Abstract
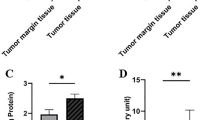
Lactate dehydrogenase (LDH), marker of anaerobic metabolism, is associated with highly invasive and metastatic breast cancer. Novel studies show that increased anaerobic metabolism (LDH), as well as activity of antioxidative enzymes (superoxide dismutase (SOD) and catalase (CAT)), is correlated with higher mammographic density, as known predictor of breast cancer risk. In this study, we measured LDH, MDH, and SOD activity in tumor and adjacent tissues of breast cancer patients by spectrophotometric assay. Mammograms were evaluated according to the American College of Radiology Breast Imaging Reporting and Data system. Mammographically dense breast tissue is associated with higher activity of LDH in tumor tissue of breast cancer patients. Moreover, patients with masses have significantly higher activity of LDH compared to patients with focal asymmetries or architectural distortion. Patients with spiculated mass margin had higher activity of LDH compared to patients with focal asymmetries or architectural distortion. Activity of LDH in patients significantly increases, while activity of CAT significantly decreases with the increase of BIRADS category. These results suggest that the association of activity of LDH and CAT in tumor tissue with mammographic characteristics could help in defining aggressive breast cancers.






Similar content being viewed by others
Abbreviations
- LDH:
-
Lactate dehydrogenase
- SOD:
-
Superoxide dismutase
- CAT:
-
Catalase
- HER2:
-
Human epidermal growth factor receptor 2
- DAB-3:
-
3Diaminobenzidine
- ER:
-
Estrogen receptor
- PR:
-
Progesterone receptor
References
Puliti, D., & Zappa, M. (2012). Breast cancer screening: are we seeing the benefit? BMC Medicne, 10, 106.
Boyd, N. F., Martin, L. J., Yaffe, M. J., & Minkin, S. (2011). Mammographic density and breast cancer risk: current understanding and future prospects. Breast Cancer Research, 13(6), 223.
Boyd, N. F., Melnichouk, O., Martin, L. J., Hislop, G., Chiarelli, A. M., Yaffe, M. J., et al. (2011). Mammographic density, response to hormones, and breast cancer risk. Journal of Clinical Oncology, 29(22), 2985–2992.
Boyd N.F. (2011) Tamoxifen, mammographic density, and breast cancer prevention. Journal of the National Cancer Institute, 103(9), 704–5.
Boyd, N. F., Martin, L. J., Bronskill, M., Yaffe, M. J., Duric, N., & Minkin, S. (2010). Breast tissue composition and susceptibility to breast cancer. Journal of the National Cancer Institute, 102(16), 1224–1237.
Boyd, N. F., & McGuire, V. (1990). Evidence of lipid peroxidation in premenopausal women with mammographic dysplasia. Cancer Letters, 50, 31–37.
Basu, A. K., & Marnett, L. J. (1983). Unequivocal demonstration that malondialdehyde is a mutagen. Carcinogenesis, 4, 331–333.
Jaganjac, M., Cacev, T., Cipak, A., Kapitanović, S., Gall Troselj, K., & Zarković, N. (2012). Even stressed cells are individuals: second messengers of free radicals in pathophysiology of cancer. Croatian Medical Journal, 53(4), 304–309.
Fantin, V. R., St-Pierre, J., & Leder, P. (2006). Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer Cell, 9, 425–434.
Zhou, M., Zhao, Y., Ding, Y., Liu, H., Liu, Z., Fodstad, O., et al. (2010). Warburg effect in chemosensitivity: targeting lactate dehydrogenase-A re-sensitizes taxol-resistant cancer cells to taxol. Molecular Cancer, 9, 33.
Konjević, G., Spuzić, I., & Jurisić, V. (2001). Association of NK cell dysfunction with changes in LDH characteristics of peripheral blood lymphocytes (PBL) in breast cancer patients. Breast Cancer Research and Treatment, 66(3), 255–263.
De Berardinis, R. J., Lum, J. J., Hatzivassiliou, G., & Thompson, C. B. (2008). The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metabolism, 7, 11–20.
Ursin, G., Hovanessian-Larsen, L., Parisky, Y. R., Pike, M. C., & Wu, A. H. (2005). Greatly increased occurrence of breast cancers in areas of mammographically dense tissue. Breast Cancer Research, 7(5), 605–608.
Filipits, M., Rudas, M., Jakesz, R., Dubsky, P., Fitzal, F., Singer, C. F., et al. (2011). A new molecular predictor of distant recurrence in ER-positive, HER2-negative breast cancer adds independent information to conventional clinical risk factors. Clinical Cancer Research, 17(18), 6012–6020.
Wang, Y., Ikeda, D. M., Narasimhan, B., Longacre, T. A., Bleicher, R. J., Pal, S., et al. (2008). Estrogen receptor-negative invasive breast cancer: imaging features of tumors with and without human epidermal growth factor receptor type 2 overexpression. Radiology, 246(2), 367–375.
Stankovic, S., Konjevic, G., Gopcevic, K., Jovic, V., Inic, M., & Jurisic, V. (2010). Activity of MMP-2 and MMP-9 in sera of breast cancer patients. Pathology, Research and Practice, 206(4), 241–247.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 1976(72), 248–254.
Van der Helm, H. J. (1962). Interference of the measurement of lactate dehydrogenase (LDH) activity in human serum and plasma by LDH from blood cells. Clinica Chimica Acta, 1962(7), 124–128.
Cunningham, V. R., Phillips, J., & Field, E. J. (1965). Lactic dehydrogenase isoenzymes in normal and pathological spinal fluids. Journal of Clinical Pathology, 18(6), 765–770.
Weydert, C. J., & Cullen, J. J. (2010). Measurement of superoxide dismutase, catalase and glutathione peroxidase in cultured cells and tissue. Nature Protocols, 5(1), 51–66.
Burnside, E. S., Sickles, E. A., Bassett, L. W., Rubin, D. L., Lee, C. H., Ikeda, D. M., et al. (2009). The ACR BI-RADS experience: learning from history. Journal American College Radiology, 6(12), 851–860.
Boyd, N. F., Martin, L. J., Yaffe, M., & Minkin, S. (2009). Mammographic density. Breast Cancer Research, 11(3), S4.
Boyd, N. F., Rommens, J. M., Vogt, K., Lee, V., Hopper, J. L., Yaffe, M. J., et al. (2005). Mammographic breast density as an intermediate phenotype for breast cancer. Lancet Oncology, 6(10), 798–808.
Fu, L., Tsuchiya, S., Matsuyama, I., & Ishii, K. (1998). Clinicopathologic features and incidence of invasive lobular carcinoma in Japanese women. Pathology International, 48, 348–354.
Seo, B. K., Pisano, E. D., Kuzimak, C. M., Koomen, M., Pavic, D., Lee, Y., et al. (2006). Correlation of HER-2/neu overexpression with mammography and age distribution in primary breast carcinomas. Academic Radiology, 13(10), 1211–1218.
Tabar, L., Tony Chen, H.H., Amy Yen, M.F., Tot, T., Tung, T.H., Chen, L.S. et al. (2004) Mammographic tumor features can predict long-term outcomes reliably in women with 1-14-mm invasive breast carcinoma. Cancer, 101(8), 1745–59.
Habel, L. A., Dignam, J. J., Land, S. R., Salane, M., Capra, A. M., & Julian, T. B. (2004). Mammographic density and breast cancer after ductal carcinoma in situ. Journal of the National Cancer Institute, 96, 1467–1472.
Dang, C. V., & Semenza, G. L. (1999). Oncogenic alterations of metabolism. Trends in Biochemical Sciences, 24(2), 68–72.
Hong, C. C., Tang, B. K., Rao, V., Agarwal, S., Martin, L., Tritchler, D., et al. (2004). Cytochrome P450 1A2 (CYP1A2) activity, mammographic density, and oxidative stress: a cross-sectional study. Breast Cancer Research, 6(4), R338–R351.
Tsanou, E., Ioachim, E., Briasoulis, E., Damala, K., Charchanti, A., Karavasilis, V., et al. (2004). Immunohistochemical expression of superoxide dismutase (MnSOD) anti-oxidant enzyme in invasive breast carcinoma. Histology and Histopathology, 19(3), 807–813.
Kattan, Z., Minig, V., Leroy, P., Dauça, M., & Becuwe, P. (2008). Role of manganese superoxide dismutase on growth and invasive properties of human estrogen-independent breast cancer cells. Breast Cancer Research and Treatment, 108(2), 203–215.
Acknowledgments
This work was supported by the grant No. 175056 of the Ministry of Science and Technology of the Republic of Serbia. We wish to thank Prof. Dr. M. Susa for suggestions and help during this investigation. Also, we wish to thank Mrs. Jasna Popovic-Basic and Mrs. Marijana Topalovic for excellent technical work during this investigation and Mrs. Dusica Gavrilovic for extensive statistical analyses.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Radenkovic, S., Milosevic, Z., Konjevic, G. et al. Lactate dehydrogenase, Catalase, and Superoxide dismutase in Tumor Tissue of Breast Cancer Patients in Respect to Mammographic Findings. Cell Biochem Biophys 66, 287–295 (2013). https://doi.org/10.1007/s12013-012-9482-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-012-9482-7