Pediatric nonalcoholic fatty liver disease (NAFLD): a “growing” problem? J. Hepatol. 46, 113–142 (2007)
E. Scorletti, P.C. Calder, C.D. Byrne, Non-alcoholic fatty liver disease and cardiovascular risk: metabolic aspects and novel treatments. Endocrine 40(3), 332–343 (2011)
O. Pirgon, H. Bilgin, I. Tolu, D. Odabas, Correlation of insulin sensitivity with bone mineral status in obese adolescents with non-alcoholic fatty liver disease. Clin. Endocrinol. (Oxf) 75, 189–195 (2011)
I. Tasci, G. Erdem, A. Sonmez, T. Dogru, C.N. Ercin, Hepatic steatosis, visceral adiposity, insulin resistance, adiponectin, and inflammation. Metabolism 58, 141 (2009)
V. Nobili, A. Reale, A. Alisi, G. Morino, I. Trenta, M. Pisani et al., Elevated serum ALT in children presenting to the emergency unit: relationship with NAFLD Dig. Liver Dis. 41, 749–752 (2009)
D.A. Rubin, R.G. McMurray, J.S. Harrell, A.C. Hackney, D.E. Thorpe, A.M. Haqq, The association between insulin resistance and cytokines in adolescents: the role of weight status and exercise. Metabolism 57, 683–690 (2008)
T. Yatagai, S. Nagasaka, A. Taniguchi, M. Fukushima, T. Nakamura, A. Kuroe, Y. Nakai, S. Ishibashi, Hypoadiponectinemia is associated with visceral fat accumulation and insulin resistance in Japanese men with type 2 diabetes mellitus. Metabolism 52, 1274–1278 (2003)
A.R. de Dâmaso, A. Piano, P.L. Sanches, F. Corgosinho, L. Tock, L.M. Oyama et al., Hyperleptinemia in obese adolescents deregulates neuropeptides during weight loss. Peptides 32, 1384–1391 (2011)
S. Blüher, C.S. Mantzoros, Leptin in humans: lessons from translational research. Am. J. Clin. Nutr. 89, 991S–997S (2009)
M.H. Tschöp, D.Y. Hui, T.L. Horvath, Diet-induced leptin resistance: the heart of the matter. Endocrinology 148, 921–923 (2007)
G.D. Norata, S. Raselli, L. Grigore, K. Garlaschelli, E. Dozio, P. Magni, A.L. Catapano, Leptin: adiponectin ratio is an independent predictor of intima media thickness of the common carotid artery. Stroke 38, 2844–2846 (2007)
J.M. Tanner, R.H. Whithouse, Clinical longitudinal standards for height, weight velocity and stages of puberty. Arch. Dis. Child. 51, 170–179 (1976)
B. Gutin, L. Ramsey, P. Barbeau, W. Cannady, M. Ferguson, M. Litaker et al., Plasma leptin concentrations in obese children: changes during 4-mo periods with and without physical training. Am. J. Clin. Nutr. 69, 388–394 (1996)
J.B. Schwimmer, R. Deutsch, J.B. Rauch, C. Behling, R. Newbury, J.E. Lavine, Obesity, insulin resistance, and other clinicopathological correlates of pediatrics nonalcoholic fatty liver disease. J. Pediatr. 143, 500–505 (2003)
E. Black, L. Petersen, M. Kreutzer, S. Toubro, T.I. Sørensen, O. Pedersen et al., Fat mass measured by DXA varies with scan mode. Obes. Res. 10, 69–77 (2002)
F.F. Ribeiro-Filho, A.N. Faria, S. Azjen, M.T. Zanella, S.R. Ferreira, Methods of estimation of visceral fat: advantages of ultrasonography. Obes. Res. 11, 1488–1494 (2003)
N. Sabir, Y. Sermez, S. Kazil, M. Zencir, Correlation of abdominal fat accumulation and liver steatosis: importance of ultrasonographic and anthropometric measurements. Eur. J. Ultrasound. 14, 121–128 (2001)
S. Saadeh, Z.M. Younossi, E.M. Remer, T. Gramlich, J.P. Ong, M. Hurley et al., The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 123, 745–750 (2002)
S.P. Murphy, M.I. Poos, Dietary reference intakes: summary of applications in dietary assessment. Public Health Nutr. 5, 843–849 (2002)
A.R. Dâmaso, A. de Piano, L. Tock et al., Nutritional and clinical strategies on the prevention and treatment of NAFLD and metabolic syndrome, in Nutrition, diet therapy and the liver, 1st edn., ed. by V.R. Preedy, R. Srirajaskanthan, R. Lakshman, R.R. Watson (Taylor & Francis Group, Boca Raton, 2009), pp. 113–130
C.E.Garber, B. Blissmer, M.R. Deschenes, B.A. Franklin, M.J. Lamonte, I.M. Lee et al., American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med. Sci. Sports Exerc. 43, 1334–1359 (2011)
W.J. Kraemer, M.S. Fragala, Personalize it: program design in resistance training. ACSM’S Health Fit. J. 10, 7–17 (2006)
S. Freitas, C.S. Lopes, W. Coutinho, J.C. Appolianario, Tradução e adaptação para o português da Escala de Compulsão Alimentar Periódica. Rev. Bras. Psiquiatr. 23, 215–220 (2001)
T.A. Cordás, P.B. Hochgraf, O ‘‘BITE’’ Instrumento para avaliação da Bulimia nervosa: Versão para o português. J. Bras. Psiquiatr. 42, 141–144 (1993)
J. Gormally, S. Black, S. Daston, D. Rardin, The assessment of binge eating severity among obese persons. Addict. Behav. 7, 47–55 (1982)
M. Henderson, C.P.L. Freeman, A self-rating scale for bulimia. The BITE. Br. J. Psychiatry 50, 18–24 (1987)
P. Isnard, G. Michel, M.L. Frelut, G. Vila, B. Falissard, W. Naja, Binge eating and psychopathology in severely obese adolescents. Int. J. Eat. Disord. 34, 235–243 (2003)
C.T. Rubin, L.E. Lanyon, Regulation of bone mass by mechanical strain magnitude. Calcif. Tissue Int. 37, 411–417 (1985)
L.E. Lanyon, C.T. Rubin, Static vs. dynamic loads as an influence on bone remodelling. J. Biomech. 17, 897–905 (1984)
T. Notomi, Y. Okazaki, N. Okimoto, S. Saitoh, T. Nakamura, M. Suzuki, A comparison of resistance and aerobic training for mass, strength and turnover of bone in growing rats. Eur. J. Appl. Physiol. 83, 469–474 (2000)
G. Radetti, W. Kleon, J. Stuefer, K. Pittschieler, Non-alcoholic fatty liver disease in obese children evaluated by magnetic resonance imaging. Acta Paediatr. 95, 833–837 (2006)
D.F. Chan, A.M. Li, W.C. Chu, M.H. Chan, E.M. Wong, E.K. Liu et al., Hepatic steatosis in obese Chinese children. Int. J. Obes. Relat. Metab. Disord. 28, 1257–1263 (2004)
N.K. Lee, H. Sowa, E. Hinoi, M. Ferron, J.D. Ahn, C. Confavreux et al., Endocrine regulation of energy metabolism by the skeleton. Cell 130, 456–469 (2007)
A. Afghani, M.L. Cruz, M.I. Goran, Impaired glucose tolerance and bone mineral content in overweight Latino children with a family history of type 2 diabetes. Diabetes Care 28, 372–378 (2005)
N.K. Pollock, P.J. Bernard, K. Wenger, S. Misra, B.A. Gower, J.D. Allison et al., Lower bone mass in prepubertal overweight children with pre-diabetes. J. Bone Miner. Res. 25, 2484–2493 (2010)
P. McNair, S. Madsbad, M.S. Christensen, C. Christiansen, O.K. Faber, C. Binder et al., Bone mineral loss in insulin-treated diabetes mellitus: studies on pathogenesis. Acta Endocrinol. 90, 463–472 (1979)
T. Miyata, K. Notoya, K. Yoshida, K. Horie, K. Maeda, K. Kurokawa et al., Advanced glycation end products enhance osteoclast-induced bone resorption in cultured mouse unfractionated bone cells and in rats implanted subcutaneously with devitalized bone particles. J. Am. Soc. Nephrol. 8, 260–270 (1997)
J. Argente, N. Caballo, V. Barrios, J. Pozo, M.T. Muñoz, J.A. Chowen et al., Multiple endocrine abnormalities of the growth hormone and insulin like growth factor axis in prepubertal children with exogenous obesity: effect of short- and long-term weight reduction. J. Clin. Endocrinol. Metab. 82, 2076–2083 (1997)
E. Hinoi, N. Gao, D.Y. Jung, V. Yadav, T. Yoshizawa, M.G. Myers Jr. et al., The sympathetic tone mediates leptin’s inhibition of insulin secretion by modulating osteocalcin bioactivity. J. Cell Biol. 183, 1235–1242 (2008)
M.W. Hamrick, S.L. Ferrari, Leptin and the sympathetic connection of fat to bone. Osteoporos. Int. 19, 905–912 (2008)
A. Martin, V. David, L. Malaval, M.H. Lafage-Proust, L. Vico, T. Thomas, Opposite effects of leptin on bone metabolism: a dose-dependent balance related to energy intake and insulin-like growth factor-I pathway. Endocrinology 148, 3419–3425 (2007)
Y. Okamoto, S. Kihara, N. Ouchi, M. Nishida, Y. Arita, M. Kumada et al., Adiponectin reduces atherosclerosis in apolipoprotein E-deficient mice. Circulation 106(22), 2767–2770 (2002)
R. Ouedraogo, Y. Gong, B. Berzins, X. Wu, K. Mahadev, K. Hough et al., Adiponectin deficiency increases leukocyte-endothelium interactions via upregulation of endothelial cell adhesion molecules in vivo. J. Clin. Invest. 117(6), 1718–1726 (2007)
A.R. Nawrocki, S.M. Hofmann, D. Teupser, J.E. Basford, J.L. Durand, L.A. Jelicks et al., Lack of association between adiponectin levels and atherosclerosis in mice. Arterioscler. Thromb. Vasc. Biol. 30(6), 115911–115965 (2010)
R.S. Lindsay, T. Funahashi, R.L. Hanson, Y. Matsuzawa, S. Tanaka, P.A. Tataranni et al., Adiponectin and development of type 2 diabetes in the Pima Indian population. Lancet 360(9326), 57–58 (2002)
C. Snehalatha, B. Mukesh, M. Simon, V. Viswanathan, S.M. Haffner, A. Ramachandran, Plasma adiponectin is an independent predictor of type 2 diabetes in Asian Indians. Diabetes Care 26(12), 3226–3229 (2003)
S. Li, H.J. Shin, E.L. Ding, R.M. van Dam, Adiponectin levels and risk of type 2 diabetes: a systematic review and meta-analysis. JAMA 302(2), 179–188 (2008)
N. Klöting, M. Fasshauer, A. Dietrich, P. Kovacs, M.R. Schön, M. Kern et al., Insulin-sensitive obesity. Am. J. Physiol. Endocrinol. Metab. 299(3), E506–E515 (2010)
V.Z. Rocha, E.J. Folco, Inflammatory concepts of obesity. Int. J. Inflam. 2011, 529061 (2011)
A.R. Dâmaso, W.L. do Prado, A. de Piano, L. Tock, D.A. Caranti, M.C. Lofrano et al., Relationship between nonalcoholic fatty liver disease prevalence and visceral fat in obese adolescents. Dig. Liver Dis. 40(2), 132–139 (2008)
Acknowledgments
AFIP, FAPESP, CNPQ, and CAPES supported the CEPE multidisciplinary obesity therapy. CENESP, FADA, FAPESP (2006/00684-3; 2008/53069-0; 2011/50356-0; 2011/50414-0), (CEPID/Sleep #9814303-3 st), UNIFESP. Special thanks to adolescents and their parents.
Conflict of interest
There exists no conflict of interest that could be perceived as having prejudice the impartiality of the research reported herein. This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sector.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Campos, R.M.S., de Piano, A., da Silva, P.L. et al. The role of pro/anti-inflammatory adipokines on bone metabolism in NAFLD obese adolescents: effects of long-term interdisciplinary therapy. Endocrine 42, 146–156 (2012). https://doi.org/10.1007/s12020-012-9613-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-012-9613-3